Abstract
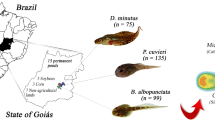
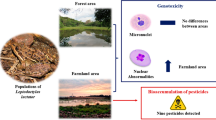
Many agricultural practices cause environmental degradation that affects the cellular integrity of anurans. In the present study, we provided in situ data of Dendropsophus minutus, Physalaemus cuvieri, and Scinax fuscovarius collected in soybean/corn and conservation units in the Brazilian Cerrado. The in situ data showed no significant variation in the micronucleus frequency between the sites, only the reniform cells had a higher rate for the agricultural environment. A combined analysis of all nuclear erythrocyte abnormalities (ENAs = nuclear buds, reniform nuclei, apoptotic cell, binucleated, and anucleated cells) was recorded higher frequencies in farmland. Overall, Scinax fuscovarius was considered the best potential bioindicator for soybean/corn plantations. Finally, we recommend expanding the micronucleus test for in situ studies to expand our understanding of the sensitivity of native anuran species and provide a more systematic assessment of the adverse effects of environmental pollutants on wildlife.


Similar content being viewed by others
References
Anderson S, Sadinski W, Shugart L, Brussard P, Depledge M, Ford T, Hose J, Stegeman J, Suk W, Wirgin I, Wogan G (1994) Genetic and molecular ecotoxicology: a research framework. Environ Health Perspect 102:3–8
Antunes SC, Castro BB, Nunes B, Pereira R, Gonçalves F (2008) In situ bioassay with Eisenia andrei to assess soil toxicity in an abandoned uranium mine. Ecotoxicol Environ Saf 71:620–631
Arcaute CR, Pérez-Inglesis JM, Nikoloff N, Natale GS, Soloneski S, Larramendy ML (2014) Genotoxicity evaluation of the insecticide imidacloprid on circulating blood cells of Montevideo tree frog Hypsiboa spulchellus tadpoles (Anura, Hylidae) by comet and micronucleus bioassays. Ecol Indic 45:632–639
Babini MS, Bionda CL, Salas NL, Martino AL (2016) Adverse effect of agroecosystem pond water on biological endpoints of common toad (Rhinella arenarum) tadpoles. Environ Monitor Assess 188:459
Barni S, Boncompagni E, Grosso A, Bertone V, Freitas I, Fasola M, Fenoglio C (2007) Evaluation of Rana snkesculenta blood cell response to chemical stressors in the environment during the larval and adult phases. Aquat Toxicol 81:45–54
Berger L (1989) Disappearance of amphibian larvae in the agricultural landscape. Ecol Int Bull 17:65–73
Borges RE, Santos LRS, Assis RA, Benvindo-Souza M, Franco-Belussi L, Oliveira C (2019) Monitoring the morphological integrity of neotropical anurans. Environ Sci Pollut Res 26:2623–2634
Brodeur JC, Svartz G, Perez-Coll CS, Marino DJG, Herkovits J (2009) Comparative susceptibility to atrazine of three developmental stages of Rhinella arenarum and influence on metamorphosis: non-monotonous acceleration of the time to climax and delayed tail resorption. Aquat Toxicol 91:161–170
Brühl CA, Pieper S, Weber B (2011) Amphibians at risk? Susceptibility of terrestrial amphibian life stages to pesticides. Environ Toxicol Chem 30:2465–2472
Castro BB, Guilhermino L, Ribeiro R (2003) In situ bioassay chambers and procedures for assessment of sediment toxicity with Chironomus riparius. Environ Pollut 125:325–335
Cavas T, Ergene-Gözükara S (2005) Induction of micronuclei and nuclear abnormalities in Oreochromis niloticus following exposure to petroleum refinery and chromium processing plant effluents. Aquat Toxicol 74:264–271
Cruz-Esquivel A, Viloria-Rivas J, Marrugo-Negrete J (2017) Genetic damage in Rhinella marina populations in habitats affected by agriculture in the middle region of the SinA River, Colombia. Environ Sci Pollut Res 24:27392–27401
Depledge MH, Fossi MC (1994) The role of biomarkers in environmental assessment. Ecotoxicology 3:161–172
