Abstract
Objective
To analyse the contribution of adverse drug reactions (ADR) to hospital readmissions.
Methods
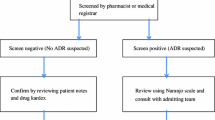
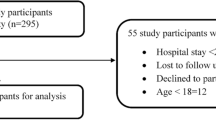
This was a case–control study in which unscheduled admissions of patients who had been admitted to the hospital during the two previous months were assessed during a 21-month period. The patient was considered a case when the main diagnosis of readmission complied with the World Health Organisation’s definition of an ADR. For each case, two controls were selected from those patients that had been admitted for ADR without readmission (n = 177). Information on drugs and other risk factors was obtained from cases by interview and from controls by clinical record review.
Results
There were 26,559 unscheduled admissions of which 81 were readmissions associated with ADR (4.5% of the unscheduled readmissions). There were no statistically significant correlations with sex, age or medical history, with the exception of arterial hypertension. The main drug products causing readmission were acenocoumarol (15, 18.5%), antihypertensive-diuretics (14, 17.3%), anticancer drugs (11, 13.6%) and digoxin (seven, 8.6%). In the multivariate logistic analysis, the variables predicting readmission were acenocoumarol [odds ratio (OR) 12.2, 95% confidence interval (CI) 3.8–38.3, P < 0.0001], a record of diabetes mellitus (OR 2.6, 95% CI 1.3–5.5, P < 0.01), the number of drugs taken at the moment of ADR (OR 1.2, 95% CI 1.1–1.4, P < 0.001) and high blood pressure (OR 0.3, 95% CI 0.2–0.6, P < 0.001) even though the latter was a negative predictor, preventing readmission. Of the 81 readmissions associated with ADR, 28 (34.6%) were preventable.
Conclusion
A medical record of diabetes mellitus, polypharmacy and acenocoumarol treatment were risk factors predicting hospital readmission related to ADR.
Similar content being viewed by others
References
Ashton CM, Del Junco DJ, Souchek J, Wray NP, Mansyur CL (1997) The association between the quality of inpatient care and early readmission: a meta-analysis of the evidence. Med Care 35:1044–1059
Jimenez-Puente A, Garcia-Alegría J, Gomez-Aracena J, Hidalgo-Rojas L, Lorenzo-Nogueiras L, Fernandez-Crehuet-Navajas J (2002) [Analysis of the causes and potential avoidability of readmissions in an acute patients’ hospital]. Med Clin (Barc) 118:500–505
Stowasser DA, Staatz CE, Stowasser M, Coombes JA, Collins DM (2000) Identifying drug-related readmissions. Is there a better way of assessing the contribution of adverse medication events? Aust J Hosp Pharm 30:47–53
Hewitt J (1995) Drug-related unplanned readmissions to hospital. Aust J Hosp Pharm 25:400–403
Bero LA, Lipton HL, Bird JA (1991) Characterization of geriatric drug-related hospital readmissions. Med Care 29:989–1003
Frankl SE, Breeling JL, Goldman L (1991) Preventability of emergent hospital readmission. Am J Med 90:667–674
Pirmohamed M, James S, Meakin S, Green C, Scott AK, Walley TJ et al. (2004) Adverse drug reactions as cause of admission to hospital: prospective analysis of 18 820 patients. Br Med J 329:15–19
Dormann H, Neubert A, Criegee-Rieck M, Egger T, Radespiel-Troger M, Azaz-Livshits T, Levy M, Brune K, Hahn EG (2004) Readmissions and adverse drug reactions in internal medicine: the economic impact. J Intern Med 255:653–663
Lagnaoui R, Moore N, Fach J, Longy-Boursier M, Begaud B (2000) Adverse drug reactions in a department of systemic diseases-oriented internal medicine: prevalence, incidence, direct costs and avoidability. Eur J Clin Pharmacol 55:181–186
Goettler M, Scheneeweiss S, Hasford J (1997) Adverse drug reaction monitoring-cost and benefit considerations Part II: Cost and preventability of adverse drug reactions leading to hospital admission. Pharmacoepidemiol Drug Saf 6[Suppl 3]:79–90
