Abstract
Purpose
The antidiabetic drug glibenclamide is metabolized by the enzyme cytochrome P450 2C9 (CYP2C9) encoded by the polymorphic gene CYP2C9. Previous studies involving healthy volunteers have shown a significant influence of variant CYP2C9 genotypes on glibenclamide metabolism. The aim of this study was to investigate the influence of genetic polymorphisms of CYP2C9 on the response to glibenclamide and on glibenclamide plasma levels in type 2 diabetes mellitus patients.
Methods
The study cohort consisted of type 2 diabetes mellitus patients (n = 80) on regular therapy with glibenclamide either alone or with concomitant metformin. Plasma levels of glibenclamide were estimated by reverse phase high pressure liquid chromatography. The variant alleles of CYP2C9, namely CYP2C9 *2 and *3, were identified by PCR–restricted fragment length polymorphism. The plasma levels of glibenclamide and occurrences of hypoglycemic adverse effects with their severity were compared between the genotype groups.
Results
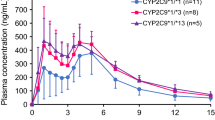
Of the 80 patients (61 males, 19 females), 78 were on concomitant treatment with two drugs, namely, glibenclamide and metformin, and two were on monotherapy with glibenclamide. There was a significant association (p < 0.001) between genotype status of CYP2C9 and the control of diabetes in patients receiving treatment with glibenclamide. There were no statistically significant differences in hypoglycemic adverse effects between the genotype groups.
Conclusion
The type 2 diabetes mellitus patients participating in this study with variant genotypes of CYP2C9 were found to respond better to treatment with glibenclamide than those with the normal genotype. The variant genotype CYP2C9 *1/*3 did not significantly influence the hypoglycemic adverse effects among those patients on long-term glibenclamide treatment.
Similar content being viewed by others
References
Wild S, Roglic G, Green A, Sicree R, King H (2004) Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diab Care 27:1047–1053
Nathan DM, Buse JB, Davidson MB, Ferrannini E, Holman RR, Sherwin R et al (2009) Medical management of hyperglycaemia in type 2 diabetes mellitus: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 52:17–30
Yin O, Tomlinson B, Chow S (2005) CYP2C9, but not CYP2C19, polymorphisms affect the pharmacokinetics and pharmacodynamics of glyburide in Chinese subjects. Clin Pharmacol Ther 78:370–377
Adithan C, Gerard N, Vasu S, Balakrishnan R, Shashindran CH, Krishnamoorthy R (2003) Allele and genotype frequency of CYP2C9 in Tamilnadu population. Eur J Clin Pharmacol 59:707–709
Kirchheiner J, Brockmöller J (2005) Clinical consequences of cytochrome P450 2C9 polymorphisms. Clin Pharmacol Ther 77:1–16
Wang B, Wang J, Huang SQ, Su HH, Zhou SF (2009) Genetic polymorphism of the human cytochrome P450 2C9 gene and its clinical significance. Curr Drug Metab 10:781–834
Niemi M, Cascorbi I, Timm R, Kroemer HK, Neuvonen PJ, Kivisto KT (2002) Glyburide and glimepiride pharmacokinetics in subjects with different CYP2C9 genotypes. Clin Pharmacol Ther 72:326–332
Kirchheiner J, Brockmoller J, Meineke I, Bauer S, Rohde W, Meisel C et al (2002) Impact of CYP2C9 amino acid polymorphisms on glyburide kinetics and on the insulin and glucose response in healthy volunteers. Clin Pharmacol Ther 71:286–296
Asplund K, Wiholm BE, Lithner F (1983) Glibenclamide-associated hypoglycaemia: a report on 57 cases. Diabetologia 24:412–417
Rajendran SD, Philip BK, Gopinath R, Suresh B (2007) RHPLC method for the estimation of glibenclamide in human serum. Indian J Pharm Sci 69:796–799
Sullivan-Klose TH, Ghanayem BI, Bell DA, Zhang ZY, Kaminsky LS, Shenfield GM et al (1996) The role of the CYP2C9-Leu359 allelic variant in the tolbutamide polymorphism. Pharmacogenetics 6:341–349
Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (2003) Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diab Care 26[Suppl 1]:S5–S20
Suzuki K, Yanagawa T, Shibasaki T, Kaniwa N, Hasegawa R, Tohkin M (2006) Effect of CYP2C9 genetic polymorphisms on the efficacy and pharmacokinetics of glimepiride in subjects with type 2 diabetes. Diabetes Res Clin Pract 72:148–154
Kirchheiner J, Roots I, Goldammer M, Rosenkranz B, Brockmoller J (2005) Effect of genetic polymorphisms in cytochrome p450 (CYP) 2C9 and CYP2C8 on the pharmacokinetics of oral antidiabetic drugs: clinical relevance. Clin Pharmacokinet 44:1209–1225
Scheen AJ (1996) Clinical pharmacokinetics of metformin. Clin Pharmacokinet 30:359–371
Melander A, Donnelly R, Rydberg T (1998) Is there a concentration-effect relationship for sulphonylureas? Clin Pharmacokinet 34:181–188
Beck-Nielsen H, Pedersen O, Lindskov HO (1979) Increased insulin sensitivity and cellular insulin binding in obese diabetics following treatment with glibenclamide. Acta Endocrinol (Copenh) 90:451–462
Hjollund E, Richelsen B, Beck-Nielsen H, Pedersen O (1983) The effect of glibenclamide on insulin receptors in normal man: comparative studies of insulin binding to monocytes and erythrocytes. J Clin Endocrinol Metab 57:1257–1262
Lee CR, Pieper JA, Hinderliter AL, Blaisdell JA, Goldstein JA (2002) Evaluation of cytochrome P4502C9 metabolic activity with tolbutamide in CYP2C9 heterozygotes. Clin Pharmacol Ther 72:562–571
Holstein A, Plaschke A, Ptak M, Egberts EH, El-Din J, Brockmoller J et al (2005) Association between CYP2C9 slow metabolizer genotypes and severe hypoglycaemia on medication with sulphonylurea hypoglycaemic agents. Br J Clin Pharmacol 60:103–106
Acknowledgments
We gratefully acknowledge the Indian Council of Medical Research, New Delhi for funding this project (Sanction number: 53/17/2003-BMS. Dated 12.3.2007). We also acknowledge Dr. Jaideep Gogtay and Dr. SD Rajendran for providing pure powders of glibenclamide and glimepiride, respectively. We thank Ms. Indumathi, Ms Kayathri and Ms. Anuradha for their technical assistance in this project.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Surendiran, A., Pradhan, S.C., Agrawal, A. et al. Influence of CYP2C9 gene polymorphisms on response to glibenclamide in type 2 diabetes mellitus patients. Eur J Clin Pharmacol 67, 797–801 (2011). https://doi.org/10.1007/s00228-011-1013-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-011-1013-8