Abstract
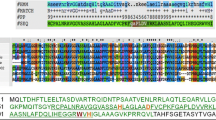
To determine the function of the C-terminal region of Bacillus amyloliquefaciens phage endolysin on Pseudomonas aeruginosa lysis, the permeabilization of the outer membrane of P. aeruginosa was analyzed. Glu-15 to His (E15H) and Thr-32 to Glu (T32E) substitutions were introduced into the Bacillus phage endolysin. Neither E15H nor T32E substitution induced enzymatic and antibacterial activities. These two, Glu-15 and Thr-32, were considered to be the active center of the enzyme. The addition of purified E15H and T32E proteins to P. aeruginosa cells induced the release of periplasmic β-lactamase from the cells, indicating that both proteins enhance permeabilization of the outer membrane. However, the addition of E15H and T32E proteins to P. aeruginosa cells did not induce the release of cytoplasmic ATP from the cells. These results indicate that the antibacterial activity of the endolysin requires both the C-terminal enhancement of the permeabilization of the P. aeruginosa outer membrane and N-terminal enzymatic activity.




Similar content being viewed by others
References
Angus BL, Carey AM, Caron DA, Kropinski AMB, Hancock REW (1982) Outer membrane permeability in Pseudomonas aeruginosa: comparison of a wild-type with an antibiotic-supersusceptible mutant. Antimicrob Agents Chemother 21:299–309
Bebrone C, Moali C, Mahy F, Rival S, Docquier JD, Rossolini GM, Jacques, F, Pratt RF, Frere JM, Galleni M (2001) CENTA as a chromogenic substrate for studying β-lactamase. Antimicrob Agents Chemother 45:1868–1871
During K, Porsch P, Mahn A, Brinkmann O, Gieffers W (1999) The non-enzymatic microbicidal activity of lysozyme. FEBS Lett 449:93–100
Hancock REW (1997) The bacterial outer membrane as a drug barrier. Trends Microbiol 5:37–42
Hancock REW (2001) Cationic peptides: effectors in innate immunity and novel antimicrobials. Lancet Infect Dis 1:156–164
Hancock REW, Scott MG (2000) The role of antimicrobial peptides in animal defenses. Proc Natl Acad Sci U S A 97:8856–8861
Hancock REW, Rozek A (2002) Role of membrane in the activities of antimicrobial cationic peptides. FEMS Microbiol Lett 206:143–149
Kuroki R, Weaver LH, Matthews BW (1999) Structural basis of the conversion of T4 lysozyme into a transglycosidase by reengineering the active site. Proc Natl Acad Sci U S A 96:8949–8954
Loeffler JM, Nelson D, Fischetti VA (2001) Rapid killing of Streptococcus pneumoniae with a bacteriophage cell wall hydrolase. Science 294:2170–2172
Morita M, Tanji Y, Mizoguchi K, Soejima A, Orito Y, Unno H (2001a) Antibacterial activity of Bacillus amyloliquefaciens phage endolysin without holin conjugation. J Biosci Bioeng 88:221–225
Morita M, Tanji Y, Orito Y, Mizoguchi K, Soejima A, Unno H (2001b) Functional analysis of antibacterial activity of Bacillus amyloliquefaciens phage endolysin against Gram-negative bacteria. FEBS Lett 500:56–59
Nelson D, Loomis L, Fischetti VA (2001) Prevention and elimination of upper respiratory colonization of mice by group A streptococci by using a bacteriophage lytic enzyme. Proc Natl Acad Sci U S A 98:4107–4112
Schuch R, Nelson D, Fischetti VA (2002) A bacteriolytic agent that detects and kills Bacillus anthracis. Nature 418:884–889
Trias J, Dufresne J, Levesque RC, Nikaido H (1989) Decrease outer membrane permeability in imipenem-resistant mutants of Pseudomonas aeruginosa. Antimicrob Agents Chemother 33:1201–1206
Weisner B (1984) Esterases, glycosidases, lyases, ligases. In: Bergmeyer J, Grabl M (eds) Methods of enzymatic analysis, 3rd edn, vol IV. Verlag Chemie, Weinheim, Deerfield Beach Fla., pp. 189–194
Zhang L, Rozek A, Hancock REW (2001) Interaction of cationic antimicrobial peptides with model membranes. J Biol Chem 276:35714–35722
Acknowledgement
This research was financially supported in part by a grant (13450340) from the Japanese Ministry of Education, Sports, Science, and Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Orito, Y., Morita, M., Hori, K. et al. Bacillus amyloliquefaciens phage endolysin can enhance permeability of Pseudomonas aeruginosa outer membrane and induce cell lysis. Appl Microbiol Biotechnol 65, 105–109 (2004). https://doi.org/10.1007/s00253-003-1522-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-003-1522-1