Abstract
Background
Ferumoxytol is an ultrasmall superparamagnetic iron oxide (USPIO) nanoparticle agent used to treat iron deficiency anemia in adults with chronic kidney disease.
Objective
We aim to determine the feasibility of using ferumoxytol for clinical pediatric cardiac and vascular imaging.
Material and methods
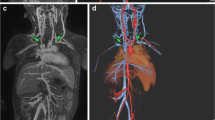
We retrospectively identified 23 consecutive children who underwent MRI with ferumoxytol (11 males; mean age: 7.4 years, range: 3 days–18 years), yielding 12 abdominal MR angiography and 15 cardiac MRI studies. Medical records were reviewed for the clinical indication, ferumoxytol dose, injection rate, sedation and any complication. A two-reader consensus scored the images on a five-point scale for overall image quality and delineation of various anatomical structures. Signal-to-background ratios for abdominal aorta and inferior vena cava for abdominal cases and blood pool-myocardium contrast ratios for cardiac cases were calculated. The confidence intervals for obtaining a score of three or above for each image parameter were calculated by using adjusted Wald method.
Results
For abdominal MR angiography, average scores for overall image quality, as well as delineation of the hepatic artery, superior mesenteric artery, renal artery and veins were 4.5, 4.3, 4.3, 3.7 and 4.7, respectively. For cardiac exams, the average scores for overall image quality, systemic arteries, pulmonary arteries, pulmonary veins, valves and ventricles were 4.4, 4.6, 4.1, 4.8, 4.1 and 4.7, respectively. For all parameters, the lower bound for the proportion of cases to have a score of 3 or above was 65%. Signal-to-background ratios for aorta and abdominal veins averaged 86 +/− 74 and 86 +/− 77 for full-dose images, and 23 and 18 for half-dose images, respectively. Mean blood pool to myocardium contrast ratio was 3:3.
Conclusion
Ferumoxytol can provide excellent image quality for pediatric body MR angiography/MR venography at a dose of 1.5 or 3 mg Fe/kg. Further investigation should be directed toward understanding the lowest dose that can be administered.









Similar content being viewed by others
References
Prince MR, Zhang HL, Chabra SG et al (2003) A pilot investigation of new superparamagnetic iron oxide (ferumoxytol) as a contrast agent for cardiovascular MRI. J Xray Sci Technol 11:231–240
Li W, Tutton S, Vu AT et al (2005) First-pass contrast-enhanced magnetic resonance angiography in humans using ferumoxytol, a novel ultrasmall superparamagnetic iron oxide (USPIO)-based blood pool agent. J Magn Reson Imaging 21:46–52
Bremerich J, Bilecen D, Reimer P (2007) MR angiography with blood pool contrast agents. Eur Radiol 17:3017–3024
Bashir MR, Mody R, Neville A et al (2014) Retrospective assessment of the utility of an iron-based agent for contrast-enhanced magnetic resonance venography in patients with endstage renal diseases. J Magn Reson Imaging 40:113–118
Sigovan M, Gasper W, Alley HF et al (2012) USPIO-enhanced MR angiography of arteriovenous fistulas in patients with renal failure. Radiology 265:584–590
Ersoy H, Jacobs P, Kent CK et al (2004) Blood pool MR angiography of aortic stent-graft endoleak. AJR Am J Roentgenol 182:1181–1186
Gahramanov S, Muldoon LL, Varallyay CG et al (2013) Pseudoprogression of glioblastoma after chemo- and radiation therapy: diagnosis by using dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging with ferumoxytol versus gadoteridol and correlation with survival. Radiology 266:842–852
Hamilton BE, Nesbit GM, Dosa E et al (2011) Comparative analysis of ferumoxytol and gadoteridol enhancement using T1- and T2-weighted MRI in neuroimaging. AJR Am J Roentgenol 197:981–988
Li W, Salanitri J, Tutton S et al (2007) Lower extremity deep venous thrombosis: evaluation with ferumoxytol-enhanced MR imaging and dual-contrast mechanism–preliminary experience. Radiology 242:873–881
McCullough BJ, Kolokythas O, Maki JH et al (2013) Ferumoxytol in clinical practice: implications for MRI. J Magn Reson Imaging 37:1476–1479
Stabi KL, Bendz LM (2011) Ferumoxytol use as an intravenous contrast agent for magnetic resonance angiography. Ann Pharmacother 45:1571–1575
Thompson EM, Guillaume DJ, Dosa E et al (2012) Dual contrast perfusion MRI in a single imaging session for assessment of pediatric brain tumors. J Neurol 109:105–114
Schiller B, Bhat P, Sharma A (2013) Safety and effectiveness of ferumoxytol in hemodialysis patients at 3 dialysis chains in the United States over a 12-month period. Clin Ther 36:70–83
Schieda N (2013) Parenteral ferumoxtol interaction with magnetic resonance imaging: a case report, review of the literature and advisory warning. Insights Imaging 4:509–512
Institute of Medicine, Food and Nutrition Board (2011) Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc : a report of the panel on micronutrients. National Academy Press, Washington, DC
Acknowledgment
This work is supported by a grant from the National Institutes of Health (R01 EB009690 and P41 EB015891) and the Tashia and John Morgridge Faculty Scholar Fund.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ruangwattanapaisarn, N., Hsiao, A. & Vasanawala, S.S. Ferumoxytol as an off-label contrast agent in body 3T MR angiography: a pilot study in children. Pediatr Radiol 45, 831–839 (2015). https://doi.org/10.1007/s00247-014-3226-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-014-3226-3