Abstract
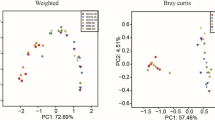
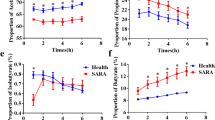
The effects of a grain-based subacute ruminal acidosis (SARA) challenge on bacteria in the rumen and feces of lactating dairy cows were determined. Six lactating, rumen-cannulated Danish Holstein cows were used in a cross-over study with two periods. Periods included two cows on a control diet and two cows on a SARA challenge. The control diet was a total mixed ration containing 45.5% dry matter (DM), 43.8% DM neutral detergent fiber, and 19.6% DM starch. The SARA challenge was conducted by gradually substituting the control diet with pellets containing 50% wheat and 50% barley over 3 days to reach a diet containing 55.6% DM, 31.3% DM neutral detergent fiber, and 31.8% DM starch, which was fed for four more days. Rumen fluid samples were collected at day 7 and 10 of experimental periods. Feces samples were collected on days 8 and 10 of these periods. Extracted DNA from the rumen and feces samples was analyzed to assess their bacterial communities using MiSeq Illumina sequencing of the V4 region of the 16S rRNA gene. The induction of SARA reduced the richness, diversity, and stability of bacterial communities and resulted in distinctly different microbiota in the rumen and feces. Bacteroidetes and Firmicutes were the most abundant phyla and, combined, they represented 76.9 and 94.4% of the bacterial community in the rumen fluid and the feces, respectively. Only the relative abundance of Firmicutes in the rumen was increased by the SARA challenge. In rumen fluid and feces, the abundances of nine out of the 90 and 25 out of the 89 taxa, respectively, were affected by the challenge. Hence, SARA challenge altered the composition of the bacterial community at the lower taxonomical level in the feces and therefore also likely in the hindgut, as well as in the rumen. However, only reductions in the bacterial richness and diversity in the rumen fluid and feces were in agreement with those of other studies and had a biological basis. Although the composition of the bacterial community of the feces was affected by the SARA challenge, bacterial taxa in the feces that can be used for accurate and non-invasive diagnosis of SARA could not be identified.





Similar content being viewed by others
References
Plaizier JC, Khafipour E, Li S, Gozho GN, Krause DO (2012) Subacute ruminal acidosis (SARA), endotoxins and health consequences. Anim. Feed Sci. Technol. 172:9–21
Kleen JL, Cannizzo C (2012) Incidence, prevalence and impact of SARA in dairy herds. Anim. Feed Sci. Technol. 172:4–8
Plaizier JC, Krause DO, Gozho GN, McBride BW (2008) Subacute ruminal acidosis in dairy cows: the physiological causes, incidence and consequences. Vet J 176:21–31
Gozho GN, Plaizier JC, Krause DO, Kennedy AD, Wittenberg KM (2005) Subacute ruminal acidosis induces ruminal lipopolysaccharide endotoxin release and triggers an inflammatory response. J. Dairy Sci. 88:1399–1403
Enemark J (2008) The monitoring, prevention and treatment of sub-acute ruminal acidosis (SARA): a review. Vet J 176:32–43
Khafipour E, Krause DO, Plaizier JC (2009a) Alfalfa pellet-induced subacute ruminal acidosis in dairy cows increases bacterial endotoxin in the rumen without causing inflammation. J. Dairy Sci. 92:1712–1724
Khafipour E, Krause DO, Plaizier JC (2009b) A grain-based subacute ruminal acidosis challenge causes translocation of lipopolysaccharide and triggers inflammation. J. Dairy Sci. 92:1060–1070
Calsamiglia S, Blanch M, Ferret A, Moya D (2012) Is subacute ruminal acidosis a pH related problem? Causes and tools for its control. Anim. Feed Sci. Technol. 172:42–50
Petri RM, Schwaiger T, Penner GB, Beauchemin KA, Forster RJ, McKinnon JJ, McAllister TA (2013) Characterization of the core rumen microbiome in cattle during transition from forage to concentrate as well as during and after an acidotic challenge. PLoS One 8:e83424
Mao SY, Zhang RY, Wang DS, Zhu WY (2013) Impact of subacute ruminal acidosis (SARA) adaptation on rumen microbiota in dairy cattle using pyrosequencing. Anaerobe 24:12–19
Khafipour E, Li S, Tun H, Derakhshani H, Moossavi S, Plaizier JC (2016) Effects of grain feeding on microbiota in the digestive tract of cattle. Anim Front 6:13–19
Russell JB, Rychlik JL (2001) Factors that alter rumen microbial ecology. Science 292:1119–1122
Krause DO, Nagaraja TG, Wright AD, Callaway TR (2013) Board-invited review: rumen microbiology: leading the way in microbial ecology. J. Anim. Sci. 91:331–341
Weimer PJ (2015) Redundancy, resiliance, and host specificity of the rumen microbiota:implications for engineering improved ruminal fermentation. Front. Microbiol. 6:296. doi:10.3389/fmicb.2015.00296
Fernando SC, Purvis HT, Najar FZ, Sukharnikov LO, Krehbiel CR, Nagaraja TG (2010) Rumen microbial population dynamics during adaptation to a high-grain diet. Appl. Environ. Microbiol. 76:7482–7490
Petri R, Forster R, Yang W, McKinnon J, McAllister T (2012) Characterization of rumen bacterial diversity and fermentation parameters in concentrate fed cattle with and without forage. J. Appl. Microbiol. 112:1152–1162
