Abstract
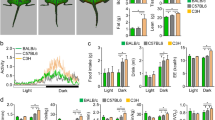
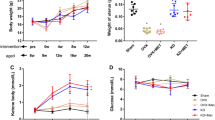
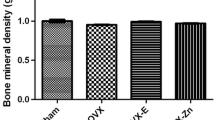
To clarify osteoporotic effects of ketogenic diet (KD) on cancellous and cortical bone compared with ovariectomy (OVX) in mice. Forty female C57BL/6J 8-week-old mice were randomly divided into SD+Sham, SD+OVX, KD+Sham, and KD+OVX groups, and fed for 12 weeks. The distal femur of trabecular bone and the middle femur of cortical bone were evaluated with Micro-CT scanning. The maximum bending force and stiffness of the tibia were calculated using a three-point bending test. Osteoblast and osteoclast expression of femur were identified using tartrate-resistant acid phosphatase (TRAP), collagen type I (CoLI), and osteocalcin (OCN) staining. A 2-factor analysis of variance was used to evaluate effects of KD and OVX on radiological, biomechanical, and histological parameters. KD resulted in not only remarkable cancellous bone decline comparable to OVX, but also unique cortical bone reduction. The maximum bending force and stiffness decreased in the KD+Sham and KD+OVX groups but did not change in the SD+OVX group. The KD+OVX led to significantly higher expression in TRAP and noticeably lower expression in CoLI when compared with other groups. Both KD+Sham and SD+OVX prominently increased expression in TRAP, but decreased expression in CoLI. There was no significant difference in OCN among the four groups. The present results suggest that KD compromises both the cancellous and cortical bone architecture of long bones while OVX only in cancellous bone architecture. A combination of KD and OVX may lead to more bone loss.





Similar content being viewed by others
References
Yudkoff M, Daikhin Y, Melo TM, Nissim I, Sonnewald U, Nissim I (2007) The ketogenic diet and brain metabolism of amino acids: relationship to the anticonvulsant effect. Annu Rev Nutr 27:415–430
Guzmán M, Blázquez C (2004) Ketone body synthesis in the brain: possible neuroprotective effects. Prostaglandins Leukot Essent Fat Acids 70:287–292
Streijger F, Plunet WT, Lee JHT, Liu J, Lam CK, Park S, Hilton BJ, Fransen BL, Matheson KAJ, Assinck P, Kwon BK, Tetzlaff W (2013) Ketogenic diet improves forelimb motor function after spinal cord injury in rodents. PloS One 8:e78765
Winesett SP, Bessone SK, Kossoff EH (2015) The ketogenic diet in pharmacoresistant childhood epilepsy. Expert Rev Neurother 15:621–628
Kossoff EH, Pyzik PL, McGrogan JR, Vining EPG, Freeman JM (2002) Efficacy of the ketogenic diet for infantile spasms 109:780–783
Puchowicz MA, Zechel JL, Valerio J, Emancipator DS, Xu K, Pundik S, LaManna JC, Lust WD (2008) Neuroprotection in diet-induced ketotic rat brain after focal ischemia. J Cereb Blood Flow Metab 28:1907–1916
Tai KK, Nguyen N, Pham L, Truong DD (2008) Ketogenic diet prevents cardiac arrest-induced cerebral ischemic neurodegeneration. J Neural Transm 115:1011–1017
Seyfried TN, Kiebish M, Mukherjee P, Marsh J (2008) Targeting energy metabolism in brain cancer with calorically restricted ketogenic diets. Epilepsia 49:114–116
Hahn TJ, Halstead LR, DeVivo DC (1979) Disordered mineral metabolism produced by ketogenic diet therapy. Calcif Tissue Int 28:17–22
Bergqvist AC, Schall JI, Stallings VA, Zemel BS (2008) Progressive bone mineral content loss in children with intractable epilepsy treated with the ketogenic diet. Am J Clin Nutr 88:1678–1684
van der Louw EJTM, Williams TJ, Henry-Barron BJ, Olieman JF, Duvekot JJ, Vermeulen MJ, Bannink N, Williams M, Neuteboom RF, Kossoff EH, Catsman-Berrevoets CE, Cervenka MC (2017) Ketogenic diet therapy for epilepsy during pregnancy: a case series. Seizure 45:198–201
Garcia-Penas JJ (2016) Autism spectrum disorder and epilepsy: the role of ketogenic diet. Rev Neurol 62(Suppl 1):S73–S78
