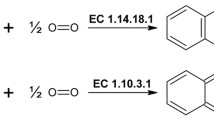
Abstract
A new tyrosinase-encoding gene (2,204 bp) and the corresponding cDNA (1,857 nucleotides) from the white-rot fungus Pycnoporus sanguineus BRFM49 were cloned. This gene consisted of seven exons and six introns and encoded a predicted protein of 68 kDa, exceeding the mature tyrosinase by 23 kDa. P. sanguineus tyrosinase cDNA was over-expressed in Aspergillus niger, a particularly suitable fungus for heterologous expression of proteins of biotechnological interest, under the control of the glyceraldehyde-3-phosphate-dehydrogenase promoter as strong and constitutive promoter. The glucoamylase preprosequence of A. niger was used to target the secretion. This construction enabled the production of recombinant tyrosinase in the extracellular medium of A. niger. The identity of the purified recombinant protein was confirmed by N-terminal amino acid sequencing. The maturation process was shown to be effective in A. niger, and the recombinant enzyme was fully active, with a molecular mass of 45 kDa. The best transformant obtained, A. niger D15#26-e, produced extracellular tyrosinase activities of 534 and 1,668 U l−1 for monophenolase and diphenolase, respectively, which corresponded to a protein yield of ca. 20 mg l−1.



Similar content being viewed by others
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSIBLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Balance DJ (1986) Sequences important for gene expression in filamentous fungi. Yeast 2:229–236
Breathnach R, Benoist C, O'Hara K, Chambon P (1978) Ovalbumin gene: evidence for a leader sequence in mRNA and DNA sequences at the exon–intron boundaries. Proc Natl Acad Sci U S A 75:4853–4857
Della Cioppa GR, Garger SJ, Holtz RB, McCulloch MJ, Sverlow GG (1996) Method for making stable, extracellular tyrosinase and synthesis of polyphenolic polymers therefrom. US Patent 5,486,351
Duran N, Esposito E (2000) Potential applications of oxidative enzymes and phenoloxidase-like compounds in wastewater and soil treatment: a review. Appl Catal, B Environ 28:83–99
Ebbelar CEM, Wichers HJ, van den Bosch T, Oyevaar JI, Recourt K (1995) URL http://www.ncbi.nlm.nih.gov/entrez/query.fcgi
Espin JC, Van Leeuwen J, Wichers HJ (1999) Kinetic study of the activation process of a latent mushroom (Agaricus bisporus) tyrosinases by serine proteases. J Agric Food Chem 47:3509–3517
Fujita Y, Uraga Y, Ichisima E (1995) Molecular cloning and nucleotide sequence of the prototyrosinase gene, melO, from Aspergillus oryzae and expression of the gene in yeast cells. Biochim Biophys Acta 1261:151–154
Garcia-Borron JC, Solano F (2002) Molecular anatomy of tyrosinase and its related proteins: beyond the histine-bound metal catalytic center. Pigment Cell Res 15:162–173
Gerdemann C, Christoph E, Krebs B (2002) The crystal structure of catechol oxidase: new insight into the function of type-3 copper proteins. Acc Chem Res 35:183–191
Gordon CL, Khalaj V, Ram AFJ, Archer DB, Brookman JL, Trinci APJ, Jeenes DJ, Doonan JH, Wells B, Punt PJ, van den Hondel CAMJJ, Robson GD (2000) Glucoamylase: fluorescent protein fusions to monitor protein secretion in Aspergillus niger. Microbiology 146:415–426
Gouka RJ, Punt PJ, van den Hondel CAMJJ (1997) Efficient production of secreted proteins by Aspergillus niger: progress, limitations and prospects. Appl Microbiol Biotechnol 47:1–11
Halaouli S, Asther M, Kruus K, Guo L, Hamdi M, Sigoillot J-C, Asther M, Lomascolo A (2005) Characterization of a new tyrosinase from Pycnoporus species with high potential for food technological applications. J Appl Microbiol 98:332–343
Haq I, Ali S, Qadeer MA (2002) Biosynthesis of L-DOPA by Aspergillus oryzae. Bioresour Technol 85:25–29
Herpoël I, Moukha S, Lesage-Meessen L, Sigoillot J-C, Asther M (2000) Selection of Pycnoporus cinnabarinus strains for laccase production. FEMS Microbiol Lett 183:301–306
Jolivet S, Arpin N, Wichers HJ, Pellon G (1998) Agaricus bisporus browning: a review. Mycol Res 102:1459–1483
Kanda K, Sato T, Ishii S, Enei H, Ejiri SI (1996) Purification and properties of tyrosinase isozymes from the gill of Lentinus edodes fruiting body. Biosci Biotechnol Biochem 60:1273–1278
Kanda S, Aimi T, Masumoto S, Nakano K, Kitamoto Y, Morigana T (2004) NCBI Database. URL http://www.ncbi.nlm.nih.gov/entrez/query.fcgi
Kupper U, Niedermann DM, Travaglini G, Lerch K (1989) Isolation and characterization of the tyrosinase gene from Neurospora crassa. J Biol Chem 264:17250–17258
Kupper U, Linden ML, Cao K, Lerch K (1990) Expression of tyrosinase in vegetative cultures of Neurospora crassa transformed with a metallothionein promoter/protyrosinase fusion gene. Curr Genet 18:331–335
Kurahashi Y, Pontzen R (1998) Carpropamid: a new melanin biosynthesis inhibitor. Pflanzenschutz-Nachr Bayer 51:245–256
Langfelder K, Glaser P, Brakhage AA (2003) NCBI Database. URL http://www.ncbi.nlm.nih.gov/entrez/query.fcgi
