Abstract
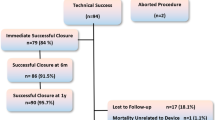
In August 2010, the Nit-Occlud® Lê (EUREVECO) became available for transcatheter coil occlusion of ventricular septal defects (VSDs). Retrospective European Registry for VSD Closure using the Nit-Occlud® Lê-VSD-Coil; analysis of the feasibility, results, safety and follow-up of VSD-closure over a 3-year period in 18 European centers. In 102 of 111 patients (female 66), successful VSD closure was performed (mean age 8.2 years, mean weight 28.82 kg), 81 perimembranous VSDs (48 with aneurysm), 30 muscular VSDs, mean procedure time was 121.1 min, and mean fluoroscopy time was 26.3 min. Short- and midterm term follow-up was possible in 100/102 patients, there was 1 embolization and 1 explantation after 24 months. Immediate complete closure occurred in 49 of 101 patients (48.5%), trivial residual shunt was present in 51 (50.0%), closure rate was 95% after 6 months and 97% after 1 year. Out of the 102 patients, there were 2 severe complications (1.8%) (1 severe hemolysis, 1 embolization) and 8 moderate/transient (=7.2%) including 1 transient AV block. During a mean follow-up period of 31.3 months (range 24–48) and a total follow-up time of 224.75 patient years, no further problems occurred. VSD closure with the Nit-Occlud® Lê VSD coil is feasible and safe with a minimal risk of severe side effects. The long-term effects and safety require further clinical follow-up studies.





Similar content being viewed by others
Abbreviations
- VSD:
-
Ventricular septum defect
- Nit-Occlud® Lê-VSD-Coil:
-
Coil used in this study
- EUREVECO:
-
European Registry for Ventricular Septal Defect Coil Occlusion
- LA:
-
Left atrium
- LV:
-
Left ventricle
- PA:
-
Pulmonary artery
- RV:
-
Right ventricle
- ECHO:
-
Echocardiography
- ECG:
-
Electrocardiogram
- TTE:
-
Transthoracic echocardiography
- TOE:
-
Transesophageal echocardiography
- RBBB:
-
Right bundle branch block
- AVBlock III°:
-
Third-degree complete atrioventricular block
References
Lock JE, Block PC, McKay RG, Baim DS, Keane JF (1988) Transcatheter closure of VSDs. Circulation 78:361–368
Bass JL, Kalra GS, Arora R, Masura J, Gavora P, Thanopoulos BD, Torres W, Sievert H, Carminati M, Fischer G, Ewert P (2003) Initial human experience with the Amplatzer perimembranous ventricular septal occluder device. Catheter Cardiovasc Interv 58:238–245
Yang J, Yang L, Yu S, Liu J, Zuo J, Chen W, Duan W, Zheng Q, Xu X, Li J, Zhang J, Xu J, Sun L, Yang X, Xiong L, Yi D, Wang L, Liu Q, Ge S, Ren J (2014) Transcatheter versus surgical closure of perimembranous ventricular septal defects in children: a randomized controlled trial. J Am Coll Cardiol 63(12):1159–1168
Oses P, Hugues N, Dahdah N, Vobecky SJ, Miro J, Pellerin M, Poirier NC (2010) Treatment of isolated ventricular septal defects in children: Amplatzer versus surgical closure. Ann Thorac Surg 90(5):1593–1598
Masura J, Gao W, Gavora P, Sun K, Zhou AQ, Jiang S, Ting-Liang L, Wang Y (2005) Percutaneous closure of perimembranous ventricular septal defects with the eccentric Amplatzer device: multicenter follow-up study. Pediatr Cardiol 26(3):216–219
Carminati M, Butera G, Chessa M, De Giovanni J, Fisher G, Gewillig M, Peuster M, Piechaud JF, Santoro G, Sievert H, Spadoni I, Walsh K, Investigators of the European VSD Registry (2007) Transcatheter closure of congenital VSDs: results of the European Registry. Eur Heart J 28:2361–2368
Chungsomprasong P, Durongpisitkul K, Vijarnsorn C, Soongswang J, Lê TP (2011) The results of transcatheter closure of VSD using Amplatzer® device and Nit Occlud® Lê coil. Catheter Cardiovasc Interv 78:1032–1040
Odemis E, Saygi M, Guzeltas A, Tanidir IC, Ergul Y, Ozyilmaz I, Bakir I (2014) Transcatheter closure of perimembranous VSDs using Nit-Occlud(®) Lê VSD coil: early and mid-term results. Pediatr Cardiol 35:817–823
Bridges ND, Perry SB, Keane JF, Goldstein SA, Mandell V, Mayer JE Jr, Jonas RA, Casteneda AR, Lock JE (1991) Preoperative transcatheter closure of congenital muscular VSDs. N Engl J Med 324(19):1312–1317
Diab KA, Cao Q-L, Mora B-N, Hijazi ZM (2007) Device closure of muscular VSDs in infants less than one year of age using the Amplatzer devices: feasibility and outcome. Catheter Cardiovasc Interv 70:90–97