Woolf EC, Syed N, Scheck AC (2016) Tumor metabolism, the ketogenic diet and β-hydroxybutyrate: novel approaches to adjuvant brain tumor therapy. Front Mol Neurosci 9:122
Vestergaard P (2005) Epilepsy, osteoporosis and fracture risk - a meta-analysis. Acta Neurol Scand 112:277–286
Baldock PA, Need AG, Moore RJ, Durbridge TC, Morris HA (1999) Discordance between bone turnover and bone loss: effects of aging and ovariectomy in the rat. J Bone Miner Res 14:1442–1448
Chiang S, Pan T (2013) Beneficial effects of phytoestrogens and their metabolites produced by intestinal microflora on bone health. Appl Microbiol Biotechnol 97:1489–1500
Tagliaferri C, Salles J, Landrier J, Giraudet C, Patrac V, Lebecque P, Davicco M, Chanet A, Pouyet C, Dhaussy A, Huertas A, Boirie Y, Wittrant Y, Coxam V, Walrand S (2015) Increased body fat mass and tissue lipotoxicity associated with ovariectomy or high-fat diet differentially affects bone and skeletal muscle metabolism in rats. Eur J Nutr 54:1139–1149
Genant HK, Baylink DJ, Gallagher JC (1989) Estrogens in the prevention of osteoporosis in postmenopausal women. Am J Obstet Gynecol 161:1842–1846
Li L, Chen X, Lv S, Dong M, Zhang L, Tu J, Yang J, Zhang L, Song Y, Xu L, Zou J (2014) Influence of exercise on bone remodeling-related hormones and cytokines in ovariectomized rats: a model of postmenopausal osteoporosis. PLoS One 9:e112845
Campbell GM, Buie HR, Boyd SK (2008) Signs of irreversible architectural changes occur early in the development of experimental osteoporosis as assessed by in vivo micro-CT. Osteoporos Int 19:1409–1419
Hsu P, Tsai M, Wang S, Chen Y, Wu J, Hsu J (2016) Cortical bone morphological and trabecular bone microarchitectural changes in the mandible and femoral neck of ovariectomized rats. PLoS One 11:e154367
Guillerminet F, Fabien-Soulé V, Even PC, Tomé D, Benhamou CL, Roux C, Blais A (2012) Hydrolyzed collagen improves bone status and prevents bone loss in ovariectomized C3H/HeN mice. Osteoporos Int 23:1909–1919
Bonucci E, Ballanti P (2014) Osteoporosis—bone remodeling and animal models. Toxicol Pathol 42:957–969
Hadjidakis DJ, Androulakis II (2006) Bone remodeling. Ann N Y Acad Sci 1092:385–396
Vääräniemi J, Halleen JM, Kaarlonen K, Ylipahkala H, Alatalo SL, Andersson G, Kaija H, Vihko P, Väänänen HK (2004) Intracellular machinery for matrix degradation in bone-resorbing osteoclasts. J Bone Miner Res 19:1432–1440
Hollberg K, Nordahl J, Hultenby K, Mengarelli-Widholm S, Andersson G, Reinholt FP (2005) Polarization and secretion of cathepsin K precede tartrate-resistant acid phosphatase secretion to the ruffled border area during the activation of matrix-resorbing clasts. J Bone Miner Metab 23:441–449
Melhus G, Brorson SH, Baekkevold ES, Andersson G, Jemtland R, Olstad OK, Reinholt FP (2010) Gene expression and distribution of key bone turnover markers in the callus of estrogen-deficient, vitamin D-depleted rats. Calcif Tissue Int 87:77–89
Wan DC, Shi YY, Nacamuli RP, Quarto N, Lyons KM, Longaker MT (2006) Osteogenic differentiation of mouse adipose-derived adult stromal cells requires retinoic acid and bone morphogenetic protein receptor type IB signaling. Proc Natl Acad Sci USA 103:12335–12340
Calvo MS, Eyre DR, Gundberg CM (1996) Molecular basis and clinical application of biological markers of bone turnover. Endocr Rev 17:333–368
Clarke B (2008) Normal bone anatomy and physiology. Clin J Am Soc Nephrol 3(Suppl 3):S131–S139
Bielohuby M, Matsuura M, Herbach N, Kienzle E, Slawik M, Hoeflich A, Bidlingmaier M (2010) Short-term exposure to low-carbohydrate, high-fat diets induces low bone mineral density and reduces bone formation in rats. J Bone Miner Res 25:275–284
Yamasaki M, Hasegawa S, Imai M, Takahashi N, Fukui T (2016) High-fat diet-induced obesity stimulates ketone body utilization in osteoclasts of the mouse bone. Biochem Biophys Res Commun 473:654–661
