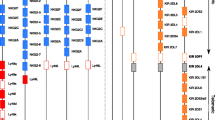
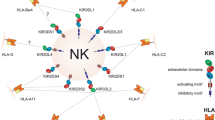
Abstract
Natural killer (NK) cells are fast-acting and versatile lymphocytes that are critical effectors of innate immunity, adaptive immunity, and placental development. Controlling NK cell function are the interactions between killer-cell immunoglobulin-like receptors (KIRs) and their HLA-A, HLA-B and HLA-C ligands. Due to the extensive polymorphism of both KIR and HLA class I, these interactions are highly diversified and specific combinations correlate with protection or susceptibility to a range of infectious, autoimmune, and reproductive disorders. Evolutionary, genetic, and functional studies are consistent with the interactions between KIR and HLA-C being the dominant control mechanism of human NK cells. In addition to their recognition of the C1 and C2 epitopes, increasing evidence points to KIR having a previously unrecognized selectivity for the peptide presented by HLA-C. This selectivity appears to be a conserved feature of activating KIR and may partly explain the slow progress made in identifying their HLA class I ligands. The peptide selectivity of KIR allows NK cells to respond, not only to changes in the surface expression of HLA-C, but also to the more subtle changes in the HLA-C peptidome, such as occur during viral infection and malignant transformation. Here, we review recent advances in understanding of human-specific KIR evolution and how the inhibitory and activating HLA-C receptors allow NK cells to respond to healthy cells, diseased cells, and the semi-allogeneic cells of the fetus.


Similar content being viewed by others
References
Abi-Rached L, Parham P (2005) Natural selection drives recurrent formation of activating killer cell immunoglobulin-like receptor and Ly49 from inhibitory homologues. J Exp Med 201(8):1319–1332
Abi-Rached L et al (2010) Human-specific evolution and adaptation led to major qualitative differences in the variable receptors of human and chimpanzee natural killer cells. PLoS Genet 6(11):e1001192
Adams EJ, Parham P (2001) Species-specific evolution of MHC class I genes in the higher primates. Immunol Rev 183:41–64
Alter G et al (2011) HIV-1 adaptation to NK-cell-mediated immune pressure. Nature 476(7358):96–100
Aranda-Romo S et al (2012) Killer-cell immunoglobulin-like receptors (KIR) in severe A (H1N1) 2009 influenza infections. Immunogenetics 64(9):653–662
Arase H et al (2002) Direct recognition of cytomegalovirus by activating and inhibitory NK cell receptors. Science 296(5571):1323–1326
Ayo CM et al (2015) Killer cell immunoglobulin-like receptors and their HLA ligands are related with the immunopathology of Chagas disease. PLoS Negl Trop Dis 9(5):e0003753
Ayo CM et al (2016) Ocular toxoplasmosis: susceptibility in respect to the genes encoding the KIR receptors and their HLA class I ligands. Sci Report 6:36632
Bari R et al (2009) Significant functional heterogeneity among KIR2DL1 alleles and a pivotal role of arginine 245. Blood 114(25):5182–5190
Beltrame LM et al (2013) Influence of KIR genes and their HLA ligands in susceptibility to dengue in a population from southern Brazil. Tissue Antigens 82(6):397–404
Besson C et al (2007) Association of killer cell immunoglobulin-like receptor genes with Hodgkin’s lymphoma in a familial study. PLoS One 2(5):e406
Bettencourt A et al (2014) The role of KIR2DS1 in multiple sclerosis—KIR in Portuguese MS patients. J Neuroimmunol 269(1–2):52–55
Bimber BN, Evans DT (2015) The killer-cell immunoglobulin-like receptors of macaques. Immunol Rev 267(1):246–258
Bimber BN et al (2008) Complete characterization of killer Ig-like receptor (KIR) haplotypes in Mauritian cynomolgus macaques: novel insights into nonhuman primate KIR gene content and organization. J Immunol 181(9):6301–6308
Blokhuis JH et al (2010) The mosaic of KIR haplotypes in rhesus macaques. Immunogenetics 62(5):295–306
Blokhuis JH et al (2017) KIR2DS5 allotypes that recognize the C2 epitope of HLA-C are common among Africans and absent from Europeans. Immunity, Inflammation and Disease, In Press.
