Abstract
Purpose
We aimed to develop a meropenem population pharmacokinetic (PK) model in critically ill children and simulate dosing regimens in order to optimize patient exposure.
Methods
Meropenem plasma concentration was quantified by high-performance liquid chromatography. Meropenem PK was investigated using a non-linear mixed-effect modeling approach.
Results
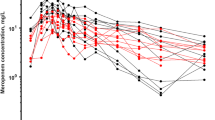
Forty patients with an age of 16.8 (1.4–187.2) months, weight of 9.1 (3.8–59) kg, and estimated glomerular filtration rate (eGFR) of 151 (19–440) mL/min/1.73 m2 were included. Eleven patients received continuous replacement renal therapy (CRRT). Concentration-time courses were best described by a two-compartment model with first-order elimination. Body weight (BW), eGFR, and CRRT were covariates explaining the between-subject variabilities on central/peripheral volume of distribution (V1/V2), inter-compartment clearance (Q), and clearance (CL): V1i = V1pop × (BW/70)1, Qi = Qpop × (BW/70)0.75, V2i = V2pop × (BW/70)1, CLi = (CLpop × (BW/70)0.75) × (eGFR/100)0.378) for patients without CRRT and CLi = (CLpop × (BW/70)0.75) × 0.9 for patients with CRRT, where CLpop, V1pop, Qpop, and V2pop are 6.82 L/h, 40.6 L, 1 L/h, and 9.2 L respectively normalized to a 70-kg subject. Continuous infusion, 60 and 120 mg/kg per day, is the most adequate dosing regimen to attain the target of 50% fT > MIC and 100% fT > MIC for patients infected by bacteria with high minimum inhibitory concentration (MIC) value (> 4 mg/L) without risk of accumulation except in children with severe renal failure.
Conclusion
Continuous infusion allows reaching the fT > MIC targets safely in children with normal or increased renal clearance.




Similar content being viewed by others
Abbreviations
- ARC:
-
Augmented renal clearance
- BIC:
-
Bayesian information criteria
- BSV:
-
Between-subject variability
- BW:
-
Body weight
- CL:
-
Clearance
- CRP:
-
C-reactive protein
- CRRT:
-
Continuous renal replacement therapy
- ECMO:
-
Extracorporeal membrane oxygenation
- EMA:
-
European Medicines Agency
- eGFR:
-
Estimated glomerular filtration rate
- MIC:
-
Minimum inhibitory concentration
- PCT:
-
Procalcitonin
- PICU:
-
Pediatric intensive care unit
- PK:
-
Pharmacokinetics
- PTA:
-
Probability of target attainment
- V :
-
Volume of distribution
References
Baldwin CM, Lyseng-Williamson KA, Keam SJ (2008) Meropenem: a review of its use in the treatment of serious bacterial infections. Drugs 68:803–838
Mattoes HM, Kuti JL, Drusano GL, Nicolau DP (2004) Optimizing antimicrobial pharmacodynamics: dosage strategies for meropenem. Clin Ther 26:1187–1198
Turnidge JD (1998) The pharmacodynamics of beta-lactams. Clin Infect Dis 27:10–22
Blot SI, Pea F, Lipman J (2014) The effect of pathophysiology on pharmacokinetics in the critically ill patient--concepts appraised by the example of antimicrobial agents. Adv Drug Deliv Rev 77:3–11
Béranger A, Oualha M, Urien S, Genuini M, Renolleau S, Aboura R, Hirt D, Heilbronner C, Toubiana J, Tréluyer JM, Benaboud S (2018) Population pharmacokinetic model to optimize cefotaxime dosing regimen in critically ill children. Clin Pharmacokinet 57:867–875
Rhodin MM, Anderson BJ, Peters AM, Coulthard MG, Wilkins B, Cole M, Chatelut E, Grubb A, Veal GJ, Keir MJ, Holford NHG (2009) Human renal function maturation: a quantitative description using weight and postmenstrual age. Pediatr Nephrol 24:67–76
