Abstract
Background
Widespread white matter (WM) pathology in preterm children has been proposed.
Objective
The purpose of this study was to investigate maturational differences of WM between preterm infants with thinning of the corpus callosum and full-term infants.
Materials and methods
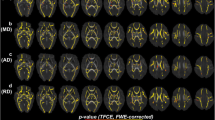
A total of 18 preterm children and 18 full-term children were divided into three subgroups according to the corrected age at the time of diffusion tensor imaging scanning. Tract-based spatial statistics was used for assessing differences in fractional anisotropy (FA) between preterm and full-term children, and between each age-related subgroup in preterm and in full-term children.
Results
In the preterm group, FA values of overall WM showed an increase with age. This trend indicates that WM maturation is a gradual occurrence during a child’s first 2 years. In the full-term group, most WM structures had reached maturation at around 1 year of age; however, centrum semiovale level showed sustained maturation during the first 2 years.
Conclusion
Results of our study demonstrate radiologic maturational differences of WM and provide evidence of the need for therapeutic intervention within 2 years of birth to prevent specific functional impairment and to improve clinical outcome in preterm children.


Similar content being viewed by others
References
Langhoff-Roos J, Kesmodel U, Jacobsson B et al (2006) Spontaneous preterm delivery in primiparous women at low risk in Denmark: population based study. BMJ 332:937–939
Latal B (2009) Prediction of neurodevelopmental outcome after preterm birth. Pediatr Neurol 40:413–419
Bhutta AT, Cleves MA, Casey PH et al (2002) Cognitive and behavioral outcomes of school-aged children who were born preterm: a meta-analysis. JAMA 288:728–737
van Kooij BJ, de Vries LS, Ball G et al (2012) Neonatal tract-based spatial statistics findings and outcome in preterm infants. AJNR Am J Neuroradiol 33:188–194
Hart AR, Whitby EW, Griffiths PD et al (2008) Magnetic resonance imaging and developmental outcome following preterm birth: review of current evidence. Dev Med Child Neurol 50:655–663
Arzoumanian Y, Mirmiran M, Barnes PD et al (2003) Diffusion tensor brain imaging findings at term-equivalent age may predict neurologic abnormalities in low birth weight preterm infants. AJNR Am J Neuroradiol 24:1646–1653
Counsell SJ, Boardman JP (2005) Differential brain growth in the infant born preterm: current knowledge and future developments from brain imaging. Semin Fetal Neonatal Med 10:403–410
Anderson NG, Laurent I, Woodward LJ et al (2006) Detection of impaired growth of the corpus callosum in premature infants. Pediatrics 118:951–960
Dudink J, Kerr JL, Paterson K et al (2008) Connecting the developing preterm brain. Early Hum Dev 84:777–782
de Bruine FT, van den Berg-Huysmans AA, Leijser LM et al (2011) Clinical implications of MR imaging findings in the white matter in very preterm infants: a 2-year follow-up study. Radiology 261:899–906
Keshavan MS, Diwadkar VA, DeBellis M et al (2002) Development of the corpus callosum in childhood, adolescence and early adulthood. Life Sci 70:1909–1922
Panigrahy A, Barnes PD, Robertson RL et al (2005) Quantitative analysis of the corpus callosum in children with cerebral palsy and developmental delay: correlation with cerebral white matter volume. Pediatr Radiol 35:1199–1207
Volpe JJ (2003) Cerebral white matter injury of the premature infant-more common than you think. Pediatrics 112(1 Pt 1):176–180
Counsell SJ, Shen Y, Boardman JP et al (2006) Axial and radial diffusivity in preterm infants who have diffuse white matter changes on magnetic resonance imaging at term-equivalent age. Pediatrics 117:376–386
Basser PJ, Pierpaoli C (2011) Microstructural and physiological features of tissues elucidated by quantitative-diffusion-tensor MRI. J Magn Reson 213:560–570
Assaf Y, Pasternak O (2008) Diffusion tensor imaging (DTI)-based white matter mapping in brain research: a review. J Mol Neurosci 34:51–61
Neil JJ (2008) Diffusion imaging concepts for clinicians. J Magn Reson Imagin 27:1–7
Dubb A, Gur R, Avants B et al (2003) Characterization of sexual dimorphism in the human corpus callosum. Neuroimage 20:512–519
van Pul C, van Kooij BJ, de Vries LS et al (2012) Quantitative fiber tracking in the corpus callosum and internal capsule reveals microstructural abnormalities in preterm infants at term-equivalent age. AJNR Am J Neuroradiol 33:678–684
Neil JJ, Shiran SI, McKinstry RC et al (1998) Normal brain in human newborns: apparent diffusion coefficient and diffusion anisotropy measured by using diffusion tensor MR imaging. Radiology 209:57–66
Huppi PS, Maier SE, Peled S et al (1998) Microstructural development of human newborn cerebral white matter assessed in vivo by diffusion tensor magnetic resonance imaging. Pediatr Res 44:584–590
