Abstract
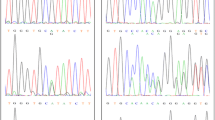
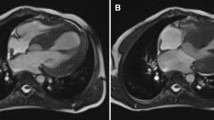
Pediatric primary cardiomyopathy is rare but serious, having high mortality; hypertrophic and dilated types are the most common. Its etiology has been mainly considered idiopathic; however, next generation sequencing techniques have revealed nearly half of idiopathic pediatric cases arose from specific genetic mutations. Therefore, our study aimed to identify the genetic causes of primary idiopathic cardiomyopathy. Newborns to 15-year old patients with this condition were recruited between March 2016 and May 2017 at Thammasat University Hospital. Complete patient history and physical examination data were collected by a geneticist with cardiac examinations and echocardiograms by pediatric cardiologists. Whole exome sequencing was performed for all. Of the 12 patients enrolled, 5 cases were dilated type and 7 hypertrophic. Two with dilated type were excluded during follow-up as cause was determined (hypocalcemia and pacemaker induced). A list of 118 genes for cardiomyopathy was analyzed in the remaining 10 cases. Pathogenic and likely pathogenic mutations were identified in 5 patients: HRAS, PTPN11, SOS1, FLNC and TXNRD2; half our patients were not actually idiopathic. Despite its high cost, genetic testing is useful for determining familial risk as well as predicting patient cardiomyopathy progress.

Similar content being viewed by others
References
Nugent AW, Daubeney PE, Chondros P, Carlin JB, Cheung M, Wilkinson LC, Davis AM, Kahler SG, Chow CW, Wilkinson JL, Weintraub RG, National Australian Childhood Cardiomyopathy Study (2003) The epidemiology of childhood cardiomyopathy in Australia. N Engl J Med 348:1639–1646
Wilkinson JD, Sleeper LA, Alvarez JA, Bublik N, Lipshultz SE (2008) The pediatric cardiomyopathy registry: 1995–2007. Prog Pediatr Cardiol 25:31–36
Richardson P, McKenna W, Bristow M, Maisch B, Mautner B, O'Connell J, Olsen E, Thiene G, Goodwin J, Gyarfas I, Martin I, Nordet P (1996) Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the definition and classification of cardiomyopathies. Circulation 93:841–842
Maron BJ, Towbin JA, Thiene G, Antzelevitch C, Corrado D, Arnett D, Moss AJ, Seidman CE, Young JB (2006) Contemporary definitions and classification of the cardiomyopathies: an American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation 113:1807–1816
Elliott P, Andersson B, Arbustini E, Bilinska Z, Cecchi F, Charron P, Dubourg O, Kuhl U, Maisch B, McKenna WJ, Monserrat L, Pankuweit S, Rapezzi C, Seferovic P, Tavazzi L, Keren A (2008) Classification of the cardiomyopathies: a position statement from the European Society Of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J 29:270–276
Elliott P (2008) The 2006 American Heart Association classification of cardiomyopathies is not the gold standard. Circ Heart Fail 1:77–80
Lee TM, Hsu DT, Kantor P, Towbin JA, Ware SM, Colan SD, Chung WK, Jefferies JL, Rossano JW, Castleberry CD, Addonizio LJ, Lal AK, Lamour JM, Miller EM, Thrush PT, Czachor JD, Razoky H, Hill A, Lipshultz SE (2017) Pediatric cardiomyopathies. Circ Res 121:855–873
Colan SD, Lipshultz SE, Lowe AM, Sleeper LA, Messere J, Cox GF, Lurie PR, Orav EJ, Towbin JA (2007) Epidemiology and cause-specific outcome of hypertrophic cardiomyopathy in children: findings from the pediatric cardiomyopathy registry. Circulation 115:773–781
