Abstract
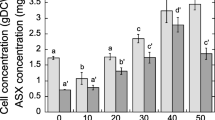
Natural carbon sources, such as those present in cane sugar molasses and grape juice, promote the synthesis of astaxanthin in different Phaffia rhodozyma yeasts. One of these, coconut milk, has a very rich nutrient composition. The aim of this work was to investigate the utility of coconut milk as sole source of energy for astaxanthin pigment production by P. rhodozyma strains. Currently, coconut pulp is widely used in industrial processes in Mexico for the production of shampoos, candies, food, etc. However, coconut milk is a waste product. We show that coconut milk enhances astaxanthin production. The fermentation yielded 850 μg/g yeast with the NRRL-10921 wild-type strain and 1,850 μg/g yeast with the mutated R1 strain. Production was better than reported results employing other natural carbon sources.




Similar content being viewed by others
References
Acheampong E, Martin A (1995) Kinetic studies on the yeast Phaffia rhodozyma. J Basic Microbiol 35:147–155
An GH, Schuman DB, Johnson EA (1989) Isolation of Phaffia rhodozyma mutants with increased astaxanthin content. Appl Environ Microbiol 55:116–124
An GH, Bielich J, Auerbach R, Johnson EA (1991) Isolation and characterization of carotenoid hyperproducing mutants of yeast by flow cytometry and cell sorting. Biotechnology 9:70–73
Bramley PM, Mackenzie A (1988) Regulation of carotenoid biosynthesis. In: Horecker BL, Stadtman ER (eds) Current topics in cellular regulation, vol 29. Academic, San Diego
Calo PT, De Miguel T, Velázquez JB, Villa TJ (1995) Mevalonic acid increases trans-astaxanthin and biosynthesis in Phaffia rhodozyma. Biotechnol Lett 17(6):575–578
Fontana JD, Guimaraes MF, Martins NT, Fontana CA, Baron M (1996) Culture of the astaxanthin ogenic yeast Phaffia rhodozyma in low-cost media. Appl Biochem Biotechnol 57–58:413–422
Haard NF (1988) Astaxanthin formation by the yeast Phaffia rhodozyma on molasses. Biotechnol Lett 10:609–614
Hayman TG, Mannarelli BN, Leathers TD (1995) Production of carotenoids by Phaffia rhodozyma grown on media composed of corn wet-milling co-products. J Ind Microbiol 115:173–183
Jian-Ping Y, Xian-Di G, Feng C (1997) Separation and analysis of carotenoids and chlorophylls in Haematococcus lacustris by high-performance liquid chromatography photodiode array detection. J Agric Food Chem 45:1952–1956
Jiménez R (1999) Obtención de una mutante de Phaffia rhodozyma hiperproductora de astaxanthin. Maters in Sc. Thesis (in Spanish). Departamento de Biotecnología y Bioingeniería, CINVESTAV-IPN, México
Johnson EA, An GH (1991) Astaxanthin from microbial sources. Crit Rev Biotechnol 11:297–326
Lewis MJ, Ragot N, Berlant MC, Miranda M (1990) Selection of astaxanthin-overproducing mutants of Phaffia rhodozyma with β-ionone. Appl Environ Microbiol 56:2944–2945
Martin AM, Acheampong E, Patel TR (1993) Production of astaxanthin by Phaffia rhodozyma using peat hydrolysates as substrate. J Chem Tech Biotechnol 58:223–230
Meyer PS, du Preez JC (1994) Astaxanthin production by Phaffia rhodozyma mutant on grape juice. World J Microbiol Biotechnol 10:178–183
Meyer PS, du Preez JC, Kilian SG (1993) Selection and evaluation of astaxanthin-overproducing mutants of Phaffia rhodozyma. World J Microbiol Biotechnol 9:514–520
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Okagbue RN, Lewis MW (1984) Autolysis of the red yeast Phaffia rhodozyma: a potential tool to facilitate extraction of astaxanthin. Biotechnol Lett 6:247
Olaizola M (2000) Commercial production of astaxanthin from Haematococcus pluvialis using 25,000-liter outdoor photobioreactors. J Appl Phycol 12:499–506
Sedmak JJ, Weerasinghe DK, Jolly SO (1990) Extraction and quantitation of astaxanthin by Phaffia rhodozyma. Biotechnol Tech 4:107–112
Sokal RR, Rohlf FJ (1981) Biometry. The principles and practice of statistics in biological research, 2nd edn. Freeman, San Francisco
Yamane Y, Higashida K, Nishio N (1997) Influence of oxygen and glucose on primary metabolism and astaxanthin production by Phaffia rhodozyma in Fed-Bath cultures. Kinetic and stoichiometric analysis. Appl Environ Microbiol 63:4471–4478
Acknowledgement
We would like to express our thanks to Dr. T. Ponce-Noyola for providing the mutated R1 yeast.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Domínguez-Bocanegra, A.R., Torres-Muñoz, J.A. Astaxanthin hyperproduction by Phaffia rhodozyma (now Xanthophyllomyces dendrorhous) with raw coconut milk as sole source of energy. Appl Microbiol Biotechnol 66, 249–252 (2004). https://doi.org/10.1007/s00253-004-1686-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-004-1686-3