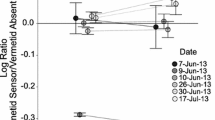
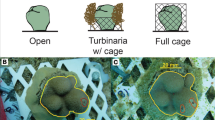
Abstract
Intraspecific variation in coral colony growth forms is common and often attributed to phenotypic plasticity. The ability of other organisms to induce variation in coral colony growth forms has received less attention, but has implications for both taxonomy and the fates of corals and associated species (e.g. fishes and invertebrates). Variation in growth forms and photochemical efficiency of massive Porites spp. in lagoons of Moorea, French Polynesia (17.48°S, 149.85°W), were quantified in 2012. The presence of a vermetid gastropod (Ceraesignum maximum) was correlated with (1) reduced rugosity of coral colonies and (2) reduced photochemical efficiency (Fv/Fm) on terminal “hummocks” (coral tissue in contact with vermetid mucus nets) relative to adjacent “interstitial” locations (tissue not in contact vermetid mucus nets). A manipulative field experiment confirmed that the relative growth rate of coral tissue was greater in interstices than hummocks when vermetids were present and similar (but with a trend for faster growth on hummocks) when vermetids were absent. Collectively, these results indicate that vermetid gastropods interact (presumably via their mucus nets) with coral colony architecture to impair photochemical efficiency, reduce growth rates of specific portions of a coral tissue, and induce a smoothed colony morphology. Given that structural complexity of coral colonies is an important determinant of “habitat quality” for many other species (fishes and invertebrates), these results suggest that the vermetid gastropod, C. maximum (with a widespread distribution and reported increases in density in some portions of its range), may have important indirect effects on many coral-associated organisms.



Similar content being viewed by others
References
Abelson A, Galil BS, Loya Y (1991) Skeletal modifications in stony corals caused by indwelling crabs: hydrodynamical advantages for crab feeding. Symbiosis 10:233–248
Aeby GS, Williams GJ, Franklin EC, Haapkyla J, Harvell CD, Neale S, Page CA, Raymundo L, Vargas-Angel B, Willis BL, Work TM, Davy SK (2011) Growth anomalies on the coral genera Acropora and Porites are strongly associated with host density and human population size across the Indo-Pacific. Plos One 6:e16887
Bergsma GS (2009) Tube-dwelling coral symbionts induce significant morphological change in Montipora. Symbiosis 49:143–150
Bergsma GS (2012) Coral mutualists enhance fish abundance and diversity through a morphology-mediated facilitation cascade. Mar Ecol Prog Ser 451:151–161
Bruno JF, Edmunds PJ (1997) Clonal variation for phenotypic plasticity in the coral Madracis mirabilis. Ecology 78:2177–2190
Chappell J (1980) Coral morphology, diversity and reef growth. Nature 286:249–252
Colgan MW (1985) Growth rate reduction and modification of a coral colony by a vermetid mollusc, Dendropoma maxima. Proceedings of the 5th international coral reef symposium 6:205–210
Connell JH (1976) Competitive interactions and the species diversity of corals. In: Mackie GO (ed) Coelenterate ecology and behavior. Plenum Press, New York, pp 51–68
Davies PS (1989) Short-term growth measurements of corals using an accurate buoyant weight technique. Mar Biol 101:389–395
Dustan P (1975) Growth and form in the reef-building coral Montastrea annularis. Mar Biol 33:101–107
Edmunds PJ (2009) Effect of acclimatization to low temperature and reduced light on the response of reef corals to elevated temperature. Mar Biol 156:1797–1808
Forsman ZH, Barshis DJ, Hunger CL, Toonen RJ (2009) Shape-shifting corals: molecular markers show morphology is evolutionarily plastic in Porites. BMC Evol Biol 9:45
Fukami H, Budd AF, Paulay G, Sole-Cava A, Chen CA, Iwao K, Knowlton N (2004) Conventional taxonomy obscures deep divergence between Pacific and Atlantic corals. Nature 427:832–835
Geange SW, Stier AC (2009) Order of arrival affects competition in two reef fishes. Ecology 90:2868–2878
Gladfelter WB, Gladfelter EH (1978) Fish community structure as a function of habitat structure on West Indian patch reefs. Rev Biol Trop 26:65–84
Golding RE, Bieler R, Rawlings TA, Collins TM (2014) Deconstructing Dendropoma: a systematic revision of a world-wide worm-snail roup, with descriptions of new genera (Caenogastropoda: Vermetidae). Malacologia 57:1–97
Graham NAJ, Wilson SK, Pratchett MS, Polunin NVC, Spalding MD (2009) Coral mortality versus structural collapse as drivers of corallivorous butterflyfish decline. Biodivers Conserv 18:3325–3336
Holbrook SJ, Schmitt RJ (2002) Competition for shelter space causes density-dependent predation mortality in damselfishes. Ecology 83:2855–2868
Kappner I, Al-Moghrabi SM, Richter C (2000) Mucus-net feeding by the vermetid gastropod Dendropoma maxima in coral reefs. Mar Ecol Prog Ser 204:309–313
Kayal M, Vercelloni J, Lison de Loma T, Bosserelle P, Chancerelle Y, Geoffroy S, Stievenart Céline, Michonneau F, Penin L, Planes S, Adjeroud M (2012) Predator Crown-of-Thorns starfish (Acanthaster planci) outbreak, mass mortality of corals, and cascading effects on reef fish and benthic communities. PLoS ONE 7(10):e47363