Towbin JA, Lowe AM, Colan SD, Sleeper LA, Orav EJ, Clunie S, Messere J, Cox GF, Lurie PR, Hsu D, Canter C, Wilkinson JD, Lipshultz SE (2006) Incidence, causes, and outcomes of dilated cardiomyopathy in children. JAMA 296:1867–1876
Kindel SJ, Miller EM, Gupta R, Cripe LH, Hinton RB, Spicer RL, Towbin JA, Ware SM (2012) Pediatric cardiomyopathy: importance of genetic and metabolic evaluation. J Card Fail 18:396–403
Norton N, Li D, Hershberger RE (2012) Next-generation sequencing to identify genetic causes of cardiomyopathies. Curr Opin Cardiol 27:214–220
Ellepola CD, Knight LM, Fischbach P, Deshpande SR (2017) Genetic testing in pediatric cardiomyopathy. Pediatr Cardiol 3:491–500
Long PA, Evans JM, Olson TM (2017) Diagnostic yield of whole exome sequencing in pediatric dilated cardiomyopathy. J Cardiovasc Dev Dis 4:1–10
Cirino AL, Ho C (1993) [Updated 2019 Jun 6] Hypertrophic cardiomyopathy overview. In: Adam, MP, Ardinger, HH, Pagon, RA, Wallace, SE, Bean, LJH, Stephens, K, Amemiya, A (eds) GeneReviews®. University of Washington, Seattle, 1993–2019. https://www.ncbi.nlm.nih.gov/books/NBK1768/. Accessed 2 Sep 2019
Hershberger RE, Morales A (1993) [Updated 2018 Aug 23] Dilated cardiomyopathy overview. In: Adam, MP, Ardinger, HH, Pagon, RA, Wallace, SE, Bean, LJH, Stephens, K, Amemiya, A (eds) GeneReviews®. University of Washington, Seattle; 1993–2019. https://www.ncbi.nlm.nih.gov/books/NBK1309/. Accessed 13 Feb 2019
Pettersen MD, Du W, Skeens ME, Humes RA (2008) Regression equations for calculation of z scores of cardiac structures in a large cohort of healthy infants, children, and adolescents: an echocardiographic study. J Am Soc Echocardiogr 21:922–934
Daubeney PE, Nugent AW, Chondros P, Carlin JB, Colan SD, Cheung M, Davis AM, Chow CW, Weintraub RG (2006) Clinical features and outcomes of childhood dilated cardiomyopathy: results from a national population-based study. Circulation 114:2671–2678
Gersh BJ, Maron BJ, Bonow RO, Dearani JA, Fifer MA, Link MS, Naidu SS, Nishimura RA, Ommen SR, Rakowski H, Seidman CE, Towbin JA, Udelson JE, Yancy CW (2011) 2011 ACCF/AHA guideline for the diagnosis and treatment of hypertrophic cardiomyopathy: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Thorac Cardiovasc Surg 142:e153–203
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, Voelkerding K, Rehm HL, On behalf of the ALQAC (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–424
Yang Y, Muzny DM, Xia F, Niu Z, Person R, Ding Y, Ward P, Braxton A, Wang M, Buhay C, Veeraraghavan N, Hawes A, Chiang T, Leduc M, Beuten J, Zhang J, He W, Scull J, Willis A, Landsverk M, Craigen WJ, Bekheirnia MR, Stray-Pedersen A, Liu P, Wen S, Alcaraz W, Cui H, Walkiewicz M, Reid J, Bainbridge M, Patel A, Boerwinkle E, Beaudet AL, Lupski JR, Plon SE, Gibbs RA, Eng CM (2014) Molecular findings among patients referred for clinical whole-exome sequencing. JAMA 312:1870–1879
Lee H, Deignan JL, Dorrani N, Strom SP, Kantarci S, Quintero-Rivera F, Das K, Toy T, Harry B, Yourshaw M, Fox M, Fogel BL, Martinez-Agosto JA, Wong DA, Chang VY, Shieh PB, Palmer CG, Dipple KM, Grody WW, Vilain E, Nelson SF (2014) Clinical exome sequencing for genetic identification of rare Mendelian disorders. JAMA 312:1880–1887