Dias LCP, Pimenta FM, Santos AB, Costa MH, Ladle RJ (2016) Patterns of land use, extensification, and intensification of Brazilian agriculture. Glob Change Biol 22:2887–2903
Ferreira CM, Guimarães HMB, Ranzani-Paiva MJT, Soares SR, Riviero DHRF, Saldiva PHN (2003) Hematological markers of copper toxicity in Rana catesbeiana tadpoles (Bullfrog). Revista Brasil Toxicol 16:83–88
Garaj-Vrhovac V, Gajski G, Ravlić S (2008) Efficacy of HUMN criteria for scoring the micronucleus assay in human lymphocytes exposed to a low concentration of p,p′-DDT. Braz J Med Biol Res 41:473–476
Gehara M, Crawford AJ, OrricoVGD RodríguezA et al (2014) High levels of diversity uncovered in a widespread nominal taxon: continental phylogeography of the neotropical tree frog Dendropsophus minutus. PLoS ONE 9:e111829
GökalpMuranli FD, Güner U (2011) Induction of micronuclei and nuclear abnormalities in erythrocytes of mosquito fish (Gambusia affinis) following exposure to the pyrethroid insecticide lambda-cyhalothrin. Mut Res 726:104–108
Gonçalves MW, Vieira TB, Maciel NM, Carvalho WF, Lima LSF, Gambale PG, da Cruz AD, Nomura F, Bastos RP, Silva DM (2015) Detecting genomic damages in the frog Dendropsophus minutus: preserved versus perturbed areas. Environ Sci Pollut Res 22:3947–3954
Gonçalves MW, Gambale PG, Godoy FR, Alves AA, Rezende PHD, Maciel NM, Nomura F, Bastos RP, Marco P (2017a) The agricultural impact of pesticides on Physalaemus cuvieri tadpoles (Amphibia: Anura) ascertained by comet assay. Zoologia 4:e19865. http://zoobank.org/A65FFC07-75B6-4DE4-BE59-8CE6BB2D4448
Gonçalves MW, Campos CBM, Batista VG, Cruz AD, Marco Junior P, Bastos RP, Silva DDM (2017b) Genotoxic and mutagenic effects of Atrazine Atanor 50 SC on Dendropsophus minutus Peters, 1872 (Anura: Hylidae) developmental larval stages. Chemosphere 182:730–737
Gosner KL (1960) A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16:183–190. https://www.jstor.org/stable/3890061
Hayes TB, Case P, Chui S, Chung D, Haeffele C, Haston K, Lee M, Mai VP, Marjuoa Y, Parker J (2006) Pesticide mixtures, endocrine disruption, and Amphibian declines: are we underestimating the impact? Environ Health Perspect 114:40–50
Hayes TB, Falso P, Gallipeau S, Stice M (2010) The cause of global amphibian declines: a developmental endocrinologist’s perspective. J Exp Biol 213:921–933
Jing X, Yao GJ, Liu DH, Liu C, Wang F, Wang P, Zhou ZQ (2017) Exposure of frogs and tadpoles to chiral herbicide fenoxaprop-ethyl. Chemosphere 186:832–838
Josende ME, Tozetti MA, Alalan TM, Filho MV, Ximenez SS, Silva Júnior FMR, Martins ES (2015) Genotoxic evaluation in two amphibian species from Brazilian subtropical wetlands. Ecol Indic 49:83–87
Krauter PW, Anderson SL, Harrison FL (1987) Radiation-induced micronuclei in peripheral erythrocytes of Rana catesbeiana: an aquatic animal model for in vivo genotoxicity studies. Environ Molec Mutagenesis 10:285–296
Kurelec B (1993) The genetic disease syndrome. Mar Environ Res 35:341–348
Lajmanovich RC, Cabagna-Zenklusen MC, Attademo AM, Junges CM, Peltzer PM, Basso A, Lorenzatti E (2014) Induction of micronuclei and nuclear abnormalities in tadpoles of the common toad (Rhinella arenarum) treated with the herbicides Liberty® and glufosinate–ammonium. Mutat Res Genetic Toxicol Environ Mutagenesis 769:7–12
Mann RM, Hyne RV, Choung CB, Wilson SP (2009) Amphibians and agricultural chemicals: review of the risks in a complex environment. Environ Pollut 157:2903–2927
Margarido TCS, Felicio AA, Rossa-Feres DD, de Almeida EA (2013) Biochemical biomarkers in Scinax fuscovarius tadpoles exposed to a commercial formulation of the pesticide fipronil. Mar Environ Res 91:61–67
Mijares A, Rodrigues MT, Baldo D (2010) Physalaemus cuvieri. Lista Vermelha de Espécies Ameaçadas da IUCN 2010: e.T57250A11609155. http://dx.doi.org/10.2305/IUCN.UK.2010-2.RLTS.T57250A11609155.en
