Abstract
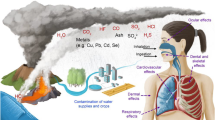
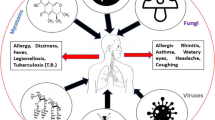
Greater Mexico City is one of the largest urban centers in the world, with an estimated population by 2010 of more than 20 million inhabitants. In urban areas like this, biological material is present at all atmospheric levels including live bacteria. We sampled the low atmosphere in several surveys at different points by the gravity method on LB and blood agar media during winter, spring, summer, and autumn seasons in the years 2008, 2010, 2011, and 2012. The colonial phenotype on blood agar showed α, β, and γ hemolytic activities among the live collected bacteria. Genomic DNA was extracted and convenient V3 hypervariable region libraries of 16S rDNA gene were high-throughput sequenced. From the data analysis, Firmicutes, Proteobacteria, and Actinobacteria were the more abundant phyla in all surveys, while the genera from the family Enterobacteriaceae, in addition to Bacillus spp., Pseudomonas spp., Acinetobacter spp., Erwinia spp., Gluconacetobacter spp., Proteus spp., Exiguobacterium spp., and Staphylococcus spp. were also abundant. From this study, we conclude that it is possible to detect live airborne nonspore-forming bacteria in the low atmosphere of GMC, associated to the microbial cloud of its inhabitants.





Similar content being viewed by others
Change history
04 August 2017
An erratum to this article has been published.
References
Konstantinidis KT (2014) Do airborne microbes matter for atmospheric chemistry and cloud formation? Environ Microbiol 16:1482–1484
Cho I, Blaser MJ (2012) The human microbiome: at the interface of health and disease. Nat Rev Genet 13:260–270
Meadow JF, Altrichter AE, Bateman AC, Stenson J, Brown GZ, Green JL, Bohannan BJ (2015) Humans differ in their personal microbial cloud. Peer J 3:e1258
Demographia World Urban Areas: 11th annual edition: 2015.01. http://www.demographia.com/
Rosas I, McCartney HA, Payne RW, Calderon C, Lacey J, Chapela R et al (1998) Analysis of the relationship between environmental factors (aeroallergens, air polution, and weather) and asthma emergency admissions to a hospital in Mexico City. Allergy 53:394–401
Valverde M, Lopez M, Lopez I, Sanchez I, Fortoul T, Ostroky-Wegman P et al (1997) DNA damage in leukocytes and buccal nasal epithelial cells of individuals exposed to air pollution in Mexico City. Environ Mol Mutagen 30:147–152
Borja-Aburto VH, Loomis DP, Bangdiwala SI, Shy CM, Rascon-Pacheco RA (1997) Ozone, suspended particulates, and daily mortality in Mexico City. Am J Epidemiol 145:258–268
Hernández-Garduño E, Pérez-Neira J, Paccagnella A, Piña-García M, Munguía-Castro M, Catalan-Vazquez M et al (1997) Air pollution and respiratory health in Mexico City. J Occup Environ Med 39:299–307
SUIVE/DGE/SALUD/Información Epidemiológica de Morbilidad, Anuario 2011. Versión Ejecutiva
Eduard W, Heederik D, Duchaine C, Green BJ (2012) Bioaerosol exposure assessment in the workplace: the past, present and recent advances. J Environ Monit 14:334–339
Stetzenbach DL, Buttner PM, Cruz P (2004) Detection and enumeration of airborne biocontaminants. Curr Opin Biotechnol 15:170–174
Rivera-González LO, Zhang Z, Sánchez BN, Zhang K, Brown DG, Rojas-Bracho L et al (2015) An assessment of air pollutant exposure methods in Mexico City, Mexico. J Air Waste Manag Assoc 65:581–591
Sánchez-Monedero AM, Roig A, Cayuela LM, Stentiford IE (2006) Emisión de Bioaerosoles Asociada a la Gestión de Residuos Orgánicos. Ingeniería 10:39–47
MacNeil L, Kauri T, Robertson W (1995) Molecular techniques and their potential application in monitoring the microbiological quality of indoor air. Can J Microbiol 41:657–665
Walter MV, Marthi B, Fieland VP, Ganio LM (1990) Effect of aerosolization on subsequent bacterial survival. Appl Environ Microbiol 56:3468–3472
Marthi B, Fieland VP, Walter M, Seidler RJ (1990) Survival of bacteria during aerosolization. Appl Environ Microbiol 56:3463–3467
Domínguez-Malfavón L, Cervantes-González E, García-Romero JR, Torres-Mota AM, Rojas-Avelizapa NG, García-Mena J (2007) Study of the microbial diversity of outdoor bioaerosols of Mexico City. In: Wang Y, Li S, Huang P, Yang Y, An Y, Sun X (eds) Progress in environmental science and technology (vol. I). Proceedings of the 2007 International Symposium on Environmental Science and Technology. Beijing, China, November 13–16, 2007. ISBN 978-7-03-020403-5. Published by Science Press 16 Donghuangchenggen North Street, Beijing, 100717, P. R. China pp 502–509.