Wang J, Zuo J, Yu S, Yi D, Yang X, Zhu X, Li J, Yang L, Xiong L, Ge S, Ren J, Yang J (2016) Effectiveness and safety of transcatheter closure of perimembranous ventricular septal defects in adults. Am J Cardiol 117(6):980–987
Ewert P, Kretschmar O, Peters B, Abdul-Khaliq H, Nagdyman N, Schulze-Neick I, Bass J, Lê TP, Lange PE (2004) Transcatheter closure of congenital septal defects. Z Kardiol 93:147–155
Michel- Behnke I, Le T-P, Waldecker B, Akintuerk H, Valeske K, Schranz D (2005) Percutaneous closure of congenital and acquired VSDs. J Interv Cardiol 18:89–99
Zartner P, Christians C, Stelter JC, Hraška V, Schneider MB (2014) Transvascular closure of single and multiple muscular VSDs in neonates and infants <20 kg. Catheter Cardiovasc Interv 83:564–570
Zhou D, Pan W, Guan L, Ge J (2012) Transcatheter closure of perimembranous VSDs and intracristal ventricular defects with the SHSMA Occluder. Catheter Cardiovasc Interv 79:666–674
Yang J, Yang L, Wan Y, Zuo J, Zhang J, Chen W, Li J, Sun L, Yu S, Liu J, Chen T, Duan W, Xiong L, Yi D (2010) Transcatheter device closure of perimembranous VSDs. Eur Heart J 31(18):2238–2245
Bjørnstad PG, Smevik B, Fischer G (2010) Catheter based closure of VSDs. Scand Cardiovasc J 44:9–14
Bentham JR, Gujral A, Adwani S, Archer N, Wilson N (2011) Does the technique of interventional closure of perimembranous VSD reduce the incidence of heart block? Cardiol Young 21:271–280
Bass JL, Gruenstein D (2012) Transcatheter closure of the perimembranous VSD-preclinical trial of a new Amplatzer device. Catheter Cardiovasc Interv 79:1153–1160
Tzikas A, Ibrahim R, Velasco-Sanchez D, Freixa X, Alburquenque M, Khairy P, Bass JL, Ramirez J, Aguirre D, Miro J (2014) Transcatheter closure of perimembranous VSD with the Amplatzer(®) membranous VSD occluder 2. Catheter Cardiovasc Interv 83:571–580
El Said HG, Bratincsak A, Gordon BM, Moore JW (2012) Closure of perimembranous VSDs with aneurysmal tissue using the Amplazter Duct Occluder I. Catheter Cardiovasc Interv 80:895–903
Lee SM, Song JY, Choi JY, Lee SY, Paik JS, Chang SI, Shim WS, Kim SH (2013) Transcatheter closure of perimembranous VSD using Amplatzer ductal occluder. Catheter Cardiovasc Interv 82:1141–1146
Koneti NR, Verma S, Bakhru S, Vadlamudi K, Kahtare P, Penumatsa RR, Qureshi S (2013) Transcatheter trans-septal antegrade closure of muscular VSD in young children. Cath Cardiovasc Interv 82(4):E500–E506
Kanaan M, Ewert P, Berger F, Assa S, Schubert S (2015) Follow-up of patients with interventional closure of VSDs with Amplatzer Duct Occluder II. Pediatr Cardiol 36:379–385
Nogi S, Haneda N, Tomita H, Yasuda K (2008) Trancatheter coil occlusion of perimembranous VSDs. Catheter Cardiovasc Interv 72:683–690
Yang L, Tai BC, Khin LW, Quek SC (2014) A systematic review on the efficacy and safety of transcatheter device closure of VSDs. J Interv Cardiol 27:260–272
Erdem S, Kizlltaş A, Küçükosmanoğlu O, Ozbarlas N (2012) Temporary atrioventricular complete block that develops following the transcatheter closure of VSD. Turk J Pediatr 54:80–82
Holzer R, de Giovanni J, Walsh KP, Tometzki A, Goh T, Hakim F, Zabal C, de Lezo JS, Cao QL, Hijazi ZM (2006) Transcatheter closure of perimembranous VSD using the Amplatzer membranous VSD occluder. Catheter Cardiovasc Interv 68:620–628
Zuo J, Xie J, Yi W, Yang J, Zhang J, Li J, Yi D (2010) Results of transcatheter closure of perimembranous VSD. Am J Cardiol 106:1034–1037
Li X, Li L, Wang X, Zhao H-B, Zhang S-Y (2011) Clinical analysis of transcatheter closure of perimembranous VSD with occluders made in China. Chin Med J 2011(124):2117–2122
Fu YC, Bass J, Amin Z, Radtke W, Cheatham JP, Hellenbrand WE, Balzer D, Cao QL, Hijazi ZM (2006) Transcatheter closure of perimembranous VSDs using the new Amplatzer membranous VSD occluder. J Am Coll Cardiol 47:319–325
Funding
The funding is by hospital funding only.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest by any of the authors regarding this report.
Additional information
Nikolaus A. Haas and Laura Kock have equally shared the authorship in this publication.
Rights and permissions
About this article
Cite this article
Haas, N.A., Kock, L., Bertram, H. et al. Interventional VSD-Closure with the Nit-Occlud® Lê VSD-Coil in 110 Patients: Early and Midterm Results of the EUREVECO-Registry. Pediatr Cardiol 38, 215–227 (2017). https://doi.org/10.1007/s00246-016-1502-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-016-1502-8