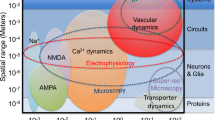
Abstract
Our central nervous system is based on the generation and propagation of electrical signals along the neuronal pathways. These variations of the membrane potential are arranged by the concerted action of ion channels in the neuronal membrane. Therefore, the exact measurement of the electric field in the central nervous system is the focus of intensive investigation. While electrophysiological methods provide exact measurements on the single-cell or single-molecule level with high temporal resolution, they are limited in their spatial resolution ranging from a few single cells to a single molecule. To thoroughly understand how the voltage-dependent ion channels sense the membrane potential and are precisely gated by it, the electric field within the protein has to be investigated. Likewise, the propagation of electrical impulses in a network of neurons involves a large number of cells, which have to be monitored simultaneously. For these endeavors, optical methods have proven to be useful due to their scalability, temporal and spatial resolution. Here, we will summarize the properties of the optical probes that we used to determine the electrical field strength within voltage-sensitive ion channels and discuss the hybrid approach to detect membrane potential changes in genetically specified neurons in terms of design, limitations and future developments.






Similar content being viewed by others
Notes
di-1-ANEPMI: 1-[3-(ß-Maleimidopropionyl) aminopropyl]-4-[ß-[2-(dimethylamino)-6-naphtyl] vinyl] pyridinium bromide; di-1-ANEPIA: l-[3-(Iodoacetyl) aminopropyl]-4-[ß-[2-(dimethylamino)-6-naphtyl] vinyl] pyridinium bromide.
During the onset of a membrane action potential, the current is carried mainly by the opening of the sodium channels (I Na). Therefore, the change in membrane potential is approximately given by dV/dt = −I Na/C, and since INa remains approximately constant, an increase in C reduces dV/dt so that the transmembrane voltage develops more slowly.
In supplementary note.
References
Aggarwal S.K., MacKinnon R. 1996. Contribution of the S4 segment to gating charge in the Shaker K+ channel. Neuron 16:1169–1177
Ahern C.A., Horn R. 2005. Focused electric field across the voltage sensor of potassium channels. Neuron 48:25–29
Asamoah O.K., Wuskell J.P., Loew L.M., Bezanilla F. 2003. A fluorometric approach to local electric field measurements in a voltage-gated ion channel. Neuron 37:85–97
Ataka K., Pieribone V.A. 2002. A genetically targetable fluorescent probe of channel gating with rapid kinetics. Biophys. J. 82:509–516
Baubet V., Le Mouellic H., Campbell A.K., Lucas-Meunier E., Fossier P., Brulet P. 2000 Chimeric green fluorescent protein-aequorin as bioluminescent Ca2+ reporters at the single-cell level. Proc. Natl. Acad. Sci. USA 97:7260–7265
Bedlack R.S., Jr., Wei M.D., Fox S.H., Gross E., Loew L.M. 1994. Distinct electric potentials in soma and neurite membranes. Neuron 13:1187–1193
Bezanilla F. 2000. The voltage sensor in voltage-dependent ion channels. Physiol. Rev. 80:555–592
Blunck R., Starace D.M., Correa A.M., Bezanilla F. (2004) Detecting rearrangements of shaker and NaChBac in real-time with fluorescence spectroscopy in patch-clamped mammalian cells. Biophys. J. 86:3966–3980
Cacciatore T.W., Brodfuehrer P.D., Gonzalez J.E., Jiang T., Adams S.R., Tsien R.Y., Kristan W.B., Jr., et al. 1999. Identification of neural circuits by imaging coherent electrical activity with FRET-based dyes. Neuron 23:449–459
Cha A., Bezanilla F. 1997. Characterizing voltage-dependent conformational changes in the Shaker K+ channel with fluorescence. Neuron 19:1127–1140
Cha A., Bezanilla F. 1998. Structural implications of fluorescence quenching in the Shaker K+ channel. J. Gen. Physiol. 112:391–408
Cha A., Snyder G.E., Selvin P.R., Bezanilla F. 1999. Atomic scale movement of the voltage-sensing region in a potassium channel measured via spectroscopy. Nature 402:809–813