Kawasaki T, Kyotani T, Ushiogi T, Lee H (2013) Distribution of airborne bacteria in railway stations in Tokyo, Japan. J Occup Health 55:495–502
Gangamma S (2014) Characteristics of airborne bacteria in Mumbai urban environment. Sci Total Environ 488–489:70–74
Brooks PJ, Gerba PC, Pepper LI (2004) Aerosol emission, fate, and transport from municipal and animal wastes. J Residuals Sci Technol 1:13–25
Tanner BD, Brooks JP, Haas CN, Gerba CP, Pepper IL (2005) Bioaerosol emission rate and plume characteristics during land application of liquid class B biosolids. Environ Sci Technol 39:1584–1590
Paez-Rubio T, Viau E, Romero-Hernández S, Peccia J (2005) Source bioaerosol concentration and rRNA gene-based identification of microorganisms aerosolized at a flood irrigation wastewater reuse site. Appl Environ Microbiol 71:804–810
Thorne PS, Kiekhaefer MS, Whitten P, Donham KJ (1992) Comparison of bioaerosol sampling methods in barns housing swine. Appl Environ Microbiol 58:2543–2551
Dybwad M, Granum PE, Bruheim P, Blatny JM (2012) Characterization of airborne bacteria at an underground subway station. Appl Environ Microbiol 78:1917–1929
Okten S, Asan A (2012) Airborne fungi and bacteria in indoor and outdoor environment of the Pediatric Unit of Edirne Government Hospital. Environ Monit Assess 184:1739–1751
Fang Z, Ouyang Z, Zheng H, Wang X, Hu L (2007) Culturable airborne bacteria in outdoor environments in Beijing, China. Microb Ecol 54:487–496
Rodríguez-Gómez S, Sauri MR, Peniche I, Pacheco J, Ramírez JM (2005) Aerotransportables viables en el área de tratamiento y disposición final de residuos sólidos municipales de Mérida, Yucatán. Ingeniería 9:19–29
CONAPO (2012) Delimitación de las zonas metropolitanas de México 2010. http://www.conapo.gob.mx/es/CONAPO/Zonas_metropolitanas_2010
Buttner MP, Willeke K, Grinshpun SA (1997) Sampling and analysis of airborne microorganisms. In Section VII, Aerobiology. Stetzenbach, LD (vol. ed.). Manual of environmental microbiology. Hurst CJ (ed. in chief). ASM Press, Washington. pp 629–640.
Lohmann C, Sabou M, Moussaoui W, Prévost G, Delarbre JM, Candolfi E et al (2013) Comparison between the Biflex III-Biotyper and the Axima-SARAMIS systems for yeast identification by matrix-assisted laser desorption ionization-time of flight mass spectrometry. J Clin Microbiol 51:1231–1236
Brosius J, Palmer ML, Poindexter JK, Noller HF (1978) Complete nucleotide sequence of a 16S Ribosomal RNA gene from Escherichia coli. Proc Natl Acad Sci U S A 75:4801–4805
Ehresmann C, Stiegler P, Fellner P, Ebel JP (1972) The determination of the primary structure of the 16S ribosomal RNA of Escherichia coli. 2. Nucleotide sequences of products from partial enzymatic hydrolysis. Biochimie 54:901–967
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acids Symp Ser 41:95–98
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Johnson M, Zaretskaya I, Raytselis Y, Merezhuk Y, McGinnis S, Madden TL (2008) NCBI BLAST: a better web interface. Nucleic Acids Res 36:W5–W9
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Fierer N, Hamady M, Lauber CL, Knight R (2008) The influence of sex, handedness, and washing on the diversity of hand surface bacteria. Proc Natl Acad Sci U S A 105:17994–17999
Whiteley AS, Jenkins S, Waite I, Kresoje N, Payne H, Mullan B et al (2012) Microbial 16S rRNA Ion Tag and community metagenome sequencing using the Ion Torrent (PGM) Platform. J Microbiol Meth 91:80–88
Murugesan S, Ulloa-Martínez M, Martínez-Rojano H, Galván-Rodríguez FM, Miranda-Brito C, Romano MC et al (2015) Study of the diversity and short-chain fatty acids production by the bacterial community in overweight and obese Mexican children. Eur J Clin Microbiol Infect Dis 34:1337–1346
