Abstract
This paper investigates the influence of cell density on cell membrane electropermeabilization. The experiments were performed on dense cell suspensions (up to 400 × 106 cells/ml), which represent a simple model for studying electropermeabilization of tissues. Permeabilization was assayed with a fluorescence test using Propidium iodide to obtain the mean number of permeabilized cells (i.e. fluorescence positive) and the mean fluorescence per cell (amount of loaded dye). In our study, as the cell density increased from 10 × 106 to 400 × 106 cells/ml, the fraction of permeabilized cells decreased by approximately 50%. We attributed this to the changes in the local electric field, which led to a decrease in the amplitude of the induced transmembrane voltage. To obtain the same fraction of cell permeabilization in suspensions with 10 × 106 and 400 × 106 cells/ml, the latter suspension had to be permeabilized with higher pulse amplitude, which is in qualitative agreement with numerical computations. The electroloading of the cells also decreased with cell density. The decrease was considerably larger than expected from the differences in the permeabilized cell fractions alone. The additional decrease in fluorescence was mainly due to cell swelling after permeabilization, which reduced extracellular dye availability to the permeabilized membrane and hindered the dye diffusion into the cells. We also observed that resealing of cells appeared to be slower in dense suspensions, which can be attributed to cell swelling resulting from electropermeabilization.









Similar content being viewed by others
Abbreviations
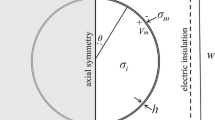
- ΔΨ :
-
induced transmembrane voltage, V
- R :
-
cell radius, m
- E :
-
applied electric field, V/m
- ϕ:
-
angle between E and the normal vector to the membrane, °
- ϕ c :
-
critical angle where permeabilization occurs, °
- E S :
-
critical amplitude of the electric field, V/m
- A perm :
-
permeabilized surface of the membrane, m2
- A tot :
-
total area of the membrane, m2
- α:
-
angle, °
- Φ :
-
flow of molecules, mol/s
- P :
-
permeability coefficient, m/s
- ΔS :
-
concentration difference of the molecule S, mol/m3
- F(t):
-
fraction of membrane defects in the permeabilized region, –
- F *(N,T):
-
fraction of membrane defects immediately after the onset of permeabilizing pulse, –
- t :
-
time, s
- N :
-
number of pulses, –
- T :
-
pulse duration, s
- k :
-
resealing rate, 1/s
- F NC :
-
fraction of permeabilized surface where cell contacts are not present, –
- c :
-
total concentration of PI after permeabilization (cells + external medium), mol/m3
- c 1 :
-
concentration of PI in external medium, mol/m3
- V :
-
total volume (cells + external medium), m3
- V 1 :
-
volume of external medium, m3
- EMEM:
-
eagle’s minimum essential medium, –
- PI:
-
propidium iodide, –
- CHO:
-
Chinese hamster ovary cells, –
References
Abidor IG, Barbul AI, Zhelev DV, Doinov P, Bandrina IN, Osipova EM, Sukharev SI (1993) Electrical properties of cell pellets and cell electrofusion in a centrifuge. Biochim Biophys Acta 1152:207–218
Abidor IG, Li LH, Hui SW (1994) Studies of cell pellets: II. Osmotic properties, electroporation, and related phenomena: membrane interactions. Biophys J 67:427–435
Barnett A, Weaver JC (1991) Electroporation: a unified, quantitative theory of reversible electrical breakdown and rupture. Bioelectrochem Bioenerg 25:163–182
Canatella PJ, Black MM, Bonnichsen DM, McKenna C, Prausnitz MR (2004) Tissue electroporation: quantification and analysis of heterogeneous transport in multicellular environments. Biophys J 86:3260–3268
Čemažar M, Grošel A, Glavač D, Kotnik V, Škobrne M, Kranjc S, Mir LM, Andre F, Opolon P, Serša G (2003) Effects of electrogenetherapy with p53wt combined with cisplatin on survival of human tumor cell lines with different p53 status. DNA Cell Biol 22:765–775
Fattori E, Cappelletti M, Zampaglione I, Mennuni C, Calvaruso F, Arcuri M, Rizzuto G, Costa P, Perretta G, Ciliberto G, La Monica N (2005) Gene electro-transfer of an improved erythropoietin plasmid in mice and non-human primates. J Gene Med 7:228–236
Golzio M, Mora MP, Raynaud C, Delteil C, Teissié J, Rols MP (1998) Control by osmotic pressure of voltage-induced permeabilization and gene transfer in mammalian cells. Biophys J 74:3015–3022
Golzio M, Teissié J, Rols MP (2002) Direct visualization at the single-cell level of electrically mediated gene delivery. Proc Natl Acad Sci 99:1292–1297
Gothelf A, Mir LM, Gehl J (2003) Electrochemotherapy: results of cancer treatment using enhanced delivery of bleomycin by electroporation. Cancer Treat Rev 29:371–387