Chanda B., Asamoah O.K., Blunck R., Roux B., Bezanilla F. 2005a. Gating charge displacement in voltage-gated ion channels involves limited transmembrane movement. Nature 436:852–856
Chanda B., Bezanilla F. 2002. Tracking voltage-dependent conformational changes in skeletal muscle sodium channel during activation. J. Gen. Physiol. 120:629–645
Chanda B., Blunck R., Faria L.C., Schweizer F.E., Mody I., Bezanilla F. 2005b. A hybrid approach to measuring electrical activity in genetically specified neurons. Nat. Neurosci. 8:1619–1626
Cohen L.B., Salzberg B.M. 1978. Optical measurement of membrane potential. Rev. Physiol. Biochem. Pharmacol. 83:35–88
Fernandez J.M., Taylor R.E., Bezanilla F. 1983. Induced capacitance in the squid giant axon. Lipophilic ion displacement currents. J. Gen. Physiol. 82:331–346
Gonzalez J.E., Tsien R.Y. 1995. Voltage sensing by fluorescence resonance energy transfer in single cells. Biophys. J. 69:1272–1280
Gonzalez J.E., Tsien R.Y. 1997. Improved indicators of cell membrane potential that use fluorescence resonance energy transfer. Chem. Biol. 4:269–277
Guerrero G., Isacoff E.Y. 2000. Genetically encoded optical sensors of neuronal activity and cellular function. Curr. Opin. Neurobiol. 11:601–607
Guerrero G., Siegel M.S., Roska B., Loots E., Isacoff E.Y. 2002. Tuning FlaSh: Redesign of the dynamic voltage range, and color of the genetically encoded optical sensor of membrane potential. Biophys. J. 83:3607–3618
Helmchen F., Waters J. 2002. Ca2+ imaging in the mammalian brain in vivo. Eur. J. Pharmacol. 447:119–129
Jiang W., Hunter T. 1998. Analysis of cell-cycle profiles in transfected cells using a membrane-targeted GFP. Biotechniques 24:349–50, 352, 354
Knopfel T., Tomita K., Shimazaki R., Sakai R. 2003. Optical recordings of membrane potential using genetically targeted voltage-sensitive fluorescent proteins. Methods 30:42–48
Konnerth A., Obaid A.L., Salzberg B.M. 1987. Optical recording of electrical activity from parallel fibres and other cell types in skate cerebellar slices in vitro. J. Physiol. 393:681–702
Kuner T., Augustine G.J. 2000. A genetically encoded ratiometric indicator for chloride: capturing chloride transients in cultured hippocampal neurons. Neuron 27:447–459
Lakowicz, J.R. 1999. Principles of Fluorescence Spectroscopy. 2nd Ed.
Mannuzzu L.M., Moronne M.M., Isacoff E.Y. 1996. Direct physical measure of conformational rearrangement underlying potassium channel gating. Science 271:213–216
Miesenbock G., De Angelis D.A., Rothman J.E. 1998. Visualizing secretion and synaptic transmission with pH-sensitive green fluorescent proteins. Nature 394:192–195
Miyawaki A., Llopis J., Heim R., McCaffery J.M., Adams J.A., Ikura M., Tsien R.Y. 1997. Fluorescent indicators for Ca2+ based on green fluorescent proteins and calmodulin. Nature 388:882–887
Murata Y., Iwasaki H., Sasaki M., Inaba K., Okamura Y. 2005. Phosphoinositide phosphatase activity coupled to an intrinsic voltage sensor. Nature 435:1239–1243
Nakai J., Ohkura M., Imoto K. 2001. A high signal-to-noise Ca2+ probe composed of a single green fluorescent protein. Nat. Biotechnol. 19:137–141
Ng M., Roorda R.D., Lima S.Q., Zemelman B.V., Morcillo P., Miesenbock G. 2002. Transmission of olfactory information between three populations of neurons in the antennal lobe of the fly. Neuron 36:463–474
Noda M., Shimizu S., Tanabe T., Takai T., Kayano T., Ikeda T., Takahashi H. et al. 1984. Primary structure of Electrophorus electricus sodium channel deduced from cDNA sequences. Nature 312:121–127
Obaid A.L., Loew L.M., Wuskell J.P., Salzberg B.M. 2004. Novel naphthylstyryl-pyridium potentiometric dyes offer advantages for neural network analysis. J. Neurosci. Methods 134:179–190
Ormo M., Cubitt A.B., Kallio K., Gross L.A., Tsien R.Y., Remington S.J. 1996. Crystal structure of the Aequorea victoria green fluorescent protein. Science 273:1392–1395
