Abstract
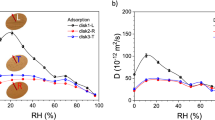
As water moves inside the wood, the gluelines might act as barriers that are potentially leading to local moistening. Even low amounts of water may influence the mechanical performance of glued wood products. Dynamic vapor sorption analysis was performed to assess the sorption processes of six commercial wood adhesives. Vapor sorption isotherms were compared with vapor uptake dynamics. Phenol–resorcinol–formaldehyde showed high moisture uptake of 18 %, while vapor diffusion speed was low. Fish glue showed a water uptake of 45 % at otherwise moderate vapor uptake speed. Melamine–formaldehyde resin gained 22 % water, and polyvinyl acetate absorbed 10 %. The latter was also the fastest vapor absorbing adhesive. Polyurethane only absorbed 3.5 % of moisture at medium uptake speed. Mechanisms of water diffusion seem to be driven by (1) the available free volume in the polymer and (2) the interacting ionic groups of the polymer chain. While the free volume could be linked to the accumulated moisture, the ionic group interaction might determine the measured vapor diffusion dynamics.





Similar content being viewed by others
References
Bell LN, Labuza TP (2000) Moisture sorption: practical aspects of isotherms measurements and use. AACC International, St. Paul
Benz RW, Neville H (1949) Water content of hydrophilic phenol–formaldehyde resins: vapor pressure–temperature relationships. J Polym Sci 4:673–688
Bowditch MR (1996) The durability of adhesive joints in the presence of water. Int J Adhes Adhes 16:73–79
Brunauer S, Emmet PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60:309–319
Brunner M, Lehmann M, Kraft S, Fankhauser U, Richter K, Conzett J (2010) A flexible adhesive layer to strengthen glulam beams. J Adhes Sci Technol 24(8–10):1665–1701
Buck SL (1990) A study of the properties of commercial liquid hide glue and traditional hot hide glue in response to changes in relative humidity and temperature. In: Wooden artifacts group. Specialty sessions, June 2 and 3, 1990. AIC Annual meeting, Richmond, VA
Clauss S (2011) Structure–property relationships of one-component moisture-curing polyurethane adhesives under thermal load. Dissertation at ETH Zürich, No. 20060
Crank J, Park GS (1968) Diffusion in polymers. Academic Press, London
Dhanpal P, Yiu CKY, King NM, Tay FR, Hiraishi N (2009) Effect of temperature on water sorption and solubility of dental adhesive resins. J Dent 37:122–132
Di Vona ML, Sgreccia E, Licoccia S, Khadhraoui M, Denoyel R, Knauth P (2008) Composite proton-conducting hybrid polymers: water sorption isotherms and mechanical properties of blends of sulfonated PEEK and substituted PPSU. Chem Mater 20:4327–4334
Dolmaire N, Espuche E, Mechin F, Pascault JP (2003) Water transport properties of thermoplastic polyurethane films. J Polym Sci B 42:473–492
Dunky M, Niemz P (2002) Holzwerkstoffe und Leime: Technologie und Einflussfaktoren. Springer, Heidelberg
Fabre HS, Fabre S, Cefaly DF, de Oliveira Carrilho MR, Garcia FC, Wang L (2007) Water sorption and solubility of dentin bonding agents light-cured with different light sources. J Dent 35:253–258
Frihart CR (2005) Wood adhesion and adhesives. In: Rowell RM (ed) Handbook of wood chemistry and wood composites. CRC Press, Boca Raton, pp 215–287
Frihart CR, Hunt CG (2010) Adhesives with wood materials: bond formation and performance. Wood handbook: wood as an engineering material, chap 10. Centennial ed. General technical report FPL; GTR-190. US Department of Agriculture, Forest Service, Forest Products Laboratory, Madison, pp 10.1–10.24
Habenicht G (2012) Kleben- erfolgreich und fehlerfrei, 6th edn. Springer Fachmedien, Wiesbaden
Hakala H, Vatanparast R, Vuorimaa E, Lemmetyinen H (2001) Monitoring water uptake of polyurethanes by in situ fluorescence technique. J Appl Polym Sci 82:1593–1599
Herrera-Gómez A, Velázquez-Cruz G, Martín-Polo MO (2001) Analysis of the water bound to a polymer matrix by infrared spectroscopy. J Appl Phys 89:5431–5437