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Haas BJ, Gevers D, Earl AM, Feldgarden M, Ward DV, Giannoukos G et al (2011) Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Res 21:494–504
DeSantis TZ, Hugenholtz P, Larsen N, Rojas M, Brodie EL, Keller K et al (2006) Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol 72:5069–5072
Vázquez-Baeza Y, Pirrung M, Gonzalez A, Knight R (2013) EMPeror: a tool for visualizing high-throughput microbial community data. Gigascience 2:16
Bartram AK, Lynch MD, Stearns JC, Moreno-Hagelsieb G, Neufeld JD (2011) Generation of multimillion-sequence 16S rRNA gene libraries from complex microbial communities by assembling paired-end Illumina reads. Appl Environ Microbiol 77:3846–3852
Huse SM, Dethlefsen L, Huber JA, Welch DM, Relman DA, Sogin ML (2008) Exploring microbial diversity and taxonomy using SSU rRNA hypervariable tag sequencing. PLoS Genet 4(11), e1000255
Yamaguchi N, Park J, Kodama M, Ichijo T, Baba T, Nasu M (2014) Changes in the airborne bacterial community in outdoor environments following Asian dust events. Microbes Environ 29:82–88
Bowers RM, McCubbin MB, Hallar AG, Fierer N (2012) Seasonal variability in airborne bacterial communities at a high-elevation site. Atmos Environ 50:41–49
Tanaka D, Terada Y, Nakashima T, Sakatoku A, Nakamura S (2015) Seasonal variations in airborne bacterial community structures at a suburban site of central Japan over a 1-year time period using PCR-DGGE method. Aerobiologia 31:143–157
McDonald LC, Banerjee SN, Jarvis WR (1999) Seasonal variation of Acinetobacter infections: 1987–1996. Nosocomial Infections Surveillance System. Clin Infect Dis 29:1133–1137
Eber MR, Shardell M, Schweizer ML, Laxminarayan R, Perencevich EN (2011) Seasonal and temperature-associated increases in gram-negative bacterial bloodstream infections among hospitalized patients. PLoS One 6, e25298
Rule AM, Schwab KJ, Kesavan J, Buckley TJ (2009) Assessment of bioaerosol generation and sampling efficiency based on Pantoea agglomerans. Aerosol Sci Technol 43:620–628
Środowiskai O, Naturalnych Z (2014) Bioaerosol concentration in the air surrounding municipal solid waste landfill. Environ Protect Nat Res 25:17–25
Eskin N, Vessey K, Tian L (2014) Research progress and perspectives of nitrogen fixing bacterium, Gluconacetobacter diazotrophicus, in monocot plants. Int J Agron 2014:208383
Bahashwan SA, El Shafey HM (2013) Antimicrobial resistance patterns of Proteus isolates from clinical specimens. Eur Sci J 9:188–202
Rodrigues DF, Tiedje JM (2007) Multi-locus real-time PCR for quantitation of bacteria in the environment reveals Exiguobacterium to be prevalent in permafrost. FEMS Microbiol Ecol 59:489–499
Tena D, Martínez NM, Casanova J, García JL, Román E, Medina MJ et al (2014) Possible Exiguobacterium sibiricum skin infection in human [letter]. Emerg Infect Dis. doi:10.3201/eid2012.140493 [Internet]
Klein EY, Sun L, Smith DL, Laxminarayan R (2013) The changing epidemiology of methicillin-resistant Staphylococcus aureus in the United States: a national observational study. Am J Epidemiol 7:666–674
Acknowledgments
This work was financed by Cinvestav, REMAS-CONACyT 0123119, CONACyT AP LIC 104881, ICYTDF/324/2009 FOLIO: 0649, CONACyT 163235 INFR-2011-01, and FONSEC SS/IMSS/ISSSTE-CONACYT-233361 granted to JGM. We thank a Postdoctoral Fellowship from FONSEC SS/IMSS/ISSSTE-CONACYT-233361 granted to SM. We also thank Mrs. Antonia López Salazar for clerical assistance and Mr. José Rodrigo García Gutiérrez for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest. The authors alone are responsible for the content and writing of the paper. The authors declared that they have no financial and personal relationships with other people or organizations that can inappropriately influence their work. There is no professional or other personal interest of any nature or kind in any product, service, and/or company.
