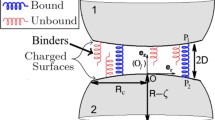
Abstract
Two-dimensional (2D) kinetics of receptor–ligand interactions governs cell adhesion in many biological processes. While the dissociation kinetics of receptor–ligand bond is extensively investigated, the association kinetics has much less been quantified. Recently receptor–ligand interactions between two surfaces were investigated using a thermal fluctuation assay upon biomembrane force probe technique (Chen et al. in Biophys J 94:694–701, 2008). The regulating factors on association kinetics, however, are not well characterized. Here we developed an alternative thermal fluctuation assay using optical trap technique, which enables to visualize consecutive binding–unbinding transition and to quantify the impact of microbead diffusion on receptor–ligand binding. Three selectin constructs (sLs, sPs, and PLE) and their ligand P-selectin glycoprotein ligand 1 were used to conduct the measurements. It was indicated that bond formation was reduced by enhancing the diffusivity of selectin-coupled carrier, suggesting that carrier diffusion is crucial to determine receptor–ligand binding. It was also found that 2D forward rate predicted upon first-order kinetics was in the order of sPs > sLs > PLE and bond formation was history-dependent. These results further the understandings in regulating association kinetics of surface-bound receptor–ligand interactions.






Similar content being viewed by others
References
Albelda SM (1993) Biology of disease—role of integrins and other cell-adhesion molecules in tumor progression and metastasis. Lab Invest 68:4–17
Alon R, Hammer DA, Springer TA (1995) Lifetime of the P-selectin-carbohydrate bond and its response to tensile force in hydrodynamic flow. Nature 374:539–542. doi:10.1038/374539a0
Bell GI (1978) Models for the specific adhesion of cells to cells. Science 200:618–627. doi:10.1126/science.347575
Bell GI (1981) Estimate of the sticking probability for cells in uniform shear flow with adhesion caused by specific bonds. Cell Biophys 3:289–304. doi:10.1007/BF02782629
Chang KC, Hammer DA (1999) The forward rate of binding of surface-tethered reactants: effect of relative motion between two surfaces. Biophys J 76:1280–1292. doi:10.1016/S0006-3495(99)77291-7
Chen S, Alon R, Fuhlbrigge RC, Springer TA (1997) Rolling and transient tethering of leukocytes on antibodies reveal specializations of selectins. Proc Natl Acad Sci USA 94:3172–3177. doi:10.1073/pnas.94.7.3172
Chen W, Evans EA, McEver RP, Zhu C (2008) Monitoring receptor–ligand interactions between surfaces by thermal fluctuations. Biophys J 94:694–701. doi:10.1529/biophysj.107.117895
Chesla SE, Selvaraj P, Zhu C (1998) Measuring two-dimensional receptor-ligand binding kinetics by micropipette. Biophys J 75:1553–1572. doi:10.1016/S0006-3495(98)74074-3
Dammer U, Hegner M, Anselmetti D, Wagner P, Dreier M, Huber W et al (1996) Specific antigen/antibody interactions measured by force microscopy. Biophys J 70:2437–2441. doi:10.1016/S0006-3495(96)79814-4
Dustin ML, Ferguson LM, Chan PY, Springer TA, Golan DE (1996) Visualization of CD2 interaction with LFA-3 and determination of the two-dimensional dissociation constant for adhesion receptors in a contact area. J Cell Biol 132:465–474. doi:10.1083/jcb.132.3.465
Evans CW, Proctor J (1978) A collision analysis of lymphoid cell aggregation. J Cell Sci 33:17–36
Evans E, Ritchie K (1997) Dynamic strength of molecular adhesion bonds. Biophys J 72:1541–1555. doi:10.1016/S0006-3495(97)78802-7
Evans E, Leung A, Hammer D, Simon S (2001) Chemically distinct transition states govern rapid dissociation of single l-selectin bonds under force. Proc Natl Acad Sci USA 98:3784–3789. doi:10.1073/pnas.061324998
Finger EB, Puri KD, Alon R, Lawrence MB, von Andrian UH, Springer TA (1996) Adhesion through l-selectin requires a threshold hydrodynamic shear. Nature 379:266–269. doi:10.1038/379266a0
Florin EL, Moy VT, Gaub HE (1994) Adhesion forces between individual ligand–receptor pairs. Science 264:415–417. doi:10.1126/science.8153628
Fritz J, Katopodis AG, Kolbinger F, Anselmetti D (1998) Force-mediated kinetics of single P-selectin ligand complexes observed by atomic force microscopy. Proc Natl Acad Sci USA 95:12283–12288. doi:10.1073/pnas.95.21.12283