Grosse C, Schwan HP (1992) Cellular membrane potentials induced by alternating fields. Biophys J 63:1632–1642
Kinosita K, Tsong TY (1977) Formation and resealing of pores of controlled sizes in human erythrocyte membrane. Nature 268:438–441
Kotnik T, Bobanović F, Miklavčič D (1997) Sensitivity of transmembrane voltage induced by applied electric fields—a theoretical analysis. Bioelectrochem Bioenerg 43:285–291
Li LH, Ross P, Hui SW (1999) Improving electrotransfection efficiency by post pulse centrifugation. Gene Ther 6:364–372
Mir LM, Orlowski S (1999) Mechanisms of electrochemotherapy. Adv Drug Deliver Rev 35:107–118
Neumann E, Ridder MS, Wang Y, Hofschneider PH (1982) Gene transfer into mouse lyoma cells by electroporation in high electric fields. EMBO J 1:841–845
Neumann E, Toensing K, Kakorin S, Budde P, Frey J (1998) Mechanism of electroporative dye uptake by mouse B cells. Biophys J 74:98–108
Neumann E, Kakorin S, Toensing K (1999) Fundamentals of electroporative delivery of drugs and genes. Bioelectrochem Bioenerg 48:3–16
Okino M, Mohri H (1987) Effects of high-voltage electrical impulse and an anticancer drug on in vivo growing tumors. Jpn J Cancer Res 78:1319–1321
Pauly H, Schwan HP (1959) Über die impedanz einer suspension von kugelförmigen teilchen mit einer schale. Z Naturforsch 14B:125–131
Pavlin M, Pavšelj N, Miklavčič D (2002) Dependence of induced transmembrane potential on cell density, arrangement, and cell position inside a cell system. IEEE Trans Biomed Eng 49:605–612
Pavlin M, Kandušer M, Reberšek M, Pucihar G, Hart FX, Magjarević R, Miklavčič D (2005) Effect of cell electroporation on the conductivity of a cell suspension. Biophys J 88:4378–4390
Pucihar G, Kotnik T, Valič B, Miklavčič D (2006) Numerical determination of transmembrane voltage induced on irregularly shaped cells. Annals Biomed Eng 34:642–652
Rols MP, Teissié J (1990) Electropermeabilization of mammalian cells: quantitative analysis of the phenomenon. Biophys J 58:1089–1098
Rols MP, Teissié J (1998) Electropermeabilization of mammalian cells to macromolecules: control by pulse duration. Biophys J 75:1415–1423
Rols MP, Dahhou F, Mishra KP, Teissie J (1990) Control of electric field induced cell membrane permeabilization by membrane order. Biochemistry 29:2960–2966
Rols MP, Delteil C, Golzio M, Teissié J (1998) Control by ATP and ADP of voltage-induced mammalian-cell-membrane permeabilization, gene transfer and resulting expression. Eur J Biochem 254:382–388
Šatkauskas S, Bureau MF, Puc M, Mahfoudi A, Scherman D, Miklavčič D, Mir LM (2002) Mechanisms of in vivo DNA electrotransfer: respective contributions of cell electropermeabilization and DNA electrophoresis. Mol Ther 5:133–140
Šatkauskas S, Batiuškaite D, Šalomskaite-Davalgiene S, Venslauskas MS (2005) Effectiveness of tumor electrochemotherapy as a function of electric pulse strength and duration. Bioelectrochemistry 65:105–111
Schmeer M, Seipp T, Pliquett U, Kakorin S, Neumann E (2004) Mechanism for the conductivity changes caused by membrane electroporation of CHO cell-pellets. PCCP 6:5564–5574
Schwan HP (1957) Electrical properties of tissue and cell suspensions. Adv Biol Med Phys 5:147–209
Serša G, Čemažar M, Miklavčič D (1995) Antitumor effectiveness of electrochemotherapy with cis-diammindichloroplatinum(II) in mice. Cancer Res 55:3450–3455
Sixou S, Teissié J (1993) Exogeneous uptake and release of molecules by electroloaded cells: a digitized videomicroscopy study. Bioelectrochem Bioenerg 31:237–257
Susil R, Šemrov D, Miklavčič D (1998). Electric field induced transmembrane potential depends on cell density and organization. Electro Magnetobiol 17:391–399
Teissié J, Eynard N, Gabriel B, Rols MP (1999) Electropermeabilization of cell membranes. Adv Drug Deliver Rev 35:3–19
Tsong TY (1991) Electroporation of cell membranes. Biophys J 60:297–306
Weaver JC, Chizmadzhev YA (1996) Theory of electroporation: a review. Bioelectrochem Bioenerg 41:135–160
Acknowledgments
The author (G. P.) would like to thank Dr. M. Golzio, Dr. M. P. Rols and Dr. B. Gabriel for their valuable discussions during the experiments, and Ms. C. Millot for her help with cell cultures. This work was supported by the Ministry of the Higher Education and Science of the Republic of Slovenia. G. P. was also a recipient of a scholarship from the French government. The two institutes are partners in a Slovenian-French CNRS PICS program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pucihar, G., Kotnik, T., Teissié, J. et al. Electropermeabilization of dense cell suspensions. Eur Biophys J 36, 173–185 (2007). https://doi.org/10.1007/s00249-006-0115-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-006-0115-1