Abstract
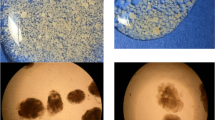
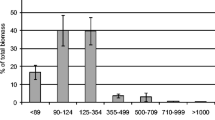
Activated sludge and acetate-fed granules were used as microbial inocula to start up two sequencing batch reactors (R1, R2) for phenol biodegradation. The reactors were operated in 4-h cycles at a phenol loading of 1.8 kg m−3 day−1. The biomass in R1 failed to remove phenol and completely washed out after 4 days. R2 experienced initial difficulty in removing phenol, but the biomass acclimated quickly and effluent phenol concentrations declined to 0.3 mg l−1 from day 3. The acetate-fed granules were covered with bacterial rods, but filamentous bacteria with sheaths, presumably to shield against toxicity, quickly emerged as the dominant morphotype upon phenol exposure. Bacterial adaptation to phenol also took the form of modifications in enzyme activity and increased production of extracellular polymers. 16S rRNA gene fingerprints revealed a slight decrease in bacterial diversity from day 0 to day 3 in R1, prior to process failure. In R2, a clear shift in community structure was observed as the seed evolved into phenol-degrading granules without losing species-richness. The results highlight the effectiveness of granules over activated sludge as seed for reactors treating toxic wastewaters.






Similar content being viewed by others
References
APHA (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, Washington, D.C.
Beun JJ, Hendriks A, Van Loosdrecht MCM, Morgenroth E, Wilderer PA, Heijnen JJ (1999) Aerobic granulation in a sequencing batch reactor. Water Res 33:2283–2290
Brummer IHM, Felske A, Wagner-Dobler I (2003) Diversity and seasonal variability of beta-proteobacteria in biofilms of polluted rivers: analysis by temperature gradient gel electrophoresis and cloning. Appl Environ Microbiol 69:4463–4473
Caravelli A, Contreras EM, Giannuzzi L, Zaritzky N (2003) Modeling of chlorine effect on floc forming and filamentous micro-organisms of activated sludges. Water Res 37:2097–2105
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Eichner CA, Erb RW, Timmis KN, Wagner-Döbler I (1999) Thermal gradient gel electrophoresis analysis of bioprotection from pollutant shocks in the activated sludge microbial community. Appl Environ Microbiol 65:102–109
Fang HHP, Xu LC, Chan KY (2002) Effects of toxic metals and chemicals on biofilm and biocorrosion. Water Res 36:4709–4716
Fedorak PM, Hrudey SE (1988) Anaerobic degradation of phenolic compounds with application to treatment of industrial waste waters. In: Wise DL (ed) Biotreatment systems, vol 1. CRC, Boca Raton, pp 170–212
Filonov AE, Duetz WA, Karpov AV, Gaiazov RR, Kosheleva IA, Breure AM, Filonova IF, Andel JG van, Boronin AM (1997) Competition of plasmid-bearing Pseudomonas putida strains catabolizing naphthalene via various pathways in chemostat culture. Appl Microbiol Biotechnol 48:493–498
Flemming HC, Wingender J (2001) Relevance of microbial extracellular polymeric substances (EPSs)—part I: structural and ecological aspects. Water Sci Technol 43:1–8
Jenkins D, Richard MG, Daigger GT (1993) Manual on the causes and control of activated sludge bulking and foaming, 2nd edn. Lewis, Boca Raton
Jiang HL, Tay JH, Tay STL (2002) Aggregation of immobilized activated sludge cells into aerobically grown microbial granules for the aerobic biodegradation of phenol. Lett Appl Microbiol 35:439–445
Jiang HL, Tay JH, Tay STL (2004) Changes in structure, activity and metabolism of aerobic granules as a microbial response to high phenol loading. Appl Microbiol Biotechnol 63:602–608
Kiesel B, Müller RH (2002) The meta pathway as a potential energy-generating sequence and its effects on the growth rate during the utilization of aromatics. Acta Biotechnol 22:221–234
Martins AMP, Pagilla K, Heijnen JJ, Van Loosdrecht MCM (2004) Filamentous bulking sludge—a critical review. Water Res 38:793–817
Morgenroth E, Sherden T, Van Loosdrecht MCM, Heijnen JJ, Wilderer PA (1997) Aerobic granular sludge in a sequencing batch reactor. Water Res 31:3191–3194
Moy BYP, Tay JH, Toh SK, Liu Y, Tay STL (2002) High organic loading influences the physical characteristics of aerobic granules. Lett Appl Microbiol 34:407–412
Müller RH, Babel W (1996) Growth rate-dependent expression of phenol-assimilation pathways in Alcaligenes eutrophus JMP 134-the influence of formate as an auxiliary energy source on phenol conversion characteristics. Appl Microbiol Biotechnol 46:156–162
Muyzer G, De Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Neumann G, Teras R, Monson L, Kivisaar M, Schauer F, Heipieper HJ (2004) Simultaneous degradation of atrazine and phenol by Pseudomonas sp strain ADP: effects of toxicity and adaptation. Appl Environ Microbiol 70:1907–1912
Pernelle JJ, Gaval G, Cotteux E, Duchene P (2001) Influence of transient substrate overloads on the proliferation of filamentous bacterial populations in an activated sludge pilot plant. Water Res 35:129–134
Stoscheck CM (1990) Quantitation of protein. Methods Enzymol 182:50–69
Sutherland IW (2001) Biofilm exopolysaccharides: a strong and sticky framework. Microbiology 147:3–9
Tay STL, Ivanov V, Yi S, Zhuang WQ, Tay JH (2002) Presence of anaerobic Bacteroides in aerobically grown microbial granules. Microb Ecol 44:278–285
Watanabe K, Teramoto M, Harayama S (1999) An outbreak of nonflocculation catabolic populations caused the breakdown of a phenol-digesting activated sludge process. Appl Environ Microbiol 65:2813–2819
Watanabe K, Teramoto M, Harayama S (2002) Stable augmentation of activated sludge with foreign catabolic genes harboured by an indigenous dominant bacterium. Environ Microbiol 4:577–583
Wingender J, Neu TR, Flemming HC (1999) What are bacterial extracellular polymeric substances. In: Wingender J, Neu TR, Flemming HC (eds) Microbial extracellular polymeric substances: characterization, structure and function. Springer, Berlin Heidelberg New York, pp 1–19
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tay, S.TL., Moy, B.YP., Maszenan, A.M. et al. Comparing activated sludge and aerobic granules as microbial inocula for phenol biodegradation. Appl Microbiol Biotechnol 67, 708–713 (2005). https://doi.org/10.1007/s00253-004-1858-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-004-1858-1