Lonsdale DO, Baker EH, Kipper K, Barker C, Philips B, Rhodes A, Sharland M, Standing JF (2019) Scaling beta-lactam antimicrobial pharmacokinetics from early life to old age. Br J Clin Pharmacol 85:316–346
Avedissian SN, Bradley E, Zhang D, Bradley JS, Nazer LH, Tran TM, Nguyen A, le J (2017) Augmented renal clearance using population-based pharmacokinetic modeling in critically ill pediatric patients. Pediatr Crit Care Med 18:e388–e394
De Cock PA, Standing JF, Barker CI et al (2015) Augmented renal clearance implies a need for increased amoxicillin-clavulanic acid dosing in critically ill children. Antimicrob Agents Chemother 59:7027–7035
Béranger A, Benaboud S, Urien S, Moulin F, Bille E, Lesage F, Zheng Y, Genuini M, Gana I, Renolleau S, Hirt D, Tréluyer JM, Oualha M (2019) Piperacillin population pharmacokinetics and dosing regimen optimization in critically ill children with normal and augmented renal clearance. Clin Pharmacokinet 58:223–233
Leteurtre S, Duhamel A, Salleron J, Grandbastien B, Lacroix J, Leclerc F (2013) PELOD-2: an update of the pediatric logistic organ dysfunction score. Crit Care Med 41:1761–1773
Schwartz GJ, Brion LP, Spitzer A (1987) The use of plasma creatinine concentration for estimating glomerular filtration rate in infants, children, and adolescents. Pediatr Clin N Am 34:571–590
Akcan-Arikan A, Zappitelli M, Loftis LL, Washburn KK, Jefferson LS, Goldstein SL (2007) Modified RIFLE criteria in critically ill children with acute kidney injury. Kidney Int 71:1028–1035
Polsfuss S, Bloemberg GV, Giger J, Meyer V, Hombach M (2012) Comparison of European Committee on Antimicrobial Susceptibility Testing (EUCAST) and CLSI screening parameters for the detection of extended-spectrum β-lactamase production in clinical Enterobacteriaceae isolates. J Antimicrob Chemother 67:159–166
Legrand T, Chhun S, Rey E et al (2008) Simultaneous determination of three carbapenem antibiotics in plasma by HPLC with ultraviolet detection. J Chromatogr B Analyt Technol Biomed Life Sci 875:551–556
Schuster C, Sterz S, Teupser D et al (2018) Multiplex therapeutic drug monitoring by isotope-dilution HPLC-MS/MS of antibiotics in critical illnesses. J Vis Exp 30:138
Anderson BJ, Holford NHG (2008) Mechanism-based concepts of size and maturity in pharmacokinetics. Annu Rev Pharmacol Toxicol 48:303–332
Imani S, Buscher H, Marriott D, Gentili S, Sandaradura I (2017) Too much of a good thing: a retrospective study of β-lactam concentration-toxicity relationships. J Antimicrob Chemother 72:2891–2897
van den Anker JN, Pokorna P, Kinzig-Schippers M, Martinkova J, de Groot R, Drusano GL, Sorgel F (2009) Meropenem pharmacokinetics in the newborn. Antimicrob Agents Chemother 53:3871–3879
Ohata Y, Tomita Y, Nakayama M, Kozuki T, Sunakawa K, Tanigawara Y (2011) Optimal dosage regimen of meropenem for pediatric patients based on pharmacokinetic/pharmacodynamic considerations. Drug Metab Pharmacokinet 26:523–531
Cies JJ, Moore WS, Enache A et al (2017) Population pharmacokinetics and pharmacodynamic target attainment of meropenem in critically ill young children. J Pediatr Pharmacol Ther 22:276–285
Cies JJ, Moore WS, Conley SB et al (2016) Pharmacokinetics of continuous infusion meropenem with concurrent extracorporeal life support and continuous renal replacement therapy: a case report. J Pediatr Pharmacol Ther 21:92–97
Kongthavonsakul K, Lucksiri A, Eakanunkul S, Roongjang S, Issaranggoon na Ayuthaya S, Oberdorfer P (2016) Pharmacokinetics and pharmacodynamics of meropenem in children with severe infection. Int J Antimicrob Agents 48:151–157