Tartaglia M, Pennacchio LA, Zhao C, Yadav KK, Fodale V, Sarkozy A, Pandit B, Oishi K, Martinelli S, Schackwitz W, Ustaszewska A, Martin J, Bristow J, Carta C, Lepri F, Neri C, Vasta I, Gibson K, Curry CJ, Siguero JP, Digilio MC, Zampino G, Dallapiccola B, Bar-Sagi D, Gelb BD (2007) Gain-of-function SOS1 mutations cause a distinctive form of Noonan syndrome. Nat Genet 39:75–79
Aoki Y, Niihori T, Kawame H, Kurosawa K, Ohashi H, Tanaka Y, Filocamo M, Kato K, Suzuki Y, Kure S, Matsubara Y (2005) Germline mutations in HRAS proto-oncogene cause Costello syndrome. Nat Genet 37:1038–1040
Takahashi K, Kogaki S, Kurotobi S, Nasuno S, Ohta M, Okabe H, Wada K, Sakai N, Taniike M, Ozono K (2005) A novel mutation in the PTPN11 gene in a patient with Noonan syndrome and rapidly progressive hypertrophic cardiomyopathy. Eur J Pediatr 164:497–500
Ortiz-Genga MF, Cuenca S, Dal Ferro M, Zorio E, Salgado-Aranda R, Climent V, Padrón-Barthe L, Duro-Aguado I, Jiménez-Jáimez J, Hidalgo-Olivares VM, García-Campo E, Lanzillo C, Suárez-Mier MP, Yonath H, Marcos-Alonso S, Ochoa JP, Santomé JL, García-Giustiniani D, Rodríguez-Garrido JL, Domínguez F, Merlo M, Palomino J, Peña ML, Trujillo JP, Martín-Vila A, Stolfo D, Molina P, Lara-Pezzi E, Calvo-Iglesias FE, Nof E, Calò L, Barriales-Villa R, Gimeno-Blanes JR, Arad M, García-Pavía P, Monserrat L (2016) Truncating FLNC mutations are associated with high-risk dilated and arrhythmogenic cardiomyopathies. J Am Coll Cardiol 68:2440–2451
Tucker NR, McLellan MA, Hu D, Ye J, Parsons VA, Mills RW, Clauss S, Dolmatova E, Shea MA, Milan DJ, Scott NS, Lindsay M, Lubitz SA, Domian IJ, Stone JR, Lin H, Ellinor PT (2017) Novel mutation in FLNC (filamin C) causes familial restrictive cardiomyopathy. Circulation 10:e001780
Valdes-Mas R, Gutierrez-Fernandez A, Gomez J, Coto E, Astudillo A, Puente DA, Reguero JR, Alvarez V, Moris C, Leon D, Martin M, Puente XS, Lopez-Otin C (2014) Mutations in filamin C cause a new form of familial hypertrophic cardiomyopathy. Nat Commun 5:5326
Begay RL, Graw SL, Sinagra G, Asimaki A, Rowland TJ, Slavov DB, Gowan K, Jones KL, Brun F, Merlo M, Miani D, Sweet M, Devaraj K, Wartchow EP, Gigli M, Puggia I, Salcedo EE, Garrity DM, Ambardekar AV, Buttrick P, Reece TB, Bristow MR, Saffitz JE, Mestroni L, Taylor MRG (2018) Filamin C truncation mutations are associated with arrhythmogenic dilated cardiomyopathy and changes in the cell–cell adhesion structures. JACC Clin Electrophysiol 4:504–514
Mangum KD, Ferns SJ (2019) A novel familial truncating mutation in the filamin C gene associated with cardiac arrhythmias. Eur J Med Genet 62:282–285
Berndt C, Lillig CH, Holmgren A (2007) Thiol-based mechanisms of the thioredoxin and glutaredoxin systems: implications for diseases in the cardiovascular system. Am J Physiol Heart Circ Physiol 292:H1227–1236
Conrad M, Jakupoglu C, Moreno SG, Lippl S, Banjac A, Schneider M, Beck H, Hatzopoulos AK, Just U, Sinowatz F, Schmahl W, Chien KR, Wurst W, Bornkamm GW, Brielmeier M (2004) Essential role for mitochondrial thioredoxin reductase in hematopoiesis, heart development, and heart function. Mol Cell Biol 24:9414–9423
Sibbing D, Pfeufer A, Perisic T, Mannes AM, Fritz-Wolf K, Unwin S, Sinner MF, Gieger C, Gloeckner CJ, Wichmann HE, Kremmer E, Schafer Z, Walch A, Hinterseer M, Nabauer M, Kaab S, Kastrati A, Schomig A, Meitinger T, Bornkamm GW, Conrad M, von Beckerath N (2011) Mutations in the mitochondrial thioredoxin reductase gene TXNRD2 cause dilated cardiomyopathy. Eur Heart J 32:1121–1133