Galkina E, Ley K (2007) Vascular adhesion molecules in atherosclerosis. Arterioscl Throm Vas 27:2292–2301. doi:10.1161/ATVBAHA.107.149179
Gelles J, Schnapp BJ, Sheetz MP (1988) Tracking kinesin-driven movements with nanometre-scale precision. Nature 331:450–453. doi:10.1038/331450a0
Geng JG, Bevilacqua MP, Moore KL, McIntyre TM, Prescott SM, Kim JM et al (1990) Rapid neutrophil adhesion to activated endothelium mediated by GMP-140. Nature 343:757–760. doi:10.1038/343757a0
Grinnell F (1992) Wound repair, keratinocyte activation and integrin modulation. J Cell Sci 101:1–5
Hammer DA, Lauffenburger DA (1987) A dynamical model for receptor-mediated cell adhesion to surfaces. Biophys J 52:475–487. doi:10.1016/S0006-3495(87)83236-8
Huang J, Chen J, Chesla SE, Yago T, Mehta P, McEver RP et al (2004) Quantifying the effects of molecular orientation and length on two-dimensional receptor–ligand binding kinetics. J Biol Chem 279:44915–44923. doi:10.1074/jbc.M407039200
Ishijima A, Harada Y, Kojima H, Funatsu T, Higuchi H, Yanagida T (1994) Single-molecule analysis of the actomyosin motor using nano-manipulation. Biochem Biophys Res Commun 199:1057–1063. doi:10.1006/bbrc.1994.1336
Kaplanski G, Farnarier C, Tissot O, Pierres A, Benoliel AM, Alessi MC et al (1993) Granulocyte endothelium initial adhesion—analysis of transient binding events mediated by E-selectin in a laminar shear-flow. Biophys J 64:1922–1933. doi:10.1016/S0006-3495(93)81563-7
Kulin S, Kishore R, Hubbard JB, Helmerson K (2002) Real-time measurement of spontaneous antigen–antibody dissociation. Biophys J 83:1965–1973. doi:10.1016/S0006-3495(02)73958-1
Kuo SC, Sheetz MP (1993) Force of single kinesin molecules measured with optical tweezers. Science 260:232–234. doi:10.1126/science.8469975
Kusumi A, Sako Y, Yamamoto M (1993) Confined lateral diffusion of membrane-receptors as studied by single-particle tracking (Nanovid microscopy)—effects of calcium-induced differentiation in cultured epithelial-cells. Biophys J 65:2021–2040. doi:10.1016/S0006-3495(93)81253-0
Levin JD, Ting-Beall HP, Hochmuth RM (2001) Correlating the kinetics of cytokine-induced E-selectin adhesion and expression on endothelial cells. Biophys J 80:656–667. doi:10.1016/S0006-3495(01)76046-8
Ley K (2003) The role of selectins in inflammation and disease. Trends Mol Med 9:263–268. doi:10.1016/S1471-4914(03)00071-6
Li F, Erickson HP, James JA, Moore KL, Cummings RD, McEver RP (1996) Visualization of P-selectin glycoprotein ligand-1 as a highly extended molecule and mapping of protein epitopes for monoclonal antibodies. J Biol Chem 271:6342–6348. doi:10.1074/jbc.271.11.6342
Long M, Zhao H, Huang KS, Zhu C (2001) Kinetic measurements of cell surface E-selectin/carbohydrate ligand interactions. Ann Biomed Eng 29:935–946. doi:10.1114/1.1415529
Lü SQ, Ye ZY, Zhu C, Long M (2006) Quantifying the effects of contact duration, loading rate, and approach velocity on P-selectin–PSGL-1 interactions using AFM. Polymer (Guildf) 47:2539–2547. doi:10.1016/j.polymer.2005.11.095
Marshall BT, Sarangapani KK, Lou JH, McEver RP, Zhu C (2005) Force history dependence of receptor–ligand dissociation. Biophys J 88:1458–1466. doi:10.1529/biophysj.104.050567
Mehta P, Patel KD, Laue TM, Erickson HP, McEver RP (1997) Soluble monomeric P-selectin containing only the lectin and epidermal growth factor domains binds to P-selectin glycoprotein ligand-1 on leukocytes. Blood 90:2381–2389
Merkel R, Nassoy P, Leung A, Ritchie K, Evans E (1999) Energy landscapes of receptor–ligand bonds explored with dynamic force spectroscopy. Nature 397:50–53. doi:10.1038/16219
Moore KL, Eaton SF, Lyons DE, Lichenstein HS, Cummings RD, McEver RP (1994) The P-selectin glycoprotein ligand from human neutrophils displays sialylated, fucosylated, O-linked poly-N-acetyllactosamine. J Biol Chem 269:23318–23327
Neuman KC, Block SM (2004) Optical trapping. Rev Sci Instrum 75:2787–2809. doi:10.1063/1.1785844
Paschall CD, Guilford WH, Lawrence MB (2008) Enhancement of L-selectin, but not P-selectin, bond formation frequency by convective flow. Biophys J 94:1034–1045. doi:10.1529/biophysj.106.098707
Peterman EJG, Gittes F, Schmidt CF (2003) Laser-induced heating in optical traps. Biophys J 84:1308–1316. doi:10.1016/S0006-3495(03)74946-7