Ikawa K, Morikawa N, Ikeda K, Miki M, Kobayashi M (2010) Population pharmacokinetics and pharmacodynamics of meropenem in Japanese pediatric patients. J Infect Chemother 16:139–143
Du X, Li C, Kuti JL, Nightingale CH et al (2006) Population pharmacokinetics and pharmacodynamics of meropenem in pediatric patients. J Clin Pharmacol 46:69–75
Bradley JS, Sauberan JB, Ambrose PG, Bhavnani SM, Rasmussen MR, Capparelli EV (2008) Meropenem pharmacokinetics, pharmacodynamics, and Monte Carlo simulation in the neonate. Pediatr Infect Dis J 27:794–799
Parker EM, Hutchison M, Blumer JL (1995) The pharmacokinetics of meropenem in infants and children: a population analysis. J Antimicrob Chemother 36(Suppl A):63–71
Germovsek E, Lutsar I, Kipper K, Karlsson MO, Planche T, Chazallon C, Meyer L, Trafojer UMT, Metsvaht T, Fournier I, Sharland M, Heath P, Standing JF, NeoMero Consortium, Auriti C, Esposito S, Lorenza P, Ilmoja ML, Drazdiene N, Sarafidis K, Mitsiakos G, van der Flier M, Clarke P, Collinson A, Gupta S, Anthony M, Thomas M, Pattnayak S, Davis J, Rabe H, Pilling E, Bandi S, Sinha A (2018) Plasma and CSF pharmacokinetics of meropenem in neonates and young infants: results from the NeoMero studies. J Antimicrob Chemother 73:1908–1916
Pettit RS, Neu N, Cies JJ, Lapin C, Muhlebach MS, Novak KJ, Nguyen ST, Saiman L, Nicolau DP, Kuti JL (2016) Population pharmacokinetics of meropenem administered as a prolonged infusion in children with cystic fibrosis. J Antimicrob Chemother 71:189–195
Christensson BA, Nilsson-Ehle I, Hutchison M, Haworth SJ, Oqvist B, Norrby SR (1992) Pharmacokinetics of meropenem in subjects with various degrees of renal impairment. Antimicrob Agents Chemother 36:1532–1537
Sime FB, Udy AA, Roberts JA (2015) Augmented renal clearance in critically ill patients: etiology, definition and implications for beta-lactam dose optimization. Curr Opin Pharmacol 24:1–6
Andersen TB (2012) Estimating renal function in children: a new GFR-model based on serum cystatin C and body cell mass. Dan Med J 59:B4486
Craig WA (1997) The pharmacology of meropenem, a new carbapenem antibiotic. Clin Infect Dis 24(Suppl 2):S266–S275
Goldstein SL, Murry DJ, May S et al (2001) Meropenem pharmacokinetics in children and adolescents receiving hemodialysis. Pediatr Nephrol 16:1015–1018
Roberts JA, Abdul-Aziz MH, Lipman J, Mouton JW, Vinks AA, Felton TW, Hope WW, Farkas A, Neely MN, Schentag JJ, Drusano G, Frey OR, Theuretzbacher U, Kuti JL, International Society of Anti-Infective Pharmacology and the Pharmacokinetics and Pharmacodynamics Study Group of the European Society of Clinical Microbiology and Infectious Diseases (2014) Individualised antibiotic dosing for patients who are critically ill: challenges and potential solutions. Lancet Infect Dis 14:498–509
Bradley JS, Dudley MN, Drusano GL (2003) Predicting efficacy of antiinfectives with pharmacodynamics and Monte Carlo simulation. Pediatr Infect Dis J 22:982–992
Taccone FS (2012) Continuous infusion of meropenem in critically ill patients: practical considerations. Crit Care 16:444
Mattioli F, Fucile C, Del Bono V et al (2016) Population pharmacokinetics and probability of target attainment of meropenem in critically ill patients. Eur J Clin Pharmacol 72:839–848
Courter JD, Kuti JL, Girotto JE, Nicolau DP (2009) Optimizing bactericidal exposure for beta-lactams using prolonged and continuous infusions in the pediatric population. Pediatr Blood Cancer 53:379–385
