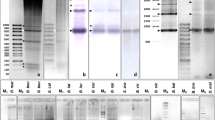
Abstract
Due to a high evolutionary turnover many satellite DNAs are restricted to a group of closely related species. Here we demonstrate that the satellite DNA family PSUB, abundant in the beetle Palorus subdepressus, is distributed in a low number of copies among diverse taxa of Coleoptera (Insecta), some of them separated for an evolutionary period of up to 60 Myr. Comparison of PSUB cloned from the species Tribolium brevicornis with the PSUB family previously characterized in Palorus subdepressus revealed high sequence conservation and absence of fixed species-specific mutations. The most polymorphic sites are those with ancestral mutations shared among clones of both species. Since the ancestral mutations contribute significantly to overall diversity, it could be proposed that a similar mutational profile already existed in an ancestral species. The pattern of variability along the satellite monomer is characterized by the presence of conserved and variable regions. The nonrandom pattern of variability as well as the absence of sequence divergence is also discerned for PRAT satellite DNA, cloned previously from two Palorus species and a distantly related Pimelia elevata. Since PRAT and PSUB are present in parallel in diverse taxa of Coleoptera, we propose that their long evolutionary preservation suggests a possible functional significance. This indication is additionally supported not only by the high evolutionary conservation of the sequences, but also by the presence of significantly conserved and variable regions along the monomers.





Similar content being viewed by others
References
Alvarez-Fuster A, Juan C, Petitpierre E (1991) Genome size in Tribolium flour-beetles: inter- and intraspecific variation. Genet Res 58:1–5
Aravin AA, Lagos-Quintana M, Yalcin A, Zavolan M, Marks D, Snyder B, Gaasterland T, Meyer J, Tuschl T (2003) The small RNA profile during Drosophila melanogaster development. Cell 5:337–350
Cafasso D, Cozzolino S, De Luca P, Chinali G (2003) An unusual satellite DNA from Zamia paucijuga (Cyclades) characterized by two different organizations of the repetitive units in the plant genome. Gene 311:71–79
Charlesworth B, Sniegowski P, Stephan W (1994) The evolutionary dynamics of repetitive DNA in eukaryotes. Nature 371:215–220
De la Herrán R, Fontana F, Lanfredi M, Congiu L, Leis M, Rossi R, Ruiz Rejón C, Ruiz Rejón M, Garrido-Ramos MA (2001) Slow rates of evolution and sequence homogenization in an ancient satellite DNA family of sturgeons. Mol Biol Evol 18:432–436
Durajlija Žinić S, Ugarković Đ, Cornudella L, Plohl M (2000) A novel interspersed type of organization of satellite DNA in Tribolium madens heterochromatin. Chromosome Res 8:201–212
Frazer KA, Tao H, Osoegawa K, de Jong PJ, Chen X, Doherty MF, Cox DR (2004) Noncoding sequences conserved in a limited number of mammals in the SIM2 interval are frequently functional. Genome Res 14:367–372
Garrido-Ramos MA, de la Herrán R, Jamilena M, Lozano R, Ruiz Rejón C, Ruiz Rejón M (1999) Evolution of centromeric satellite DNA and its use in phylogenetic studies of Sparidae family (Pisces, Perciformes). Mol Phylogenet Evol 12:200–204
Grétarsdóttir G, Arnason U (1992) Evolution of the common cetacean highly repetitive DNA component and the systematic position of Orcaella brevirostris. J Mol Evol 34:201–208
Hall SE, Kettler G, Preuss D (2003) Centromere satellites from Arabidopsis populations: maintenance of conserved and variable domains. Genome Res 13:195–205
Henikoff S, Ahmad K, Malik HS (2001) The centromere paradox: stable inheritance with rapidly evolving DNA. Science 293:1098–1102
Hinton HE, (1948) A synopsis of the genus Tribolium MacLey with some remarks on the evolution of its species groups. Bull Entomol Res 39:13–55
Kipling D, Warburton PE (1997) Centromeres, CENP-B and Tigger too. Trends Genet 13:141–145
Kwieton E, (1977) Esquisse phylogenetique du genre Pimelia F. Acta Ent Mus Nat Pragae 39:559–589