Posson D.J., Ge P., Miller C., Bezanilla F., Selvin P.R. 2005. Small vertical movement of a K+ channel voltage sensor measured with luminescence energy transfer. Nature 436:848–851
Riven I., Kalmanzon E., Segev L., Reuveny E. 2003. Conformational rearrangements associated with the gating of the G protein-coupled potassium channel revealed by FRET microscopy. Neuron 38:225–235
Rogers, Kelly L., Stinnakre, Jacques, Agulhon, Cendra, Jublot, Delphine, Shorte, Spencer L., Kremer, Eric J., Brulet, Philippe. 2005. Visualization of local Ca2+ dynamics with genetically encoded bioluminescent reporters. Eur. J. Neurosci. 21:597–610
Rohr S., Salzberg B.M. 1994. Multiple site optical recording of transmembrane voltage (MSORTV) in patterned growth heart cell cultures: assessing electrical behavior, with microsecond resolution, on a cellular and subcellular scale. Biophys. J. 67:1301–1315
Romoser V.A., Hinkle P.M., Persechini A. 1997. Detection in living cells of Ca2+ dependent changes in the fluorescence emission of an indicator composed of two green fluorescent protein variants linked by a calmodulin-binding sequence. A new class of fluorescent indicators. J. Biol. Chem. 272:13270–13274
Roorda R.D., Hohl T.M., Toledo-Crow R., Miesenbock G. 2004. Video-rate nonlinear microscopy of neuronal membrane dynamics with genetically encoded probes. J. Neurophysiol. 92:609–621
Rumyantsev S.L., Shur M.S., Bilenko Y., Kosterin P.V., Salzberg B.M. 2004. Low frequency noise and long-term stability of noncoherent light sources. J. Appl. Physics 96:966–969
Sakai R., Repunte-Canonigo V., Raj C.D., Knopfel T. 2001. Design and characterization ft of a DNA-encoded, voltage-sensitive fluorescent protein. Eur. J. Neurosci. 13:2314–2318
Salzberg B.M., Grinvald A., Cohen L.B., Davila H.V., Ross W.N. 1977. Optical recording of neuronal activity in an invertebrate central nervous system: simultaneous monitoring of several neurons. J. Neurophysiol. 40:1281–1291
Salzberg B.M., Kosterin P.V., Muschol M., Obaid A.L., Rumyantsev S.L., Bilenko Y., Shur M.S. 2005. An ultra-stable non-coherent light source for optical measurements in neuroscience and cell physiology. J. Neurosci. Methods 141:165–169
Salzberg B.M., Obaid A.L., Bezanilla F. 1993. Microsecond response of a voltage-sensitive merocyanine dye: fast voltage-clamp measurements on squid giant axon. Jpn. J. Physiol. 43:S37–S41
Schonherr R., Mannuzzu L.M., Isacoff E.Y., Heinemann S.H. 2002. Conformational switch between slow and fast gating modes: allosteric regulation of voltage sensor mobility in the EAG K+ channel. Neuron 35:935–949
Seoh S.A., Sigg D., Papazian D.M., Bezanilla F. 1996. Voltage-sensing residues in the S2 and S4 segments of the Shaker K+ channel. Neuron 16:1159–1167
Siegel M.S. Isacoff E.Y. 1997. A genetically encoded optical probe of membrane voltage. Neuron 19:735–741
Starace D.M. Bezanilla F. 2001. Histidine scanning mutagenesis of basic residues of the S4 segment of the shaker K+ channel. J. Gen. Physiol. 117:469–490
Starace D.M., Bezanilla F. 2004. A proton pore in a potassium channel voltage sensor reveals a focused electric field. Nature 427:548–553
Wang J.W., Wong A.M., Flores J., Vosshall L.B., Axel R. 2003. Two-photon calcium imaging reveals an odor-evoked map of activity in the fly brain. Cell 112:271–282
Zhang J., Davidson R.M., Wei M.D., Loew L.M. 1998. Membrane electric properties by combined patch clamp and fluorescence ratio imaging in single neurons. Biophys. J. 74:48–53
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blunck, R., Chanda, B. & Bezanilla, F. Nano to Micro — Fluorescence Measurements of Electric Fields in Molecules and Genetically Specified Neurons. J Membrane Biol 208, 91–102 (2005). https://doi.org/10.1007/s00232-005-0822-z
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s00232-005-0822-z