Abstract
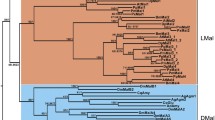
The βGRP/GNBP/β-1,3-glucanase protein family of insects includes several proteins involved in innate immune recognition, such as the β-glucan recognition proteins of Lepidoptera and the Gram-negative bacteria-binding proteins of Drosophila. A phylogenetic analysis supported the existence of two distinct subfamilies, designated the pattern recognition receptor (PRR) and glucanase subfamilies, which originated by gene duplication prior to the origin of the Holometabola. In the C-terminal region (CTR) shared by both subfamilies, the PRR subfamily has evolved significantly more rapidly at the amino acid sequence level than has the glucanase subfamily, implying a relative lack of constraint on the amino acid sequence of this region in the PRR subfamily. PRR subfamily members also include an N-terminal region (NTR), involved in carbohydrate recognition, which is not shared by glucanase subfamily members. In comparisons between paralogous PRR subfamily members, there were no conserved amino acid residues in the NTR. However, when pairs of putatively orthologous PRR subfamily members were compared, the NTR was most often as conserved as the CTR or more so. This pattern suggests that the NTR may be important in functions specific to the different paralogs, while amino acid sequence changes in the NTR may have been important in functional differentiation among paralogs, specifically with regard to the types of carbohydrates that they recognize.


Similar content being viewed by others
References
Aggrawal K, Silverman N (2007) Peptidoglycan recognition in Drosophila. Biochem Sci Trans 35:1496–1500
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Bragatto I, Genta FA, Ribeiro AF, Terra WR, Ferreira C (2010) Characterization of a β-1,3-glucanase active in the alkaline midgut of Spodoptera frugiperda larvae and its relation to β-glucan-binding proteins. Insect Biochem Mol Biol 40:861–872
Bulmer MS, Crozier R (2006) Variation in positive selection in termite GNBPs and relish. Mol Biol Evol 23:317–326
Edgecombe GD (2010) Arthropod phylogeny: an overview from the perspectives of morphology, molecular data and the fossil record. Arthropod Struct Devel 39:74–87
Ferrandon D, Imler J-L, Hoffman JA (2004) Sensing infection in Drosophila: Toll and beyond. Semin Immunol 16:43–53
Filipe SR, Tomasz A, Ligoxygakis P (2005) Requirements of peptidoglycan structure that allow detection by the Drosophila Toll pathway. EMBO Rep 6:327–333
Filipe SR, Gay NJ, Ligoxygakis P (2006) Sensing of Gram-positive bacteria in Drosophila: GNBP1 is needed to process and present peptidoglycan to PGRP-SA. EMBO J 25:5005–5014
Genta FA, Bragatto I, Terra WR, Ferreira C (2009) Purification, characterization and sequencing of the major β-1,3-glucanase from the midgut of Tenebrio molitor larvae. Insect Biochem Mol Biol 39:861–874
Gobert V, Gottar M, Matskevich AA, Rutschmann S, Royet J, Belvin M, Hoffmann JA, Ferrandon D (2003) Dual activation of the Drosophila Toll pathway by two pattern recognition receptors. Science 302:2126–2130
Gottar M, Gobert V, Matskevich AA, Reichhart JM, Wang C, Butt TM, Belvin M, Hoffmann JA, Ferrandon D (2006) Dual detection of fungal infections in Drosophila via recognition of glucans and sensing of virulence factors. Cell 127:1425–1437
Giribet G, Edgecombe GD, Wheeler WC (2001) Arthropod phylogeny based on eight molecular loci and morphology. Nature 413:157–161
Grimaldi D, Engel MS (2005) Evolution of the insects. Cambridge University Press, Cambridge
Hamilton C, Lay F, Bulmer MS (2011) Subteranean termite prophylactic secretions and external antifungal defenses. J Insect Physiol 57:1259–1266
Hoffman JA, Katafos FC, Janeway CA, Ezekowitz RA (1999) Phylogenetic perspectives in innate immunity. Science 284:1313–1318
Hughes AL (2007) Looking for Darwin in all the wrong places: the misguided quest for positive selection at the molecular level. Heredity 99:364–373
Hughes AL, Friedman R (2005) Variation in the pattern of synonymous and nonsynonymous difference between two fungal genomes. Mol Biol Evol 22:1320–1324
Hughes AL, Friedman R (2008) Codon-based tests of positive selection, branch lengths, and the evolution of mammalian immune system genes. Immunogenetics 60:495–506
Kanost MR, Jiang H, Yu X-Q (2004) Innate immune responses of a lepidopteran insect, Manduca sexta. Immunol Rev 198:97–105
Lee WJ, Lee JD, Kravchenko VV, Ulevitch RJ, Brey PT (1996) Purification and molecular cloning of an inducible gram-negative bacteria-binding protein from the silkworm, Bombyx mori. Proc Natl Acad Sci USA 93:7888–7893
Lemaitre B, Hoffmann J (2007) The host defense of Drosophila melanogaster. Annu Rev Immunol 25:697–743
