Abstract
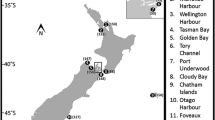
Despite their ubiquity and importance to intertidal ecosystems, information is currently lacking regarding the genetic diversity of trematode parasites within coastal organisms and the distribution of their genetic variation among intertidal habitats. In this study, we quantified the clonal diversity of the coastal marine trematode Maritrema novaezealandensis within Zeacumantus subcarinatus snail hosts from three coastal bays in Otago Harbour, New Zealand, using five microsatellite loci to determine if differences exist in the frequency of occurrence of multi-clone infections. In addition, we examined gene flow among M. novaezealandensis collected from the three bays. The frequency of mixed-clone infections varied fourfold among bays and no genetic differentiation was detected among intertidal bays. Across the coastal bays studied, M. novaezealandensis comprises a single population that is potentially infecting multiple Z. subcarinatus populations with varying life history traits.


Similar content being viewed by others
References
Baudoin M (1975) Host castration as a parasitic strategy. Evolution 29:335–352
Bell AS, de Roode JC, Sim D, Read AF (2006) Within-host competition in genetically diverse malaria infections: parasite virulence and competitive success. Evolution 60:1358–1371
Bremermann HJ, Pickering J (1983) A game-theoretical model of parasite virulence. J Theor Biol 100:411–426
Criscione CD, Poulin R, Blouin MS (2005) Molecular ecology of parasites: elucidating ecological and microevolutionary processes. Mol Ecol 14:2247–2257
Davies CM, Webster JP, Krüger O, Munatsi A, Ndamba J, Woolhouse MEJ (1999) Host–parasite population genetics: a cross-sectional comparison of Bulinus globosus and Schistosoma haematobium. Parasitology 119:295–302
Davies CM, Webster JP, Woolhouse MEJ (2001) Trade-offs in the evolution of virulence in an indirectly transmitted macroparasite. Proc R Soc Lond B 268:251–257
Davies CM, Fairbrother E, Webster JP (2002) Mixed strain schistosome infections of snails and the evolution of parasite virulence. Parasitology 124:31–38
de Roode JC, Pansini R, Cheesman SJ, Helinski MEH, Huijben S, Wargo AR, Bell AS, Chan BHK, Walliker D, Read AF (2005) Virulence and competitive ability in genetically diverse malaria infections. Proc Natl Acad Sci USA 102:7624–7628
Esch GW, Curtis LA, Barger MA (2001) A perspective on the ecology of trematode communities in snails. Parasitology 123:S57–S75
Eppert A, Lewis FA, Grzywacz C, Coura-Filho P, Caldas I, Minchella DJ (2002) Distribution of schistosome infections in molluscan hosts at different levels of parasite prevalence. J Parasitol 88:232–236
Frank SA (1992) A kin selection model for the evolution of virulence. Proc R Soc Lond B 250:195–197
Frank SA (1996) Models of parasite virulence. Q Rev Biol 71:37–78
Fredensborg BL, Poulin R (2006) Parasitism shaping host life-history evolution: adaptive responses in a marine gastropod to infection by trematodes. J Anim Ecol 75:44–53
Fredensborg BL, Mouritsen KN, Poulin R (2006) Relating bird host distribution and spatial heterogeneity in trematode infections in an intertidal snail—from small to large scale. Mar Biol 149:275–283
Gandon S, Michalakis Y (2002) Local adaptation, evolutionary potential and host–parasite coevolution: interactions between migration, mutation, population size and generation time. J Evol Biol 15:451–462
Gandon S, Capowiez Y, Dubois Y, Michalakis Y, Olivieri I (1996) Local adaptation and gene-for-gene coevolution in a metapopulation model. Proc R Soc Lond B 263:1003–1009
Gibson DI, Bray RA (1994) The evolutionary expansion and host–parasite relationships of the Digenea. Int J Parasitol 24:1213–1226
Goudet J (1995) FSTAT (vers. 1.2): a computer program to calculate F-statistics. J Hered 86:485–486
Goudet J, Raymond M, de Meeus T, Rousset F (1996) Testing differentiation in diploid populations. Genetics 144:1933–1940
Gower CM, Webster JP (2005) Intraspecific competition and the evolution of virulence in a parasitic trematode. Evolution 59:544–553
Huyse T, Poulin R, Theron A (2005) Speciation in parasites: a population genetics approach. Trends Parasitol 21:469–475
Jarne P, Theron A (2001) Genetic structure in natural populations of flukes and snails: a practical approach and review. Parasitology 123:S27–S40
Keeney DB, Waters JM, Poulin R (2006) Microsatellite loci for the New Zealand trematode Maritrema novaezealandensis. Mol Ecol Notes 6:1042–1044
Keeney DB, Waters JM, Poulin R (2007) Clonal diversity of the marine trematode Maritrema novaezealandensis within intermediate hosts: the molecular ecology of parasite life cycles. Mol Ecol 16:431–439