Directive 2001/83/EU (2000) European Parliament and of the Council of 6 November 2000, Strasbourg
Thurmann PA (2001) Methods and systems to detect adverse drug reactions in hospitals. Drug Safety 24:961–968
Schumock GT, Thornton JP (1992) Focusing on the preventability of adverse drug reactions. Hosp Pharm 27:538
Einarson TR (1993) Drug-related hospital admissions. Ann Pharmacother 27:832–840
Lazarou J, Pomeranz BH, Corey PN (1998) Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA 279:1200–1205
Muehlberger N, Schneeweiss S, Hasford J (1997) Adverse drug reaction monitoring-cost and benefit considerations Part I: Frequency of adverse drug reactions causing hospital admissions. Pharmacoepidemiol Drug Saf 6[Suppl 3]:71–77
Roughead EE, Gilbert AL, Primrose JG, Sansom LN (1998) Drug-related hospital admissions: a review of Australian studies published 1988–1996. Med J Aust 168:405–408
Bannwarth B, Queneau P, Carpentier F, Guliana JM, Bouget J, Trombert B, Association Pedagogique Nationale pour l’Enseignement de la Therapeutique (APNET) (2003) Hospital visits caused by adverse drug reactions: incidence and preventability assessed in French primary care/emergency departments. Drug Safety 26:133–134
Pouyanne P, Haramburu F, Imbs JL, Begaud B, for the French Pharmacovigilance Centres (2000) Admissions to hospital caused by adverse drug reactions: cross sectional incidence study. Br Med J 320:1036
Gurwitz JH, Field TS, Harrold LR, Rothschild J, Debellis K, Seger AC et al. (2003) Incidence and preventability of adverse drug events among older persons in the ambulatory setting. JAMA 289:1107–1116
Grymonpre RE, Mitenko PA, Sitar DS, Aoki FY, Montgomery PR (1988) Drug-associated hospital admissions in older medical patients. J Am Geriatr Soc 36:1092–1098
Fortuny J, Silverman D, Malats N, Tardón A, García-Closas R, Serra C et al. (2005) Uso de analgesicos y acido acetilsalicilico en un estudio multicentrico en España. Gac Sanit 19:316–320
Carvajal A, Arias LH, Vega E, Sánchez JA, Rodríguez IM, Ortega PG et al. (2004) Gastroprotection during the administration of non-steroidal anti-inflammatory drugs. A drug-utilization study. Eur J Clin Pharmacol 60:439–444
Otero MJ, Bajo A, Maderuelo JA, Dominguez-Gil A (1999) Evitabilidad de los acontecimientos adversos inducidos por medicamentos detectados en un Servicio de Urgencias. Rev Clin Esp 199:796–805
Forster AJ, Murff HJ, Peterson JF, Gandhi TK, Bates DW (2003) The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med 138:161–167
Howard RL, Avery AJ, Slavenburg S, Royal G, Pipe P, Lucassen P, Pirmohamed M (2006) Which drugs cause preventable admissions to hospital? A systematic review. Br J Clin Pharmacol 63:136–147
McDonnell PJ, Jacobs MR (2002) Hospital admissions resulting from preventable adverse drug reactions. Ann Pharmacother 36:1331–1336
Pearson TF, Pittman DG, Longley JM Grapes ZT, Vigliotti DJ, Mullis SR (1994) Factors associated with preventable adverse drug reactions. Am J Hosp Pharm 51:2268–2272
Hampton T (2006) New oral anticoagulants show promise. JAMA 295:743–744
Acknowledgements
This study was supported by the Galdakao Hospital Research Committee. C. Aguirre benefitted from partial leave from the Basque Foundation for Health Innovation and Research BIOEF (BIO04/LF/22).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ruiz, B., García, M., Aguirre, U. et al. Factors predicting hospital readmissions related to adverse drug reactions. Eur J Clin Pharmacol 64, 715–722 (2008). https://doi.org/10.1007/s00228-008-0473-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-008-0473-y