Pierres A, Benoliel AM, Bongrand P, vander Merwe PA (1997) The dependence of the association rate of surface-attached adhesion molecules CD2 and CD48 on separation distance. FEBS Lett 403:239–244. doi:10.1016/S0014-5793(97)00060-4
Pierres A, Feracci H, Delmas V, Benoliel AM, Thiery JP, Bongrand P (1998) Experimental study of the interaction range and association rate of surface-attached cadherin 11. Proc Natl Acad Sci USA 95:9256–9261. doi:10.1073/pnas.95.16.9256
Pierres A, Benoliel AM, Zhu C, Bongrand P (2001) Diffusion of microspheres in shear flow near a wall: use to measure binding rates between attached molecules. Biophys J 81:25–42. doi:10.1016/S0006-3495(01)75677-9
Rinko LJ, Lawrence MB, Guilford WH (2004) The molecular mechanics of P- and L-selectin lectin domains binding to PSGL-1. Biophys J 86:544–554. doi:10.1016/S0006-3495(04)74133-8
Shao JY, Xu JB (2002) A modified micropipette aspiration technique and its application to tether formation from human neutrophils. J Biomech Eng-T ASME 124:388–396. doi:10.1115/1.1486469
Springer TA (1995) Traffic signals on endothelium for lymphocyte recirculation and leukocyte emigration. Annu Rev Physiol 57:827–872. doi:10.1146/annurev.ph.57.030195.004143
Svoboda K, Schmidt CF, Schnapp BJ, Block SM (1993) Direct observation of kinesin stepping by optical trapping interferometry. Nature 365:721–727. doi:10.1038/365721a0
Tees DFJ, Woodward JT, Hammer DA (2001) Reliability theory for receptor–ligand bond dissociation. J Chem Phys 114:7483–7496. doi:10.1063/1.1356030
Tolentino TP, Wu J, Zarnitsyna VI, Fang Y, Dustin ML, Zhu C (2008) Measuring diffusion and binding kinetics by contact area FRAP. Biophys J 95:920–930. doi:10.1529/biophysj.107.114447
Ushiyama S, Laue TM, Moore KL, Erickson HP, McEver RP (1993) Structural and functional-characterization of monomeric soluble P-selectin and comparison with membrane P-selectin. J Biol Chem 268:15229–15237
Veigel C, Bartoo ML, White DC, Sparrow JC, Molloy JE (1998) The stiffness of rabbit skeletal actomyosin cross-bridges determined with an optical tweezers transducer. Biophys J 75:1424–1438. doi:10.1016/S0006-3495(98)74061-5
Veigel C, Molloy JE, Schmitz S, Kendrick-Jones J (2003) Load-dependent kinetics of force production by smooth muscle myosin measured with optical tweezers. Nat Cell Biol 5:980–986. doi:10.1038/ncb1060
Williams TE, Nagarajan S, Selvaraj P, Zhu C (2001) Quantifying the impact of membrane microtopology on effective two-dimensional affinity. J Biol Chem 276:13283–13288. doi:10.1074/jbc.M010427200
Wu L, Xiao BT, Jia XL, Zhang Y, Lu SQ, Chen J et al (2007) Impact of carrier stiffness and microtopology on two-dimensional kinetics of P-selectin and P-selectin glycoprotein ligand-1 (PSGL-1) interactions. J Biol Chem 282:9846–9854. doi:10.1074/jbc.M609219200
Yago T, Leppanen A, Qiu H, Marcus WD, Nollert MU, Zhu C et al (2002) Distinct molecular and cellular contributions to stabilizing selectin-mediated rolling under flow. J Cell Biol 158:787–799. doi:10.1083/jcb.200204041
Yago T, Zarnitsyna VI, Klopocki AG, McEver RP, Zhu C (2007) Transport governs flow-enhanced cell tethering through L-selectin at threshold shear. Biophys J 92:330–342. doi:10.1529/biophysj.106.090969
Zarnitsyna VI, Huang J, Zhang F, Chien YH, Leckband D, Zhu C (2007) Memory in receptor–ligand-mediated cell adhesion. Proc Natl Acad Sci USA 104:18037–18042. doi:10.1073/pnas.0704811104
Zhang Y, Sun G, Lu S, Li N, Long M (2008) Low spring constant regulates P-selectin-PSGL-1 bond rupture. Biophys J 95:5439–5448. doi:10.1529/biophysj.108.137141
Zhu C (2000) Kinetics and mechanics of cell adhesion. J Biomech 33:23–33. doi:10.1016/S0021-9290(99)00163-3
Acknowledgments
We thank Dr. R. P. McEver for the generous gifts of sPs, S12, sLs, CA21, PLE, 1478 and PL2 proteins, Dr. Z. Z. Ye for PSGL-1 purification. This work was supported by National Natural Science Foundation of China grants 30730032 and 10332060, National Key Basic Research Foundation of China grant 2006CB910303, National High Technology Research and Development Program of China grant 2007AA02Z306, and Chinese Academy of Sciences grant 2005-1-16 (M.L.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, G., Zhang, Y., Huo, B. et al. Surface-bound selectin–ligand binding is regulated by carrier diffusion. Eur Biophys J 38, 701–711 (2009). https://doi.org/10.1007/s00249-009-0428-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-009-0428-y