Additional information
An erratum to this article is available at https://doi.org/10.1007/s00248-017-1046-3.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 14 kb)
Fig. S1
LB and Blood Agar plates from gravity sampling. The figure shows digital images of representative sampling plates of the surveys. a) Typical bacterial colony growth on Luria-Bertani (LB) and b) Typical bacterial colony growth on Blood-Agar (BA); hemolysis halos can be observed around some colonies (GIF 158 kb)
Fig. S2
Escherichia coli 16S rDNA gene and primers. Figure shows a representation of the Brosius 1,541 bp rrnB 16S ribosomal gene of E. coli. Primers used for PCR are shown: First Set, forward FBac 5′-ATC ATG GCT CAG ATT GAA CGC-3′ (complementary positions 16-36), reverse RBAc 5′-ACT CCT ACG GGA GGC AGC AG-3′ (position 337-356); Second Set, CGO Forward 465 5′-CTC CTA CGG GAG GCA GCA G-3′ (positions 338-356), CGO Reverse 465 5′-CAG GAT TAG ATA CCC TGG TAG-3′, (positions 782-802); and Third Set, CGO Forward 605 5′-CAG GAT TAG ATA CCC TGG TAG-3′ (positions 782-802), CGO Reverse 605 5′-CGG TGA ATA CGT TCC CGG G-3′ (1368-1386). The primer V3-341F which indicates the complementary sequence for the seven forward primers used for 16S rDNA based high-throughput sequencing (V3-341F2, V3-341F4, V3-341F7, V3-341F8, V3-341F9, V3-341F18, V3-341F49) (position 334-354), and the reverse V3-518R primer (position 506-529) have described elsewhere [42]. Solid arrows indicate complementary regions for primers; larger gray color filled arrows from V1 to V9, indicate the polymorphic variable sequences in the molecule: GenBank: J01859.1 [31, 32] (GIF 15 kb)
Fig. S3
Mass spectroscopy identification of selected bacteria. The figure shows the spectra of selected isolated bacteria in the 2008 survey in GMC, generated using the Matrix Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry, technology AXIMA@SARAMIS from Anagnostec as described in Material and methods [30]. Identified bacteria using this method are: a) Bacillus subtilis; b) Staphylococcus epidermidis; c) Bacillus subtilis; d) Bacillus pumilus; e) Bacillus subtilis; f) Staphylococcus xylosus; g) Bacillus cereus; h) Not analyzed by Anagnostec; i) Bacillus subtilis; j) Staphylococcus epidermidis; k) Bacillus pumilus; l) Bacillus pumilus; m) Bacillus pumilus; n) Staphylococcus saprophyticus; o) Bacillus pumilus; p) Psychrobacter phenylpyruvicus; q) Not identified by the AXIMA@SARAMIS database; r) Psychrobacter phenylpyruvicus; s) Bacillus megaterium; t) Bacillus sp.; u) Bacillus subtilis; v) Bacillus coagulans; w) Psychrobacter phenylpyruvicus; x) Bacillus sp. See bacteria in Table 3 (GIF 224 kb)
(GIF 262 kb)
Fig. S4
Beta diversity. Unweighted UniFrac analyses were used to calculate distances between samples obtained from the seven surveys, determined by massive sequencing of V3-16S rDNA libraries prepared from DNA from bacterial growth on LB plates as described in Materials and methods. Three-dimensional scatter plots were generated by principal coordinates analysis (PCoA). (GIF 33 kb)
Fig. S5
Alpha diversity Rarefaction curves. Rarefaction curves were generated using massive sequencing data of V3-16S rDNA libraries from the seven surveys, as described in Materials and methods. The vertical axes display the diversity of the community, while the horizontal axes display the number of sequences considered in the diversity calculation. Each color indicates diversity of community for each survey. Panel A, displays rarefaction curves of the number of observed species was calculated at a similarity threshold of 97 %; Panel B, represents the rarefaction curves using the Shannon index; Panel C, shows rarefaction curves using the Chao1. (GIF 61 kb)
Fig. S6
Jacknifed beta diversity. Unweighted UniFrac analyses were used to calculate distances among sequencing data of the seven surveys, determined by massive sequencing of V3-16S rDNA libraries prepared as described in Materials and methods; tree chart was generated by using Jacknifing method. (GIF 16 kb)
Rights and permissions
About this article
Cite this article
García-Mena, J., Murugesan, S., Pérez-Muñoz, A.A. et al. Airborne Bacterial Diversity from the Low Atmosphere of Greater Mexico City. Microb Ecol 72, 70–84 (2016). https://doi.org/10.1007/s00248-016-0747-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-016-0747-3