Kuris AM (1990) Guild structure of larval trematodes in molluscan hosts: prevalence, dominance and significance of competition In: Esch GW, Bush AO, Aho JM (eds) Parasite communities: patterns and processes. Chapman Hall, London, pp 69–100
Kuris AM, Lafferty KD (1994) Community structure: larval trematodes in snail hosts. Annu Rev Ecol Syst 25:189–217
Lafferty KD, Sammond DT, Kuris AM (1994) Analysis of larval trematode communities. Ecology 75:2275–2285
Lively CM (1999) Migration, virulence, and the geographic mosaic of adaptation by parasites. Am Nat 153:S34–S47
Martorelli SR, Fredensborg BL, Mouritsen KN, Poulin R (2004) Description and proposed life cycle of Maritrema novaezealandensis n. sp. (Microphallidae) parasitic in red-billed gulls, Larus novaehollandiae scopulinus, from Otago Harbor, South Island, New Zealand. J Parasitol 90:272–277
May RM, Nowak MA (1995) Coinfection and the evolution of parasite virulence. Proc R Soc Lond B 261:209–215
Minchella DJ, Sollenberger KM, de Souza CP (1995) Distribution of schistosome genetic diversity within molluscan intermediate hosts. Parasitology 111:217–220
Miura O, Kuris AM, Torchin ME, Hechinger RF, Chiba S (2006) Parasites alter host phenotype and may create a new ecological niche for snail hosts. Proc R Soc Lond B 273:1323–1328
Mouritsen K, Poulin R (2002) Parasitism, community structure and biodiversity in intertidal ecosystems. Parasitology 124:S101–S117
Mouritsen K, Poulin R (2005) Parasitism can influence the intertidal zonation of non-host organisms. Mar Biol 148:1–11
Nei M (1987) Molecular evolutionary genetics. Columbia University Press, New York
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Poulin R (1999) The functional role of parasites in animal communities: many roles at many levels? Int J Parasitol 29:903–914
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Probst S, Kube J (1999) Histopathological effects of larval trematode infections in mudsnails and their impact on host growth: what causes gigantism in Hydrobia ventrosa (Gastropoda: Prosobranchia)? J Exp Mar Biol Ecol 238:49–68
Prugnolle F, Théron A, Pointier JP, Jabbour-Zahab R, Jarne P, Durand P, De Meeus T (2005) Dispersal in a parasitic worm and its two hosts: consequences for local adaptation. Evolution 59:296–303
Rauch G, Kalbe M, Reusch TBH (2005) How a complex life cycle can improve a parasite’s sex life. J Evol Biol 18:1069–1075
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225
Sorensen RE, Minchella DJ (2001) Snail-trematode life history interactions: past trends and future directions. Parasitology 123: S3–S18
Sousa WP (1983) Host life history and the effect of parasitic castration on growth: a field study of Cerithidea californica Haldeman (Gastropoda: Prosobranchia) and its trematode parasites. J Exp Mar Biol Ecol 73:273–296
Sousa WP (1990) Spatial scale and the processes structuring a guild of larval trematode parasites. In: Esch GW, Bush AO, Aho JM (eds) Parasite communities: patterns and processes. Chapman Hall, London, pp 41–67
Sousa WP (1992) Interspecific interactions among larval trematode parasites of freshwater and marine snails. Am Zool 32:583–592
Stenberg P, Lundmark M, Saura A (2003) MLGsim: a program for detecting clones using a simulation approach. Mol Ecol Notes 3:329–331
Thomas F, Cezilly F, de Meeus T, Crivelli A, Renaud F (1997) Parasitism and ecology of wetlands: a review. Estuaries 20:646–654
Thompson JN (2005) The geographic mosaic of coevolution. University of Chicago Press, Chicago
van Baalen M, Sabelis MW (1995) The dynamics of multiple infection and the evolution of virulence. Am Nat 146:881–910
Vilas R, Sanmartin ML, Paniagua E (2004) Genetic variability of natural populations of trematodes of the genus Lecithochirium parasites of eels. Parasitology 129:191–201
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370
Wood CL, Byers JE, Cottingham KL, Altman I, Donahue MJ, Blakeslee AMH (2007) Parasites alter community structure. Proc Natl Acad Sci USA 104:9335–9339
Acknowledgments
We are grateful to Tommy Leung for assistance with trematode species identifications. Funding for this research was provided by the Royal Society of New Zealand’s Marsden Fund. Kim Bryan-Walker was supported by a University of Otago summer research bursary grant. All experiments performed during this study comply with the current laws of New Zealand.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Reusch.
Rights and permissions
About this article
Cite this article
Keeney, D.B., Bryan-Walker, K., King, T.M. et al. Local variation of within-host clonal diversity coupled with genetic homogeneity in a marine trematode. Mar Biol 154, 183–190 (2008). https://doi.org/10.1007/s00227-008-0914-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-008-0914-2