Lang JC (1973) Interspecific aggression by scleractinian corals. II. Why the race is not always to the swift. Bull Mar Sci 23:260–279
Lesser MP, Weis VM, Patterson MR, Jokiel PL (1994) Effects of morphology and water motion on carbon delivery and productivity in the reef coral, Pocillopora damicornis (Linnaeus): diffusion barriers, inorganic carbon limitation, and biochemical plasticity. J Exp Mar Biol Ecol 178:153–179
Muko S, Kawasaki K, Sakai K, Tasku F, Sigesada N (2000) Morphological plasticity in the coral Porites sillimani and its adaptive significance. Bull Mar Sci 66:225–239
Nakamura Y, Shibuno T, Lecchini D, Kawamura T, Watanabe Y (2009) Spatial variability in habitat associations of pre- and post-settlement stages of coral reef fishes at Ishigaki Island, Japan. Mar Biol 156:2413–2419
Padilla-Gamino JL, Hanson KM, Stat M, Gates RD (2012) Phenotypic plasticity of the coral Porites rus: acclimatization responses to a turbid environment. J Exp Mar Biol Ecol 434:71–80
Pawlik JR, Steindler L, Henkel TP, Beer S, Ilan M (2007) Chemical warfare on coral reefs: sponge metabolites differentially affect coral symbiosis in situ. Limnol Oceanogr 52:907–911
Phillips NE, Shima JS (2010) Reproduction of the vermetid gastropod Dendropoma maximum (Sowerby, 1825) in Moorea, French Polynesia. J Molluscan Stud 76:133–137
Phillips NE, Shima JS, Osenberg CW (2014) Live coral cover may provide resilience to damage from the vermetid gastropod Dendropoma maximum by preventing larval settlement. Coral Reefs. doi:10.1007/s00338-014-1198-2
Rinkevich B, Loya Y (1985) Intraspecific competition in a reef coral: effects on growth and reproduction. Oecologia 66:100–105
River GF, Edmunds PJ (2001) Mechanisms of interaction between macroalgae and scleractinians on a coral reef in Jamaica. J Exp Mar Biol Ecol 261:159–172
Rogers CS (1990) Responses of coral reefs and reef organisms to sedimentation. Mar Ecol Prog Ser 62:185–202
Sebens KP, Johnson TJ (1991) Effect of water movement on prey capture and distribution of reef corals. Hydrobiologia 226:91–101
Shima JS (1999) Variability in relative importance of determinants of reef fish recruitment. Ecol Lett 2:304–310
Shima JS (2001a) Recruitment of a coral reef fish: roles of settlement, habitat, and postsettlement losses. Ecology 82:2190–2199
Shima JS (2001b) Regulation of local populations of a coral reef fish via joint effects of density- and number-dependent mortality. Oecologia 126:58–65
Shima JS (2002) Mechanisms of density- and number-dependent population regulation of a coral-reef fish. Mar Fresh Res 53:175–179
Shima JS, Osenberg CW (2003) Cryptic density dependence: effects of spatio-temporal covariation between density and site quality in reef fish. Ecology 84:46–52
Shima JS, Osenberg CW, St Mary CM (2008) Quantifying site quality in a heterogeneous landscape: recruitment of a reef fish. Ecology 89:86–94
Shima JS, Osenberg CW, Stier AC (2010) The vermetid gastropod Dendropoma maximum reduces coral growth and survival. Biol Lett 6:815–818
Shima JS, Phillips NE, Osenberg CW (2013) Consistent deleterious effects of vermetid gastropods on coral performance. J Exp Mar Biol Ecol 439:1–6
Smith JE, Shaw M, Edwards RA, Obura D, Pantos O, Sala E, Sandin SA, Smriga S, Hatay M, Rohwer FL (2006) Indirect effects of algae on coral: algae-mediated, microbe-induced coral mortality. Ecol Lett 9:835–845
Stier AC, McKeon CS, Osenberg CW, Shima JS (2010) Guard crabs alleviate deleterious effects of vermetid snails on a branching coral. Coral Reefs 29:1019–1022
Stimson J (2011) Ecological characterization of coral growth anomalies on Porites compressa in Hawai’i. Coral Reefs 30:133–142
Todd PA (2008) Morphological plasticity in scleractinian corals. Biol Rev 83:315–337
Veron JEN (2000) Corals of the world. Australian Institute of Marine Science, Townsville
Wainwright SA, Koehl MAR (1976) The nature of flow and the reaction of benthic Cnidaria to it. In: Mackie GO (ed) Coelenterate ecology and behaviour. Plenum Press, New York, pp 5–21
Wielgus J, Glassom D, Ben-Shaprut O, Chadwick-Furman NE (2002) An aberrant growth form of Red Sea corals caused by polychaete infestations. Coral Reefs 21:315–316
Zvuloni A, Armoza-Zvuloni R, Loya Y (2008) Structural deformation of branching corals associated with the vermetid gastropod Dendropoma maxima. Mar Ecol Prog Ser 363:103–108
Acknowledgments
Funding was provided by grants from Victoria University of Wellington and NSF (OCE-1130359). We acknowledge UC Berkeley Gump Research Station for logistic support. This is Contribution Number 212 from UC Berkeley’s Richard B. Gump South Pacific Research Station, Moorea, French Polynesia.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by L Mydlarz.
Reviewed by undisclosed experts.
Rights and permissions
About this article
Cite this article
Shima, J.S., McNaughtan, D. & Strong, A.T. Vermetid gastropods mediate within-colony variation in coral growth to reduce rugosity. Mar Biol 162, 1523–1530 (2015). https://doi.org/10.1007/s00227-015-2688-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-015-2688-7