Li YX, Kirby ML (2003) Coordinated and conserved expression of alphoid repeat and alphoid repeat-tagged coding sequences. Dev Dynamics 228:72–81
Lopez CC, Edström JE (1998) Interdispersed centromeric element with a CENP-B box-like motif in Chironomus pallidivitatus. Nucleic Acids Res 26:4168–4172
Lorite P, Carrillo JA, Tinaut A, Palomeque T (2002) Comparative study of satellite DNAs in ants of the Messor genus. Gene 297:113–122
Masumoto H, Masukata H, Muro Y, Nozaki N, Okazaki T (1989) A human centromere antigen (CENP-B) interacts with a short specific sequence in alphoid DNA, a human centromere satellite. J Cell Biol 109:1963–1973
Meštrović N, Plohl M, Mravinac B, Ugarković Đ (1998) Evolution of satellite DNAs from the genus Palorus—experimental evidence for the library hypothesis. Mol Biol Evol 15:1062–1068
Meštrović N, Mravinac B, Juan C, Ugarković Đ, Plohl M (2000) Comparative study of satellite sequences and phylogeny of five species from the genus Palorus (insecta, Coleoptera). Genome 43:776–785
Mravinac B, Plohl M, Meštrović N, Ugarković Đ (2002) Sequence of PRAT satellite DNA “frozen” in some coleopteran species. J Mol Evol 54:774–783
Mravinac B, Plohl M, Ugarković Đ (2004) Conserved patterns in the evolution of Tribolium satellite DNAs. Gene 332:169–177
Ohzeki J, Nakano M, Okada T, Masumoto H (2002) CENP-B box is required for de novo centromere chromatin assembly on human alphoid DNA. J Cell Biol 159:765–775
Plohl M, Cornudella L (1996) Characterization of a complex satellite DNA in the mollusc Donax trunculus: analysis of sequence variation and divergence. Gene 169:157–164
Plohl M, Borštnik B, Lucijanić-Justić V, Ugarković Đ (1992) Evidence for random distribution of sequence variants in Tenebrio molitor satellite DNA. Genet Res 60:7–13
Plohl M, Meštrović N, Bruvo B, Ugarković Đ (1998) Similarity of structural features and evolution of satellite DNAs from Palorus subdepressus (Coleoptera) and related species. J Mol Evol 46:234–239
Pons J, Petitpierre E, Juan C (2002) Evolutionary dynamics of satellite DNA family PIM357 in species of the genus Pimelia (Tenebrionidae, Coleoptera). Mol Biol Evol 19:1329–1340
Rozas J, Rozas R (1999) DnaSP version 3: an integrated program for molecular population genetics and molecular evolution analysis. Bioinformatics 15:174–175
Schneider S, Roessli D, Excoffier L (2000) ARLEQUIN: a software for population genetics data analysis, version 2.000. Genetics and Biometry Laboratory, University of Geneva, Switzerland
Steiner JJ, Poklemba CJ, Fjellstrom RG, Elliott LF (1995) A rapid one-tube genomic DNA extraction process for PCR and RAPD analyses. Nucleic Acids Res 23:2569–2570
Strachan T, Webb D, Dover GA (1985) Transition stages of molecular drive in multiple-copy DNA families in Drosophila. EMBO J 4:1701–1708
Swofford DL (1998) PAUP*: Phylogenetic Analysis Using Parsimony (*and other methods), version 4. Sinauer Associates, Sunderland, MA
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The ClustalX windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 24:4876–4882
Ugarković Đ, Plohl M (2002) Variation in satellite DNA profiles—causes and effects. EMBO J 21:5955–5959
Volpe TA, Kidner C, Hall IM, Teng G, Grewal SIS, Martienssen RA (2002) Regulation of heterochromatic silencing and histone H3 lysine-9 methylation by RNAi. Science 297:1833–1837
Acknowledgments
We are grateful to Dr. Joan Pons for the critical reading of and valuable comments on the manuscript. We also thank Dr. Mary Sopta for suggestions and improvement of the English. This work was supported by the Research Fund of the Republic of Croatia, Projects 0098074 and 0098075.
Author information
Authors and Affiliations
Corresponding author
Additional information
[Reviewing Editor: Dr. Jerzy Jurka]
Rights and permissions
About this article
Cite this article
Mravinac, B., Plohl, M. & Ugarković, Ð. Preservation and High Sequence Conservation of Satellite DNAs Suggest Functional Constraints. J Mol Evol 61, 542–550 (2005). https://doi.org/10.1007/s00239-004-0342-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-004-0342-y