Bonagura VR et al (2010) Activating killer cell immunoglobulin-like receptors 3DS1 and 2DS1 protect against developing the severe form of recurrent respiratory papillomatosis. Hum Immunol 71(2):212–219
Borhis G et al (2013) A peptide antagonist disrupts NK cell inhibitory synapse formation. J Immunol 190(6):2924–2930
Boyington JC, Sun PD (2002) A structural perspective on MHC class I recognition by killer cell immunoglobulin-like receptors. Mol Immunol 38(14):1007–1021
Boyington JC et al (2000) Crystal structure of an NK cell immunoglobulin-like receptor in complex with its class I MHC ligand. Nature 405(6786):537–543
Braud VM et al (1998) HLA-E binds to natural killer cell receptors CD94/NKG2A, B and C. Nature 391(6669):795–799
Cassidy S et al (2015) Peptide selectivity discriminates NK cells from KIR2DL2- and KIR2DL3-positive individuals. Eur J Immunol 45(2):492–500
Chandran V et al (2014) Killer-cell immunoglobulin-like receptor gene polymorphisms and susceptibility to psoriatic arthritis. Rheumatology (Oxford) 53(2):233–239
Colonna M et al (1993) HLA-C is the inhibitory ligand that determines dominant resistance to lysis by NK1- and NK2-specific natural killer cells. Proc Natl Acad Sci U S A 90(24):12000–12004
Diaz-Pena R et al (2015) Activating killer immunoglobulin-like receptors genes are associated with increased susceptibility to ankylosing spondylitis. Clin Exp Immunol 180(2):201–206
Estefania E et al (2007) Influence of KIR gene diversity on the course of HSV-1 infection: resistance to the disease is associated with the absence of KIR2DL2 and KIR2DS2. Tissue Antigens 70(1):34–41
Fadda L et al (2010) Peptide antagonism as a mechanism for NK cell activation. Proc Natl Acad Sci U S A 107(22):10160–10165
Fan QR, Long EO, Wiley DC (2001) Crystal structure of the human natural killer cell inhibitory receptor KIR2DL1-HLA-Cw4 complex. Nat Immunol 2(5):452–460
Fusco C et al (2010) KIRs and their HLA ligands in remitting-relapsing multiple sclerosis. J Neuroimmunol 229(1–2):232–237
Garcia KC, Adams EJ (2005) How the T cell receptor sees antigen—a structural view. Cell 122(3):333–336
Gendzekhadze K et al (2009) Co-evolution of KIR2DL3 with HLA-C in a human population retaining minimal essential diversity of KIR and HLA class I ligands. Proc Natl Acad Sci U S A 106(44):18692–18697
Gonzalez-Galarza FF et al (2011) Allele frequency net: a database and online repository for immune gene frequencies in worldwide populations. Nucleic Acids Res 39(Database issue):D913–D919
Goodridge JP et al (2013) HLA-F and MHC class I open conformers are ligands for NK cell Ig-like receptors. J Immunol 191(7):3553–3562
Graef T et al (2009) KIR2DS4 is a product of gene conversion with KIR3DL2 that introduced specificity for HLA-A*11 while diminishing avidity for HLA-C. J Exp Med 206(11):2557–2572
Guerini FR et al (2012) Activating KIR/HLA complexes in classic Kaposi’s Sarcoma. Infect Agent Cancer 7:9
Guethlein LA et al (2007) Evolution of killer cell Ig-like receptor (KIR) genes: definition of an orangutan KIR haplotype reveals expansion of lineage III KIR associated with the emergence of MHC-C. J Immunol 179(1):491–504
Guethlein LA et al (2015) Co-evolution of MHC class I and variable NK cell receptors in placental mammals. Immunol Rev 267(1):259–282
Hiby SE et al (2004) Combinations of maternal KIR and fetal HLA-C genes influence the risk of preeclampsia and reproductive success. J Exp Med 200(8):957–965
Hiby SE et al (2008) Association of maternal killer-cell immunoglobulin-like receptors and parental HLA-C genotypes with recurrent miscarriage. Hum Reprod 2008; 23 (4): 972-976. doi:10.1093/humrep/den011
Hiby SE et al (2010) Maternal activating KIRs protect against human reproductive failure mediated by fetal HLA-C2. J Clin Invest 120(11):4102–4110
Hiby SE et al (2014) Maternal KIR in combination with paternal HLA-C2 regulate human birth weight. J Immunol 192(11):5069–5073
Hilton HG et al (2012) Mutation at positively selected positions in the binding site for HLA-C shows that KIR2DL1 is a more refined but less adaptable NK cell receptor than KIR2DL3. J Immunol 189(3):1418–1430