Lantto R, Niku-Paavola ML, Schonberg C, Buchert J (2002) A tyrosinase enzyme. International Patent WO 02/14484A1
Lomascolo A, Cayol J-L, Roche M, Guo L, Robert J-L, Record E, Lesage-Meessen L, Ollivier B, Sigoillot J-C, Asther M (2002) Molecular clustering of Pycnoporus strains from various geographic origins and isolation of monokaryotic strains for laccase hyperproduction. Mycol Res 106:1193–1203
Lomascolo A, Record E, Herpoël-Gimbert I, Delattre M, Robert J-L, Georis J, Dauvrin T, Sigoillot J-C, Asther M (2003) Overproduction of laccase by a monokaryotic strain of Pycnoporus cinnabarinus using ethanol as inducer. J Appl Microbiol 94:618–624
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Marbach K, Stahl U (1996) NCBI Database. URL http://www.ncbi.nlm.nih.gov/entrez/query.fcgi
Nakamura M, Nakajima T, Ohba Y, Yamauchi S, Lee BR, Ichishima E (2000) Identification of copper ligands in Aspergillus oryzae tyrosinase by site-directed mutagenesis. Biochem J 350:537–545
Otterbein L, Record E, Longhi S, Asther M, Moukha S (2000) Molecular cloning of the cDNA encoding laccase from Pycnoporus cinnabarinus I-937 and expression in Pichia pastoris. Eur J Biochem 267:1619–1625
Punt PJ, van den Hondel CA (1992) Transformation of filamentous fungi based on hygromycin B and phleomycin resistance markers. Methods Enzymol 216:447–457
Record E, Moukha S, Asther M, Asther M (2001) Cloning and expression in phospholipid containing cultures of the gene encoding the specific phosphatidylglycerol/phosphatidylinositol transfer protein from Aspergillus oryzae: evidence that the pg/pi-tp is tandemly arranged with the putative 3-ketoacyl-CoA thiolase gene. Gene 10:61–72
Record E, Punt PJ, Chamkha M, Labat M, van den Hondel CAMJJ, Asther M (2002) Expression of the Pycnporus cinnabarinus laccase gene in Aspergillus niger and characterization of the recombinant enzyme. Eur J Biochem 269:602–609
Reese MG (2001) Application of a time-dealy neural network to promoter annotation in the Drosophila melanogaster genome. Comput Chem 26:51–56
Riley PA (1997) Molecules in focus: melanin. Int J Biochem Cell Biol 29:1235–1239
Robb DA, Gutteridge S (1981) Polypeptide composition of two fungal tyrosinases. Phytochemistry 20:1481–1485
Sato K (2002) URL http://www.ncbi.nlm.nih.gov/entrez/query.fcgi
Schulte U, Align V, Hoheisel J, Brandt P, Fartmann B, Holland R, Nyakatura G, Mewes HW, Mannhaupt G (2003) NCBI Database. URL http://www.ncbi.nlm.nih.gov/entrez/query.fcgi
Seo SY, Sharma VK, Sharma N (2003) Mushroom tyrosinase: recent prospects. J Agric Food Chem 51:2837–2853
Soler-Rivas C, Jolivet S, Arpin N, Olivier JM, Wichers HJ (1999) Biochemical and physiological aspects of brown blotch disease of Agaricus bisporus. FEMS Microbiol Rev 23:591–614
Thalmann CR, Lötzbeyer T (2002) Enzymatic cross-linking of proteins with tyrosinase. Eur Food Res Technol 214:276–281
Van Gelder CWG, Flurkey WH, Wichers HJ (1997) Sequence and structural features of plant and fungal tyrosinases. Phytochemistry 45:1309–1323
van Hartingsveldt W, Mattern IE, van Zeijl CM, Pouwels PH, van den Hondel CA (1987) Development of homologous transformation system for Aspergillus niger based on the pyrG gene. Mol Gen Genet 206:71–75
Wessels JGH, Mulder GH, Springer J (1987) Expression of dikaryon specific and non-specific mRNAs of Schizophyllum commune in relation to environmental conditions and fruiting. J Gen Microbiol 133:2557–2561
Wichers HJ, Gerritsen YA, Chapelon CGJ (1996) Tyrosinase isoforms from the fruitbodies of Agaricus bisporus. Phytochemistry 43:333–337
Wichers HJ, Recourt K, Hendriks M, Ebbelaar CE, Biancone G, Hoeberichts FA, Mooibroek H, Soler-Rivas C (2003) Cloning, expression, and characterisation of two tyrosinase cDNAs from Agaricus bisporus. Appl Microbiol Biotechnol 61:336–341
Acknowledgements
We warmly thank K. Kruus (VTT, Finland) for the internal tyrosinase peptides determination, P. Mansuelle (UMR 6560, IFR Jean Roche, Marseille, France) for the N-terminal amino sequence analysis, D. Navarro for technical assistance, S. Pagès (CNRS, Marseille, France) and J.-L. Cayol (IRD, Marseille, France) for helpful comments on molecular biology engineering. This work has been carried out with financial support from the Commission of the European Communities, specific RTD programme “Quality of Life and management of Living Resources”, proposal number QLK1-2002-02208, “Novel cross-linking enzymes and their consumer acceptance for structure engineering of foods”, acronym CROSSENZ. It does not reflect its views and in no way anticipates the Commission's future policy in this area.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Halaouli, S., Record, E., Casalot, L. et al. Cloning and characterization of a tyrosinase gene from the white-rot fungus Pycnoporus sanguineus, and overproduction of the recombinant protein in Aspergillus niger . Appl Microbiol Biotechnol 70, 580–589 (2006). https://doi.org/10.1007/s00253-005-0109-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-005-0109-4