Abstract
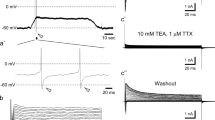
The contribution of cationic conductances in shaping the rod photovoltage was studied in light adapted cells recorded under whole-cell voltage- or current-clamp conditions. Depolarising current steps (of size comparable to the light-regulated current) produced monotonic responses when the prepulse holding potential (V h) was −40 mV (i.e. corresponding to the membrane potential in the dark). At V h = −60 mV (simulating the steady-state response to an intense background of light) current injections <35 pA (mimicking a light decrement) produced instead an initial depolarisation that declined to a plateau, and voltage transiently overshot V h at the stimulus offset. Current steps >40 pA produced a steady depolarisation to ≈−16 mV at both V h. The difference between the responses at the two V h was primarily generated by the slow delayed-rectifier-like K+ current (I Kx), which therefore strongly affects both the photoresponse rising and falling phase. The steady voltage observed at both V h in response to large current injections was instead generated by Ca-activated K+ channels (I KCa), as previously found. Both I Kx and I KCa oppose the cation influx, occurring at the light stimulus offset through the cGMP-gated channels and the voltage-activated Ca2+ channels (I Ca). This avoids that the cation influx could erratically depolarise the rod past its normal resting value, thus allowing a reliable dim stimuli detection, without slowing down the photovoltage recovery kinetics. The latter kinetics was instead accelerated by the hyperpolarisation-activated, non-selective current (I h) and I Ca. Blockade of all K+ currents with external TEA unmasked a I Ca-dependent regenerative behaviour.










Similar content being viewed by others
Abbreviations
- C m :
-
Rod membrane capacitance
- ChTx:
-
Charybdotoxin
- I Ca :
-
Voltage-activated Ca2+ current
- I cG :
-
cGMP-gated Na+ and Ca2+ current
- I ClCa :
-
Ca-activated Cl− current
- I h :
-
Hyperpolarisation-activated, non-selective cation current
- I KCa :
-
Ca-activated K+ current
- I Kx :
-
Slow delayed-rectifier-like K+ current
- OS:
-
Isolated rod outer segment
- R m :
-
Rod membrane resistance
- V h :
-
Holding potential
References
Attwell D, Wilson M (1980) Behaviour of the rod network in the tiger salamander retina mediated by membrane properties of individual rods. J Physiol 309:287–315
Bader CR, Bertrand D, Schwartz EA (1982) Voltage-activated and calcium-activated currents studied in solitary rod inner segments from the salamander retina. J Physiol 331:253–284
Barnes S (1994) After transduction: response shaping and control of transmission by ion channels of the photoreceptor inner segments. Neurosci 58:447–459. doi:10.1016/0306-4522(94)90072-8
Barnes S, Deschênes MC (1992) Contribution of Ca and Ca-activated Cl channels to regenerative depolarization and membrane bistability of cone photoreceptors. Neurophysiol 68:745–755
Barnes S, Hille B (1989) Ionic channels of the inner segment of tiger salamander cone photoreceptors. J Gen Physiol 94:719–743. doi:10.1085/jgp.94.4.719
Beech DJ, Barnes S (1989) Characterization of a voltage-gated K+ channel that accelerates the rod response to dim light. Neuron 3:573–581. doi:10.1016/0896-6273(89)90267-5
Brown DT, Flaming DG (1978) Opposing effects of calcium and barium in vertebrate rod photoreceptors. Proc Natl Acad Sci USA 75:1587–1590. doi:10.1073/pnas.75.3.1587
Cia D, Bordais A, Varela C, Forster V et al (2005) Voltage-gated channels and calcium homeostasis in mammalian rod photoreceptors. J Neurophysiol 93:1468–1475. doi:10.1152/jn.00874.2004
Corey DP, Dubinsky JM, Schwartz EA (1984) The calcium current in inner segments of rods from the salamander (Ambystoma tigrinum) retina. J Physiol 354:557–575
Fain GL, Gerschenfeld HM, Quandt FN (1980) Calcium spikes in toad rods. J Physiol 303:495–513
Frings S, Brüll N, Dzeja C et al (1998) Characterization of ether-à-go-go channels present in photoreceptors reveals similarity to I Kx, a K+ current in rod inner segments. J Gen Physiol 111:583–599. doi:10.1085/jgp.111.4.583
Gerschenfeld HM, Piccolino M, Neyton J (1980) Feed-back modulation of cone synapses by L-horizontal cells of turtle retina. J Exp Biol 89:177–192
Han Y, Jacoby RA, Wu SM (2000) Morphological and electrophysiological properties of dissociated primate retinal cells. Brain Res 875:175–186. doi:10.1016/S0006-8993(00)02614-7
Hestrin S (1987) The properties and function of inward rectification in rod photoreceptors of the tiger salamander. J Physiol 390:319–333
Hille B, Woodhull AM, Shapiro BI (1975) Negative surface charge near sodium channels of nerve: divalent ions, monovalent ions, and pH. Philos Trans R Soc Lond B Biol Sci 270:301–318. doi:10.1098/rstb.1975.0011
Hurley JB (2002) Shedding light on adaptation. J Gen Physiol 119:125–128. doi:10.1085/jgp.119.2.125
Kawai F, Horiguchi M, Suzuki H, Miyachi E (2001) Na(+) action potentials in human photoreceptors. Neuron 30:451–458. doi:10.1016/S0896-6273(01)00299-9
Maricq AV, Korenbrot JI (1988) Calcium and calcium-dependent chloride currents generate action potentials in solitary cone photoreceptors. Neuron 1:503–515. doi:10.1016/0896-6273(88)90181-X
McNaughton PA (1990) Light response of vertebrate photoreceptors. Physiol Rev 70:847–883
Moriondo A, Rispoli G (2003) A step-by-step model of phototransduction cascade shows that Ca2+ regulation of guanylate cyclase accounts only for short-term changes of photoresponse. Photochem Photobiol Sci 2:1292–1298. doi:10.1039/b303871h
Moriondo A, Pelucchi B, Rispoli G (2001) Calcium-activated potassium current clamps the dark potential of vertebrate rods. Eur J NeuroSci 14:19–26. doi:10.1046/j.0953-816x.2001.01605.x
Owen WG (1987) Ionic conductances in rod photoreceptors. Annu Rev Physiol 49:743–764. doi:10.1146/annurev.ph.49.030187.003523
Rispoli G (1998) Calcium regulation of phototransduction in vertebrate rod outer segments. J Photochem Photobiol B 44:1–20. doi:10.1016/S1011-1344(98)00083-9
Schnapf JL, Nunn BJ, Meister M et al (1990) Visual transduction in cones of the monkey Macaca fascicularis. J Physiol 427:681–713
Wollmuth LP (1994) Mechanism of Ba2+ block of M-like K channels of rod photoreceptors of tiger salamanders. J Gen Physiol 103:45–66. doi:10.1085/jgp.103.1.45
Acknowledgments
This work was supported by grants from the Ministero per l’Istruzione, l’Università e la Ricerca (MIUR), Roma.
Author information
Authors and Affiliations
Corresponding author
Additional information
Proceedings of the XIX Congress of the Italian Society of Pure and Applied Biophysics (SIBPA), Rome, September 2008.
Rights and permissions
About this article
Cite this article
Moriondo, A., Rispoli, G. The contribution of cationic conductances to the potential of rod photoreceptors. Eur Biophys J 39, 889–902 (2010). https://doi.org/10.1007/s00249-009-0419-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-009-0419-z