Hilton HG et al (2015a) Polymorphic HLA-C receptors balance the functional characteristics of KIR haplotypes. J Immunol 195(7):3160–3170
Hilton HG et al (2015b) Loss and gain of natural killer cell receptor function in an African hunter-gatherer population. PLoS Genet 11(8):e1005439
Hilton HG et al (2017a) Resurrecting KIR2DP1: a key intermediate in the evolution of human inhibitory NK cell receptors that recognize HLA-C. J Immunol 198(5):1961–1973
Hilton HG et al. (2017b) The intergenic recombinant HLA-B*46:01 has a distinctive peptidome which includes KIR2DL3 ligands. Cell Reports 19(7):1394–1405
Holm SJ et al (2005) Distinct HLA-C/KIR genotype profile associates with guttate psoriasis. J Invest Dermatol 125(4):721–730
Holzemer A et al (2015) Selection of an HLA-C*03:04-restricted HIV-1 p24 gag sequence variant is associated with viral escape from KIR2DL3+ natural killer cells: data from an observational cohort in South Africa. PLoS Med 12(11):e1001900 discussion e00
Horowitz A et al. (2016) Class I HLA haplotypes form two schools that educate NK cells in different ways. Sci Immunol 1(3)
Hou L et al (2010a) African Americans exhibit a predominant allele in the midst of extensive KIR2DL1 allelic diversity. Tissue Antigens 76(1):31–34
Hou YF et al (2010b) Disparate distribution of activating and inhibitory killer cell immunoglobulin-like receptor genes in patients with systemic lupus erythematosus. Lupus 19(1):20–26
Hou Y et al (2015) Association of killer cell immunoglobulin-like receptor and human leucocyte antigen-Cw gene combinations with systemic lupus erythematosus. Clin Exp Immunol 180(2):250–254
Jarduli LR et al (2014) Influence of KIR genes and their HLA ligands in the pathogenesis of leprosy in a hyperendemic population of Rondonopolis, Southern Brazil. BMC Infect Dis 14:438
Jiao YL et al (2008) Polymorphisms of KIRs gene and HLA-C alleles in patients with ankylosing spondylitis: possible association with susceptibility to the disease. J Clin Immunol 28(4):343–349
Jobim M et al (2008) A study of the killer cell immunoglobulin-like receptor gene KIR2DS1 in a Caucasoid Brazilian population with psoriasis vulgaris. Tissue Antigens 72(4):392–396
Kennedy PR et al (2016) Activating KIR2DS4 is expressed by uterine NK cells and contributes to successful pregnancy. J Immunol 197(11):4292–4300
Khakoo SI et al (2004) HLA and NK cell inhibitory receptor genes in resolving hepatitis C virus infection. Science 305(5685):872–874
King A et al (2000) Surface expression of HLA-C antigen by human extravillous trophoblast. Placenta 21(4):376–387
Kruse PH, Rosner C, Walter L (2010) Characterization of rhesus macaque KIR genotypes and haplotypes. Immunogenetics 62(5):281–293
Li JT et al (2016) Killer cell immunoglobulin-like receptor genes and their Hla-C ligands in Hashimoto thyroiditis in a Chinese population. Endocr Pract 22(8):935–940
Littera R et al (2016) Exploring the role of killer cell immunoglobulin-like receptors and their HLA class I ligands in autoimmune hepatitis. PLoS One 11(1):e0146086
Liu J et al (2014) Activating killer cell immunoglobulin-like receptor 2DS2 binds to HLA-A*11. Proc Natl Acad Sci U S A 111(7):2662–2667
Lu C et al (2012) Association of killer cell immunoglobulin-like receptors with pulmonary tuberculosis in Chinese Han. Genet Mol Res 11(2):1370–1378
Luszczek W et al (2004) Gene for the activating natural killer cell receptor, KIR2DS1, is associated with susceptibility to psoriasis vulgaris. Hum Immunol 65(7):758–766
Malnati MS et al (1995) Peptide specificity in the recognition of MHC class I by natural killer cell clones. Science 267(5200):1016–1018
Malnati MS et al (2017) Activating killer immunoglobulin receptors and HLA-C: a successful combination providing HIV-1 control. Sci Report 7:42470
Martin MP et al (2002) Cutting edge: susceptibility to psoriatic arthritis: influence of activating killer Ig-like receptor genes in the absence of specific HLA-C alleles. J Immunol 169(6):2818–2822