Danscher AM, Li S, Andersen PH, Khafipour E, Kristensen NB, Plaizier JC (2015) Indicators of induced subacute ruminal acidosis (SARA) in Danish Holstein cows. Acta Vet. Scand. 57:39
Derakhshani H, Tun HM, Khafipour E (2016) An extended single-index multiplexed 16S rRNA sequencing for microbial community analysis on MiSeq Illumina platforms. J Basic Microbiol 56:321–326
Magoč T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformat 27:2957–2963
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nature Meth 10:996–998
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformat 27:2194–2200
Caporaso JG, Bittinger K, Bushman FD, DeSantis TZ, Andersen GL, Knight R (2010) PyNAST: a flexible tool for aligning sequences to a template alignment. Bioinformatic 26:266–267
Price MN, Dehal PS, Arkin AP (2009) FastTree: computing large minimum evolution trees with profiles instead of a distance matrix. Molec Biol Evol 26:1641–1650
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Meth 7:335–336
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73:5261–5267
McMurdie PJ, Holmes S (2013) Phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS One 8:e61217
de Cárcer DA, Denman SE, McSweeney C, Morrison M (2011) Evaluation of subsampling-based normalization strategies for tagged high-throughput sequencing data sets from gut microbiomes. Appl. Environ. Microbiol. 77:8795–8798
Anderson MJ, Crist TO, Chase JM, Vellend M, Inouye BD, Freestone AL, Sanders NJ, Cornell HV, Comita LS, Davies KF (2011) Navigating the multiple meanings of β diversity: a roadmap for the practicing ecologist. Ecol. Lett. 14:19–28
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15:550
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Royal Stat Soc Series B (Methodol) 57:289–300
Fisher RA, Corbet AS, Williams CB (1943) The relation between the number of species and the number of individuals in a random sample of an animal population. J. Anim. Ecol. 12:42–58
Li S, Gozho GN, Gakhar N, Khafipour E, Krause DO, Plaizier JC (2012a) Evaluation of diagnostic measures for subacute ruminal acidosis in dairy cows. Can J Ani Sci 92:353–364
Li S, Khafipour E, Krause DO, Kroeker A, Rodriguez-Lecompte JC, Gozho GN, Plaizier JC (2012b) Effects of subacute ruminal acidosis challenges on fermentation and endotoxins in the rumen and hindgut of dairy cows. J. Dairy Sci. 95:294–303
Saleem F, Ametaj BN, Bouatra S, Mandal R, Zebeli Q, Dunn SM, Wishart DS (2012) A metabolomics approach to uncover the effects of grain diets on rumen health in dairy cows. J. Dairy Sci. 95:6606–6623
Levine JM, D'Antonio CM (1999) Elton revisited: a review of evidence linking diversity and invasibility. Oikos 87:15–26
Khafipour E, Li S, Plaizier JC, Krause DO (2009) Rumen microbiome composition determined using two nutritional models of subacute ruminal acidosis. Appl. Environ. Microbiol. 75:7115–7124
Hook SE, Steele MA, Northwood KS, Dijkstra J, France J, Wright AD, McBride BW (2011) Impact of subacute ruminal acidosis (SARA) adaptation and recovery on the density and diversity of bacteria in the rumen of dairy cows. FEMS Microbiol. Ecol. 78:275–284
El Kaoutari A, Armougom F, Gordon JI, Raoult D, Henrissat B (2013) The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nature Rev Microbiol 11:497–504
Henderson G, Cox F, Ganesh S, Jonker A, Young W (2015) Rumen microbial community composition varies with diet and host, but a core microbiome is found across a wide geographical range. Sci Rep 5:14567
Plaizier JC, Li S, Tun HM, Krause DO, Khafipour E (2016) Effects of experimentally induced subacute ruminal acidosis (SARA) on the rumen and hindgut microbiome in dairy cows. Front Microbiol In press
Russell JB (2002) Rumen microbiology and its role in ruminant nutrition: Cornell University
Gressley TF, Hall MB, Armentano LE (2011) Ruminant nutrition symposium: productivity, digestion, and health responses to hindgut acidosis in ruminants. J. Anim. Sci. 89:1120–1130
Callaway TR, Dowd SE, Edrington TS, Anderson RC, Krueger N, Bauer N, Kononoff PJ, Nisbet DJ (2010) Evaluation of bacterial diversity in the rumen and feces of cattle fed different levels of dried distillers grains plus solubles using bacterial tag-encoded FLX amplicon pyrosequencing. J. Anim. Sci. 88:3977–3983
Acknowledgements
This study was funded by the Danish Research Council for Technology and Production and the Manitoba Agricultural Development and Research Council. We would like to thank the students and staff at the research station Rørrendegård for practical assistance.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Approval for the project was obtained from the Danish Animal Experiments Inspectorate (file no. 2012-15-2934-00052).
Rights and permissions
About this article
Cite this article
Plaizier, J.C., Li, S., Danscher, A.M. et al. Changes in Microbiota in Rumen Digesta and Feces Due to a Grain-Based Subacute Ruminal Acidosis (SARA) Challenge. Microb Ecol 74, 485–495 (2017). https://doi.org/10.1007/s00248-017-0940-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-017-0940-z