Ito S, Hashimoto M, Wadgaonkar B et al (2005) Effects of resin hydrophilicity on water sorption and changes in modulus of elasticity. Biomaterials 26:6449–6459
Kaiser W (2007) Kunststoffchemie für Ingenieure. Von der Synthese bis zur Anwendung. Carl Hanser, München
Konnerth J, Stockel F, Muller U, Gindl W (2010) Elastic properties of adhesive polymers. III. Adhesive polymer films under dry and wet conditions characterized by means of nanoindentation. J Appl Polym Sci 118:1331–1334
Mannes D, Schmidt J-A, Volkmer T, Niemz P (2012) Untersuchungen zum Einfluss der Klebstoffart auf den kapillaren Wassertransport in Holz parallel zur Faserrichtung. Bauphysik 34:61–65
Marom G (1985) The role of water transport in composite materials, chap 9. In: Comyn J (ed) Polymer permeability. Elsevier Applied Science, Great Britain, pp 341–374
Miyazaki J, Nakano T (2005) Hygroscopicity of resorcinol–formaldehyde resin and aqueous vinyl polymer-isocyanate resin in high humidity. Holzforschung 59:342–346
Mortier E, Gerdolle DA, Jacquot B, Panighi MM (2004) Importance of water sorption and solubility studies for couple bonding agent—resin-based filling materials. Oper Dent 29:669–676
Norland RE (2006) Fish gelatin and fish flue. In: Tracton AA (ed) Coatings technology handbook, 3rd edn. CRC Press, Boca Raton, pp 65-1–65-4
Pizzi A (2003) Phenolic resin adhesives. In: Pizzi A, Mittal KL (eds) Handbook of adhesive technology, chap 29, 2nd edn. Marcel Decker, New York
Rouquerol F, Rouquerol J, Sing K (1999) Adsorption by powders and porous solids: principles, methodology and applications. Academic Press, London
Sangwichien C, Aranovich GL, Donohue MD (2002) Density functional theory predictions of adsorption isotherms with hysteresis loops. Colloids Surf A 206:313–320
Sing KSW (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl Chem 54:2201–2218
Sing KSW (1998) Adsorption methods for the characterization of porous materials. Adv Colloid Interface Sci 76–77:3–11
Sing KSW (2004) Characterization of porous materials: past, present and future. Colloid Surf A 241:3–7
Smith AL, Mulligan RB Sr, Shirazi HM (2004) Determining the effects of vapor sorption in polymers with the quartz crystal microbalance/heat conduction calorimeter. J Polym Sci Part B 42:3893–3906
Soles CL, Yee AF (2000) A discussion of the molecular mechanisms of moisture transport in epoxy resins. J Polym Sci B 38:792–802
Soles CL, Chang FT, Bolan BA, Hristov HA, Gidley DW, Yee AF (1998) Contribution of the nanovoid structure to the moisture absorption properties of epoxy resins. J Polym Sci B 36:3035–3048
Sonderegger W, Hering S, Mannes D et al (2010) Quantitative determination of bound water diffusion in multilayer boards by means of neutron imaging. Eur J Wood Prod 68:341–350
Takizawa A (1967) Analysis of the high-pressure region of certain sorption isotherms. J Phys Chem 71:1611–1616
Thommes M, Köhn R, Fröba M (2002) Sorption and pore condensation behavior of pure fluids in mesoporous MCM-48 silica, MCM-41 silica, SBA-15 silica and controlled-pore glass at temperatures above and below the bulk triple point. Appl Surf Sci 196:239–249
van der Wel GK, Adan OCG (1999) Moisture in organic coatings—a review. Prog Org Coat 37:1–14
Vick CB (1999) Adhesive bonding of wood materials. In: Wood handbook: wood as an engineering material. General technical report FPL; GTR-113. USDA Forest Service, Forest Products Laboratory, Madison, pp 9.1–9.24
Wang H, Yuan Y, Rong M, Zhang M (2009) Microencapsulation of styrene with melamine–formaldehyde resin. Colloid Polym Sci 287:1089–1097
Zeppenfeld G, Grunwald D (2005) Klebstoffe in der Holz- und Möbelindustrie, 2nd edn. DRW-Verlag Weinbrenner GmbH & Co. KG, Leinfelden-Echterdingen
Acknowledgments
The analytical assistance by Monika Funk from the University Göttingen is greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wimmer, R., Kläusler, O. & Niemz, P. Water sorption mechanisms of commercial wood adhesive films. Wood Sci Technol 47, 763–775 (2013). https://doi.org/10.1007/s00226-013-0538-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00226-013-0538-7