Miyashita R et al (2006) Association of killer cell immunoglobulin-like receptor genotypes with microscopic polyangiitis. Arthritis Rheum 54(3):992–997
Moesta AK et al (2008) Synergistic polymorphism at two positions distal to the ligand-binding site makes KIR2DL2 a stronger receptor for HLA-C than KIR2DL3. J Immunol 180(6):3969–3979
Moesta AK et al (2010) Humans differ from other hominids in lacking an activating NK cell receptor that recognizes the C1 epitope of MHC class I. J Immunol 185(7):4233–4237
Moffett A, Colucci F (2015) Co-evolution of NK receptors and HLA ligands in humans is driven by reproduction. Immunol Rev 267(1):283–297
Moffett-King A (2002) Natural killer cells and pregnancy. Nat Rev Immunol 2(9):656–663
Momot T et al (2004) Association of killer cell immunoglobulin-like receptors with scleroderma. Arthritis Rheum 50(5):1561–1565
Nakimuli A et al (2015) A KIR B centromeric region present in Africans but not Europeans protects pregnant women from pre-eclampsia. Proc Natl Acad Sci U S A 112(3):845–850
Niepieklo-Miniewska W et al (2013) Protective effect of the KIR2DS1 gene in atopic dermatitis. Gene 527(2):594–600
Nikitina-Zake L et al (2004) Killer cell immunoglobulin-like receptor genes in Latvian patients with type 1 diabetes mellitus and healthy controls. Ann N Y Acad Sci 1037:161–169
Nourse JP et al (2012) The KIR2DS2/DL2 genotype is associated with adult persistent/chronic and relapsed immune thrombocytopenia independently of FCGR3a-158 polymorphisms. Blood Coagul Fibrinolysis 23(1):45–50
Nowak I et al (2015) KIR2DS5 in the presence of HLA-C C2 protects against endometriosis. Immunogenetics 67(4):203–209
Older Aguilar AM et al (2010) Coevolution of killer cell Ig-like receptors with HLA-C to become the major variable regulators of human NK cells. J Immunol 185(7):4238–4251
Older Aguilar AM et al (2011) Natural variation at position 45 in the D1 domain of lineage III killer cell immunoglobulin-like receptors (KIR) has major effects on the avidity and specificity for MHC class I. Immunogenetics 63(8):543–547
Oliveira LM et al (2017) Reduced frequency of two activating KIR genes in patients with sepsis. Hum Immunol 78(4):363–369
Parham P, Moffett A (2013) Variable NK cell receptors and their MHC class I ligands in immunity, reproduction and human evolution. Nat Rev Immunol 13(2):133–144
Parham P et al (2010) Primate-specific regulation of natural killer cells. J Med Primatol 39(4):194–212
Pellett F et al (2007) KIRs and autoimmune disease: studies in systemic lupus erythematosus and scleroderma. Tissue Antigens 69(Suppl 1):106–108
Pende D et al (2009) Anti-leukemia activity of alloreactive NK cells in KIR ligand-mismatched haploidentical HSCT for pediatric patients: evaluation of the functional role of activating KIR and redefinition of inhibitory KIR specificity. Blood 113(13):3119–3129
Peruzzi M et al (1996) Peptide sequence requirements for the recognition of HLA-B*2705 by specific natural killer cells. J Immunol 157(8):3350–3356
Pydi SS et al (2013) Killer cell immunoglobulin like receptor gene association with tuberculosis. Hum Immunol 74(1):85–92
Pyo C-W, Guethlein LA, Vu Q, Wang R, Abi-Rached L, Norman PJ, Marsh SGE, Miller JS, Parham P, Geraghty DE, Matsunami H (2010) Different patterns of evolution in the centromeric and telomeric regions of group A and B haplotypes of the human killer cell ig-Like receptor locus. PLoS ONE 5(12):e15115
Rajagopalan S, Long EO (1997) The direct binding of a p58 killer cell inhibitory receptor to human histocompatibility leukocyte antigen (HLA)-Cw4 exhibits peptide selectivity. J Exp Med 185(8):1523–1528
Ramirez-De los Santos S et al (2012) Associations of killer cell immunoglobulin-like receptor genes with rheumatoid arthritis. Dis Markers 33(4):201–206
Salim PH et al (2010) Killer cell immunoglobulin-like receptor (KIR) genes in systemic sclerosis. Clin Exp Immunol 160(3):325–330
Saulquin X, Gastinel LN, Vivier E (2003) Crystal structure of the human natural killer cell activating receptor KIR2DS2 (CD158j). J Exp Med 197(7):933–938