Montalvão MF, Malafaia G (2017) Effects of abamectin on bullfrog tadpoles: insights on cytotoxicity. Environ Sci Pollut Res 24:23411–23416
Natale GS, Vera-Candioti J, de Arcaute CR, Soloneski S, Larramendy ML, Ronco AE (2018) Lethal and sublethal effects of the pirimicarb-based formulation Aficida (R) on Boana pulchella (Dumeril and Bibron, 1841) tadpoles (Anura, Hylidae). Ecotoxicol Environ Saf 147:471–479
Nikoloff N, Natale GS, Marino D, Soloneski S, Larramendy ML (2014) Flurochloridone-based herbicides induced genotoxicity effects on Rhinella arenarum tadpoles (Anura: Bufonidae). Ecotoxicol Environ Saf 100:275–281
Pérez-Iglesias JM, Soloneski S, Nikoloff N, Natale GS, Larramendy ML (2015) Toxic and genotoxic effects of the imazethapyr-based herbicide formulation Pivot H® on montevideo tree frog Hypsiboas pulchellus tadpoles (Anura, Hylidae). Ecotoxicol Environ Saf 119:15–24
Pérez-Iglesias JM, Franco-Belussi L, Natale GS, Oliveira C (2019) Biomarkers at different levels of organisation after atrazine formulation (SIPTRAN 500SC®) exposure in Rhinella schineideri (Anura: Bufonidae) Neotropical tadpoles. Environ Pollut 244:733–746
Pignati AW, Lima FANS, Lara SS, Correa MLM, Barbosa JR, Leão LHC, Pignatti MG (2017) Spatial distribution of pesticide use in Brazil: a strategy for health surveillance. Ciência Saúde Coletiva 22:3281–3293
Pollo FE, Bionda CL, Salinas ZA, Salas NE, Martino AL (2015) Common toad Rhinella arenarum (Hensel, 1867) and its importance in assessing environmental health: test of micronuclei and nuclear abnormalities in erythrocytes. Environ Monitor Assess 187:1–9
Pollo FE, Grenat PR, Otero MA, Salas NE, Martino AL (2016) Assessment in situ of genotoxicity in tadpoles and adults of frog Hypsiboas cordobae (Barrio 1965) inhabiting aquatic ecosystems associated to fluorite mine. Ecotoxicol Environ Saf 133:466–474
Pollo FE, Grenat PR, Salinas ZA, Otero MA, Salas NE, Martino AL (2017) Evaluation in situ of genotoxicity and stress in South American common toad Rhinella arenarumin environments related to fluorite mine. Environ Sci Pollut Res 24:18179–181872
Ramos-Neto MB, Pivello VR (2000) Lightning Fires in a Brazilian Savanna National Park: rethinking management strategies. Environ Manag 26:675–684
Redford KH, Fonseca GAB (1986) The role of Gallery Forests in the Zoogeography of the Cerrado’s non-volant mammalian fauna. Biotropica 18:126–135
Rossa-Feres DC, Nomura F (2006) Morphological characterization and taxonomic key for tadpoles (Amphibia: Anura) from northwestern region of São Paulo state, Brazil. Biota Neotrop 6:1–26
Udroiu I, Sgura A, Vignoli L, Bologna MA, D’Amen M, Salvi D, Ruzza A, Antoccia A, Tanzarella C (2015) Micronucleus test on Triturus carnifex as a tool for environmental biomonitoring. Environ Molec Mutagenesis 56:412–417
Zaya RM, Amini Z, Whitaker AS, Kohler SL, Ide CF (2011) Atrazine exposure affects growth, body condition and liver health in Xenopus laevis tadpoles. Aquat Toxicol 104:243–253
Acknowledgements
The authors are grateful to the Brazilian National Council for Scientific and Technological Development (CNPq), for supporting this study through personal research Grant Number 477044/2013-1, and the São Paulo State Research Foundation (FAPESP) for supporting the analyses (Process No. 2013/02067-5). They also thank the Chico Mendes Institute for Biodiversity Conservation (ICMBio/SISBIO) and the Emas National Park for authorizing fieldwork.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Borges, R.E., Santos, L.R.d., Benvindo-Souza, M. et al. Genotoxic Evaluation in Tadpoles Associated with Agriculture in the Central Cerrado, Brazil. Arch Environ Contam Toxicol 77, 22–28 (2019). https://doi.org/10.1007/s00244-019-00623-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-019-00623-y