Anjari M, Srinivasan L, Allsop JM et al (2007) Diffusion tensor imaging with tract-based spatial statistics reveals local white matter abnormalities in preterm infants. Neuroimage 35:1021–1027
Giuliani NR, Calhoun VD, Pearlson GD et al (2005) Voxel-based morphometry versus region of interest: a comparison of two methods for analyzing gray matter differences in schizophrenia. Schizophr Res 74:135–147
Kubicki M, Shenton ME, Salisbury DF et al (2002) Voxel-based morphometric analysis of gray matter in first episode schizophrenia. Neuroimage 17:1711–1719
Smith SM, Jenkinson M, Johansen-Berg H et al (2006) Tract-based spatial statistics: voxelwise analysis of multi-subject diffusion data. Neuroimage 31:1487–1505
Inder TE, Wells SJ, Mogridge NB et al (2003) Defining the nature of the cerebral abnormalities in the premature infant: a qualitative magnetic resonance imaging study. J Pediatr 143:171–179
Kier EL, Truwit CL (1996) The normal and abnormal genu of the corpus callosum: an evolutionary, embryologic, anatomic, and MR analysis. AJNR Am J Neuroradiol 17:1631–1641
Rose SE, Hatzigeorgiou X, Strudwick MW et al (2008) Altered white matter diffusion anisotropy in normal and preterm infants at term-equivalent age. Magn Reson Med 60:761–767
Skiöld B, Horsch S, Hallberg B et al (2010) White matter changes in extremely preterm infants, a population-based diffusion tensor imaging study. Acta Paediatr 99:842–849
van der Knaap MS, Valk J, Barkhof F (2005) Magnetic resonance of myelination and myelin disorders. 3rd edn. Springer, Berlin, New York
Provenzale JM, Liang L, DeLong D et al (2007) Diffusion tensor imaging assessment of brain white matter maturation during the first postnatal year. AJR Am J Radiol 189:476–486
Minkowski A; Council for International Organizations of Medical Sciences (1967) Regional development of the brain in early life: a symposium organized by the Council for International Organizations of Medical Sciences. Blackwell Scientific, Oxford, Edinburgh
Brody BA, Kinney HC, Kloman AS et al (1987) Sequence of central nervous system myelination in human infancy. I. An autopsy study of myelination. J Neuropathol Exp Neurol 46:283–301
Kinney HC, Brody BA, Kloman AS et al (1988) Sequence of central nervous system myelination in human infancy. II. Patterns of myelination in autopsied infants. J Neuropathol Exp Neurol 47:217–234
Barkovich AJ, Kjos BO, Jackson DE Jr et al (1988) Normal maturation of the neonatal and infant brain: MR imaging at 1.5 T. Radiology 166:173–180
Bird CR, Hedberg M, Drayer BP et al (1989) MR assessment of myelination in infants and children: usefulness of marker sites. AJNR Am J Neuroradiol 10:731–740
Christophe C, Muller MF, Baleriaux D et al (1990) Mapping of normal brain maturation in infants on phase-sensitive inversion–recovery MR images. Neuroradiology 32:173–178
Pierpaoli C, Jezzard P, Basser PJ et al (1996) Diffusion tensor MR imaging of the human brain. Radiology 201:637–648
Fuster JM (2002) Frontal lobe and cognitive development. J Neurocytol 31:373–385
Levitt P (2003) Structural and functional maturation of the developing primate brain. J Pediatr 143:S35–S45
Trivedi R, Agarwal S, Rathore RK et al (2009) Understanding development and lateralization of major cerebral fiber bundles in pediatric population through quantitative diffusion tensor tractography. Pediatr Res 66:636–641
Brooks-Gunn J, McCarton CM, Casey PH et al (1994) Early intervention in low-birth-weight premature infants. Results through age 5 years from the Infant Health and Development Program. JAMA 272:1257–1262
McCarton CM, Wallace IF, Bennett FC (1996) Early intervention for low-birth-weight premature infants: what can we achieve? Ann Med 28:221–225
Melnyk BM, Alpert-Gillis L, Feinstein NF (2001) Improving cognitive development of low-birth-weight premature infants with the COPE program: a pilot study of the benefit of early NICU intervention with mothers. Res Nurse Health 24:373–389
Cameron EC, Maehle V, Reid J (2005) The effects of an early physical therapy intervention for very preterm, very low birth weight infants: a randomized controlled clinical trial. Pediatr Phys Ther 17:107–119
Spittle AJ, Orton J, Doyle LW et al (2007) Early developmental intervention programs post hospital discharge to prevent motor and cognitive impairments in preterm infants. Cochrane Database Syst Rev 18:CD005495
Acknowledgement
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2012-013997).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, A.Y., Jang, S.H., Lee, E. et al. Radiologic differences in white matter maturation between preterm and full-term infants: TBSS study. Pediatr Radiol 43, 612–619 (2013). https://doi.org/10.1007/s00247-012-2545-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-012-2545-5