Udy AA, De Waele JJ, Lipman J (2015) Augmented renal clearance and therapeutic monitoring of β-lactams. Int J Antimicrob Agents 45:331–333
Lee B, Kim J, Park JD, Kang HM, Cho YS, Kim KS (2017) Predicting augmented renal clearance using estimated glomerular filtration rate in critically-ill children. Clin Nephrol 88:148–155
Carlier M, Carrette S, Roberts JA, Stove V, Verstraete A, Hoste E, Depuydt P, Decruyenaere J, Lipman J, Wallis SC, de Waele JJ (2013) Meropenem and piperacillin/tazobactam prescribing in critically ill patients: does augmented renal clearance affect pharmacokinetic/pharmacodynamic target attainment when extended infusions are used? Crit Care 17:R84
Roberts JA, Kirkpatrick CMJ, Roberts MS, Robertson TA, Dalley AJ, Lipman J (2009) Meropenem dosing in critically ill patients with sepsis and without renal dysfunction: intermittent bolus versus continuous administration? Monte Carlo dosing simulations and subcutaneous tissue distribution. J Antimicrob Chemother 64:142–150
Ulldemolins M, Soy D, Llaurado-Serra M, Vaquer S, Castro P, Rodríguez AH, Pontes C, Calvo G, Torres A, Martín-Loeches I (2015) Meropenem population pharmacokinetics in critically ill patients with septic shock and continuous renal replacement therapy: influence of residual diuresis on dose requirements. Antimicrob Agents Chemother 59:5520–5528
Acknowledgments
The authors thank the PICU team (physicians and nurses) that selected the children and collected the samples, making this work possible. They also thank the pharmacology laboratory of the Cochin Teaching Hospital, which analyzed the samples.
Authors’ individual contributions
MR collected the data and drafted the manuscript. MO conceived the study and critically revised the manuscript. IG and YZ performed assay analysis. EB identified pathogen agents and related MIC. SU, FF, NB, SB, and DH contributed to acquisition, analysis, and interpretation and also critically revised the manuscript. JMT and SR contributed to conception and design. FL and FM critically revised the manuscript.
Funding
Mélanie Rapp received a grant from the “société française de pédiatrie” for a 1-year research fellowship in the EA7323. The research study received funds from Necker Hospital “financement interne.”
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The Ethics Committee of Necker Hospital approved the study, which was registered at www.clinicaltrials.gov (NCT02539407). Before any inclusion, consent was obtained from children’s parent(s).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
Supplemental material (Assessment of meropenem risk of toxicity) Time course concentration for four groups defined according to renal function (A,B,C,D) with various infusion modalities. (dotted blue: 20 mg/kg every 8 h as a 20-min infusion, dotted red: 40 mg/kg every 8 h as a 20-min infusion, dotted dark green: 20 mg/kg every 8 h as a 3-h infusion, blue: 60 mg/kg per day as a continuous infusion after a bolus of 20 mg/kg, red: 120 mg/kg per day as a continuous infusion after a bolus of 20 mg/kg). Nephrotoxicity and neurotoxicity thresholds are represented by horizontal dotted lines (red and black). (DOCX 13 kb)
ESM 2
(PNG 1001 kb)
Rights and permissions
About this article
Cite this article
Rapp, M., Urien, S., Foissac, F. et al. Population pharmacokinetics of meropenem in critically ill children with different renal functions. Eur J Clin Pharmacol 76, 61–71 (2020). https://doi.org/10.1007/s00228-019-02761-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-019-02761-7