Prasad R, Chan LF, Hughes CR, Kaski JP, Kowalczyk JC, Savage MO, Peters CJ, Nathwani N, Clark AJ, Storr HL, Metherell LA (2014) Thioredoxin reductase 2 (TXNRD2) mutation associated with familial glucocorticoid deficiency (FGD). J Clin Endocrinol Metab 99:E1556–1563
Badano JL, Katsanis N (2002) Beyond Mendel: an evolving view of human genetic disease transmission. Nat Rev Genet 3:779–789
Rauen KA (2013) The RASopathies. Annu Rev Genom Hum Genet 14:355–369
Gelb BD, Roberts AE, Tartaglia M (2015) Cardiomyopathies in Noonan syndrome and the other RASopathies. Prog Pediatr Cardiol 39:13–19
Kerr B, Delrue MA, Sigaudy S, Perveen R, Marche M, Burgelin I, Stef M, Tang B, Eden OB, O'Sullivan J, De Sandre-Giovannoli A, Reardon W, Brewer C, Bennett C, Quarell O, M'Cann E, Donnai D, Stewart F, Hennekam R, Cavé H, Verloes A, Philip N, Lacombe D, Levy N, Arveiler B, Black G (2006) Genotype-phenotype correlation in Costello syndrome: HRAS mutation analysis in 43 cases. J Med Genet 43:401–405
Zhang N, Chen L, Zhang J (2019) Septal myectomy for severe neonatal hypertrophic cardiomyopathy caused by PTPN11 gene mutation: a case report. Clin Surg 4:2566
Zenker M, Horn D, Wieczorek D, Allanson J, Pauli S, van der Burgt I, Doerr HG, Gaspar H, Hofbeck M, Gillessen-Kaesbach G, Koch A, Meinecke P, Mundlos S, Nowka A, Rauch A, Reif S, von Schnakenburg C, Seidel H, Wehner LE, Zweier C, Bauhuber S, Matejas V, Kratz CP, Thomas C, Kutsche K (2007) SOS1 is the second most common Noonan gene but plays no major role in cardio-facio-cutaneous syndrome. J Med Genet 44:651–656
Hershberger RE, Givertz M, Ho CY, Judge DP, Kantor P, McBride KL, Morales A, Taylor MRG, Vatta M, Ware SM (2018) Genetic evaluation of cardiomyopathy: a heart failure society of america practice guideline. J Card Fail 24:281–302
Cox GF (2007) Diagnostic approaches to pediatric cardiomyopathy of metabolic genetic etiologies and their relation to therapy. Prog Pediatr Cardiol 24:15–25
Acknowledgements
We would like to thank patients and families who were part of this study, the Faculty of Medicine, Thammasat University for providing funding (Grant TP.2-10/2559); and Faculty of Medicine Ramathibodi Hospital, Mahidol University for providing Research Career Award to DW during the time of this study. We also thank Ms Debra Kim Liwiski, writer/international instructor, the Clinical Research Center, Faculty of Medicine, Thammasat University for language editing.
Author information
Authors and Affiliations
Contributions
KR designed the study, performed and interpreted the genetic tests and wrote the manuscript. BS and RW performed echocardiograms and reviewed the manuscript. TK performed genetic testing. SN and AK reviewed the whole exome results, and DW supervised overall concepts designed and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in the present study involving human participants were in accordance with the ethical standards of the Human Research Ethics Committee of Thammasat University No.1 (Faculty of Medicine: MTU-EC-PE-1-191/58) and the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual parents and assent from participants (if possible) included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rojnueangnit, K., Sirichongkolthong, B., Wongwandee, R. et al. Identification of Gene Mutations in Primary Pediatric Cardiomyopathy by Whole Exome Sequencing. Pediatr Cardiol 41, 165–174 (2020). https://doi.org/10.1007/s00246-019-02240-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-019-02240-x