Abstract
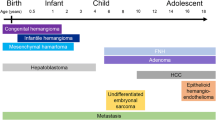
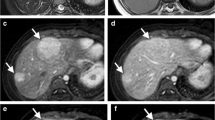
Gadoxetate disodium (Gd-EOB-DTPA) is a hepatobiliary MRI contrast agent widely used in adults for characterization of liver tumors and increasingly used in children. Hepatoblastoma is the most common primary hepatic malignancy of childhood. In this review, we describe our experience with this agent both before and after initiating therapy in children with hepatoblastoma.









Similar content being viewed by others
References
Ringe KI, Husarik DB, Sirlin CB et al (2010) Gadoxetate disodium-enhanced MRI of the liver: part 1, protocol optimization and lesion appearance in the noncirrhotic liver. AJR 195:13–28
Cruite I, Schroeder M, Merkle EM et al (2010) Gadoxetate disodium-enhanced MRI of the liver: part 2, protocol optimization and lesion appearance in the cirrhotic liver. AJR 195:29–41
Meyers AM, Towbin AJ, Serai A et al (2011) Characterization of pediatric liver lesions with gadoxetate disodium. Pediatr Radiol 41:1183–1197
Zech CJ, Herrmann KA, Reiser MF et al (2007) MR imaging in patients with suspected liver metastases: value of liver-specific contrast agent Gd-EOB-DTPA. Magn Reson Med Sci 6:43–52
Choi JY, Kim MJ, Park YM et al (2011) Gadoxetate disodium-enhanced hepatobiliary phase MRI of hepatocellular carcinoma: correlation with histological characteristics. AJR 197:399–405
Goodwin MD, Dobson JE, Sirlin CB et al (2011) Diagnostic challenges and pitfalls in MR imaging with hepatocyte-specific contrast agents. Radiographics 31:1547–1568
Vasanawala S (2010) MRI of the liver—how to do it. Pediatr Radiol 40:431–437
Marrone G, Maggiore G, Carollo V et al (2011) Biliary cystadenoma with bile duct communication depicted on liver-specific contrast agent-enhanced MRI in a child. Pediatr Radiol 41:121–124
Tamrazi A, Vasanawala SS (2011) Functional hepatobiliary MR imaging in children. Pediatr Radiol 22:1250–1258
Das CJ, Dhingra S, Gupta AK et al (2009) Imaging of paediatric liver tumours with pathological correlation. Clin Radiol 64:1015–1025
Imbach P (2006) Hepatic tumors. In: Imbach P, Kuhne T, Arceci RJ (eds) Pediatric oncology: a comprehensive guide. Springer, New York, pp 187–191
Chung EM, Lattin GE, Cube R et al (2011) Pediatric liver masses: radiologic-pathologic correlation part 2. Malignant tumors. Radiographics 31:483–507
Roebuck DJ, Aronson D, Clapuyt P et al (2005) PRETEXT: a revised staging system for primary malignant liver tumours of childhood developed by the SIOPEL group. Pediatr Radiol 37:123–132
Vogel TJ, Kummel S, Hammerstingl R et al (1996) Liver tumors: comparison of MR imaging with Gd-EOB-DTPA and Gd-DTPA. Radiology 200:59–67
Reimer P, Rummeny EJ, Shamsi K et al (1996) Phase II clinical evaluation of Gd-EOB-DTPA: dose, safety aspects, and pulse sequence. Radiology 199:177–183
Towbin AJ, Luo GG, Yin H et al (2010) Focal nodular hyperplasia in children, adolescents, and young adults. Pediatr Radiol 41:341–349
Asbach P, Klessen C, Koch M et al (2007) Magnetic resonance imaging findings of atypical focal nodular hyperplasia of the liver. Clin Imaging 31:244–252
Christison-Lagay ER, Burrows PE, Alomari A et al (2007) Hepatic hemangiomas: subtype classification and development of a clinical practice algorithm and registry. J Pediatr Surg 42:62–67
Goshima S, Kanematsu M, Watanabe H et al (2010) Hepatic hemangioma and metastasis: differentiation with gadoxetate disodium-enhanced 3-T MRI. AJR 195:941–946
Prokurat A, Kluge P, Kosciesza A et al (2002) Transitional liver cell tumors (TLCT) in older children and adolescents: a novel group of aggressive hepatic tumors expressing beta-catenin. Med Pediatr Oncol 39:510–518
Author information
Authors and Affiliations
Corresponding author
Additional information
CME activity
This article has been selected as the CME activity for the current month. Please visit the SPR Web site at www.pedrad.org on the Education page and follow the instructions to complete this CME activity.
Rights and permissions
About this article
Cite this article
Meyers, A.B., Towbin, A.J., Geller, J.I. et al. Hepatoblastoma imaging with gadoxetate disodium-enhanced MRI—typical, atypical, pre- and post-treatment evaluation. Pediatr Radiol 42, 859–866 (2012). https://doi.org/10.1007/s00247-012-2366-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-012-2366-6