Saunders PM et al (2015) A bird’s eye view of NK cell receptor interactions with their MHC class I ligands. Immunol Rev 267(1):148–166
Seymour LA et al (2014) The presence of KIR2DS5 confers protection against adult immune thrombocytopenia. Tissue Antigens 83(3):154–160
Sheereen A et al (2011) A study of KIR genes and HLA-C in Vogt-Koyanagi-Harada disease in Saudi Arabia. Mol Vis 17:3523–3528
Sim MJ et al (2017) Canonical and cross-reactive binding of NK cell inhibitory receptors to HLA-C allotypes is dictated by peptides bound to HLA-C. Front Immunol 8:193
Smith HR et al (2002) Recognition of a virus-encoded ligand by a natural killer cell activation receptor. Proc Natl Acad Sci U S A 99(13):8826–8831
Stewart CA et al (2005) Recognition of peptide-MHC class I complexes by activating killer immunoglobulin-like receptors. Proc Natl Acad Sci U S A 102(37):13224–13229
Storkus WJ et al (1992) Peptide-induced modulation of target cell sensitivity to natural killing. J Immunol 149(4):1185–1190
Suzuki Y et al (2004) Genetic polymorphisms of killer cell immunoglobulin-like receptors are associated with susceptibility to psoriasis vulgaris. J Invest Dermatol 122(5):1133–1136
Thiruchelvam-Kyle L et al. (2017) The activating human NK cell receptor KIR2DS2 recognizes a beta2-microglobulin-independent ligand on cancer cells. J Immunol
Uhrberg M et al (1997) Human diversity in killer cell inhibitory receptor genes. Immunity 7(6):753–763
Valiante NM et al (1997) Functionally and structurally distinct NK cell receptor repertoires in the peripheral blood of two human donors. Immunity 7(6):739–751
van der Ploeg K et al (2017) Modulation of human leukocyte antigen-C by human cytomegalovirus stimulates KIR2DS1 recognition by natural killer cells. Front Immunol 8:298
van der Slik AR et al (2003) KIR in type 1 diabetes: disparate distribution of activating and inhibitory natural killer cell receptors in patients versus HLA-matched control subjects. Diabetes 52(10):2639–2642
van Teijlingen NH et al (2014) Sequence variations in HIV-1 p24 Gag-derived epitopes can alter binding of KIR2DL2 to HLA-C*03:04 and modulate primary natural killer cell function. AIDS 28(10):1399–1408
VandenBussche CJ et al (2006) A single polymorphism disrupts the killer Ig-like receptor 2DL2/2DL3 D1 domain. J Immunol 177(8):5347–5357
Vilches C et al (2000) KIR2DL5, a novel killer-cell receptor with a D0-D2 configuration of Ig-like domains. J Immunol 164(11):5797–5804
Wauquier N et al (2010) Association of KIR2DS1 and KIR2DS3 with fatal outcome in Ebola virus infection. Immunogenetics 62(11–12):767–771
Williams F et al (2005) Activating killer cell immunoglobulin-like receptor gene KIR2DS1 is associated with psoriatic arthritis. Hum Immunol 66(7):836–841
Wilson MJ et al (2000) Plasticity in the organization and sequences of human KIR/ILT gene families. Proc Natl Acad Sci U S A 97(9):4778–4783
Winter CC et al (1998) Direct binding and functional transfer of NK cell inhibitory receptors reveal novel patterns of HLA-C allotype recognition. J Immunol 161(2):571–577
Xiong S et al (2013) Maternal uterine NK cell-activating receptor KIR2DS1 enhances placentation. J Clin Invest 123(10):4264–4272
Yen JH et al (2001) Major histocompatibility complex class I-recognizing receptors are disease risk genes in rheumatoid arthritis. J Exp Med 193(10):1159–1167
Zappacosta F et al (1997) Peptides isolated from HLA-Cw*0304 confer different degrees of protection from natural killer cell-mediated lysis. Proc Natl Acad Sci U S A 94(12):6313–6318
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published in the Special Issue MHC Genes and Their Ligands in Health and Disease with Editor Prof. Ronald Bontrop.
Rights and permissions
About this article
Cite this article
Hilton, H.G., Parham, P. Missing or altered self: human NK cell receptors that recognize HLA-C. Immunogenetics 69, 567–579 (2017). https://doi.org/10.1007/s00251-017-1001-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-017-1001-y