Matskevich AA, Quintin J, Ferrandon D (2010) The Drosophila PRR GNBP3 assembles effector complexes involved in antifungal defenses independently of its Toll-pathway activation function. Eur J Immunol 40:1244–1254
Mishima Y, Quintin J, Aimanianda V, Kellenberger C, Coste F, Clavaud C, Hetru C, Hoffmann JA, Latgé J-P, Ferrandon D, Roussel A (2009) The N-terminal domain of Drosophila Gram-negative binding protein 3 (GNBP3) defines a novel family of fungal pattern recognition receptors. J Biol Chem 284:28687–28697
Nei M, Kumar S (2000) Molecular evolution and phylogenetics. Oxford University Press, New York
Ochiai M, Ashida M (1988) Purification of a beta-1,3-glucan recognition protein in the prophenoloxidase activating system from hemolymph of the silkworm, Bombyx mori. J Biol Chem 263:12056–12062
Ochiai M, Ashida M (2000) A pattern-recognition protein for beta-1,3-glucan. The binding domain and the cDNA cloning of beta-1,3-glucan recognition protein from the silkworm, Bombyx mori. J Biol Chem 275:4995–5002
Ochiai M, Niki T, Ashida M (1992) Immunocytochemical localization of beta-1,3-glucan recognition protein in the silkworm, Bombyx mori. Cell Tissue Res 268:431–437
Park J-W, Kim C-H, Kim J-H, Je B-R, Roh K-B, Kim S-J, Le H-H, Ryu J-H, Lim J-H, Oh B-H, Lee W-J, Ha W-J, Lee B-L (2007) Clustering of peptidoglycan recognition protein-SA is required for sensing lysine-type peptidoglycan in insects. Proc Natl Acad Sci USA 104:6602–6607
Pauchet Y, Freitak D, Heidel-Fischer HM, Heckel DG, Vogel H (2009) Immunity or digestion: glucanase activity in a glucan-binding protein family from Lepidoptera. J Biol Chem 284:2214–2224
Pili-Floury S, Leulier F, Takahashi K, Saigo K, Samain E, Ueda R, Lemaitre B (2004) In vivo RNA interference analysis reveals an unexpected role for GNBP1 in the defense against Gram-positive bacterial infection in Drosophila adults. J Biol Chem 279:12848–12853
Regier JC, Shultz JW, Ganley AR, Hussey A, Shi D, Ball B, Zwick A, Stajich JE, Cummings MP, Martin JW, Cunningham CW (2008) Resolving arthropod phylogeny: exploring phylogenetic signal within 41 kb of protein-coding nuclear gene sequence. Syst Biol 57:920–938
Royet J (2004) Infectious non-self recognition in invertebrates: lessons from Drosophila and other insect models. Mol Immunol 41:1063–1075
Swaminathan CP, Brown PH, Roychowdhury A, Wang Q, Guan R, Silverman N, Goldman WE, Boons G-J, Mariuzza RA (2006) Dual strategies for peptidoglycan discrimination by peptidoglycan recognition proteins (PGRPs). Proc Natl Acad Sci USA 103:684–689
Takahasi K, Ochiai M, Horiuchi M, Kumeta H, Ogura K, Ashida M, Inagaki F (2009) Solution structure of the silkworm βGRP/GNBP3 N-terminal domain reveals the mechanism for β-1,3-glucan-specific recognition. Proc Natl Acad Sci USA 106:11679–11684
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: Molecular Evolutionary Genetics Analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Diggins DG (1997) The CLUSTAL X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Viljakainen L, Evans JD, Hasselmann M, Rueppell O, Tngek S, Pamilo P (2009) Rapid evolution of immune proteins in social insects. Mol Biol Evol 26:1791–1801
Wang L, Ligoxygakis O (2006) Pathogen recognition and signalling in the Drosophila innate immune response. Immunobiol 211:251–261
Wang L, Weber AN, Atilano ML, Filipe SR, Gay NJ, Ligoxygakis P (2006) Sensing of Gram-positive bacteria in Drosophila: GNBP1 is needed to process and present peptidoglycan to PGRP-SA. EMBO J 25:5005–5014
Wiegmann BM, Trautwein MD, Kim J-W, Cassel BK, Bertone MA, Winterton SL, Yeates DK (2009) Single-copy nuclear genes resolve the phylogeny of the holometabolous insects. BMC Biol 7:34
Zhang R, Cho HY, Kim HS, Ma YG, Osaki T, Kawabata S, Söderhäll K, Lee BL (2003) Characterization and properties of a 1,3-β-D_glucan pattern recognition protein of Tenebrio molitor larvae that is specifically degraded by serine protease during prophenoloxidase activation. J Biol Chem 278:42072–42079
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Supplementary Figure S1
Alignment of 90 sequences the βGRPs/GNBP/β-1,3-glucanase family (PDF 97 kb)
Supplementary Figure S2
ME tree based on the JTT + G distance (a = 1.35) at 171 aligned sites. Values on branches are confidence levels of interior branch test (PDF 16 kb)
Supplementary Figure S3
ME tree of hemolymph subfamily members from Diptera and Lepidoptera, based on the JTT + G distance (a = 1.51) at 326 aligned sites. Values on branches are confidence levels of interior branch test (PDF 47 kb)
Rights and permissions
About this article
Cite this article
Hughes, A.L. Evolution of the βGRP/GNBP/β-1,3-glucanase family of insects. Immunogenetics 64, 549–558 (2012). https://doi.org/10.1007/s00251-012-0610-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-012-0610-8