Malvi P, Piprode V, Chaube B, Pote ST, Mittal M, Chattopadhyay N, Wani MR, Bhat MK (2014) High fat diet promotes achievement of peak bone mass in young rats. Biochem Biophys Res Commun 455:133–138
Fehrendt H, Linn T, Hartmann S, Szalay G, Heiss C, Schnettler R, Lips KS (2014) Negative influence of a long-term high-fat diet on murine bone architecture. Int J Endocrinol 2014:1–9
Ionova-Martin SS, Wade JM, Tang S, Shahnazari M, Ager JW, Lane NE, Yao W, Alliston T, Vaisse C, Ritchie RO (2011) Changes in cortical bone response to high-fat diet from adolescence to adulthood in mice. Osteoporos Int 22:2283–2293
Cao JJ, Gregoire BR, Gao H (2009) High-fat diet decreases cancellous bone mass but has no effect on cortical bone mass in the tibia in mice. Bone 44:1097–1104
Lac G, Cavalie H, Ebal E, Michaux O (2008) Effects of a high fat diet on bone of growing rats. Correlations between visceral fat, adiponectin and bone mass density. Lipids Health Dis 7:16
Zernicke RF, Salem GJ, Barnard RJ, Schramm E (1995) Long-term, high-fat-sucrose diet alters rat femoral neck and vertebral morphology, bone mineral content, and mechanical properties. Bone 16:25–31
Ward WE, Kim S, Bruce WR (2003) A western-style diet reduces bone mass and biomechanical bone strength to a greater extent in male compared with female rats during development. Br J Nutr 90:589
Salem GJ, Zernicke RF, Barnard RJ (1992) Diet-related changes in mechanical properties of rat vertebrae. Am J Physiol 262:R318–R321
Smith EE, Ferguson VL, Simske SJ, Gayles EC, Pagliassotti MJ (2000) Effects of high fat or high sucrose diets on rat femora mechanical and compositional properties. Biomed Sci Instrum 36:385–390
Shu L, Beier E, Sheu T, Zhang H, Zuscik MJ, Puzas EJ, Boyce BF, Mooney RA, Xing L (2015) High-fat diet causes bone loss in young mice by promoting osteoclastogenesis through alteration of the bone marrow environment. Calcif Tissue Int 96:313–323
Inzana JA, Kung M, Shu L, Hamada D, Xing LP, Zuscik MJ, Awad HA, Mooney RA (2013) Immature mice are more susceptible to the detrimental effects of high fat diet on cancellous bone in the distal femur. Bone 57:174–183
Zengin A, Kropp B, Chevalier Y, Junnila R, Sustarsic E, Herbach N, Fanelli F, Mezzullo M, Milz S, Bidlingmaier M, Bielohuby M (2016) Low-carbohydrate, high-fat diets have sex-specific effects on bone health in rats. Eur J Nutr 55:2307–2320
Gautam J, Choudhary D, Khedgikar V, Kushwaha P, Singh RS, Singh D, Tiwari S, Trivedi R (2014) Micro-architectural changes in cancellous bone differ in female and male C57BL/6 mice with high-fat diet-induced low bone mineral density. Br J Nutr 111:1811–1821
Acknowledgements
This study is supported by National Natrual Science Foundation of China (No. 81472084). We like to thank Professor Ping Liang at Southern Hospital of Southern Medical University for his help in revising the manuscript, and Dr. Bin Huang, Song Xu, and Wenhao Wang at Southern Medical University for their help in histological analysis.
Author information
Authors and Affiliations
Contributions
Author Contributions
QZ and XW designed the experiments. XW, XW, and ZH conducted the animal experiments. XW, ZH, ZF, and XX wrote the manuscript. XW, JL, GK, and JD completed the data analysis, and QZ revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Xiuhua Wu, Zucheng Huang, Xiaomeng Wang, Zhaozong Fu, Junhao Liu, Zhiping Huang, Ganggang Kong, Xiaolin Xu, Jianyang Ding, and Qingan Zhu, declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
The present study was approved by the Animal Experiments Ethics Committee of Southern Medical University. The animal procedures were conducted in accordance with the Guidelines of Caring for Laboratory Animals by the Ministry of Science and Technology of the People’s Republic of China. Surgery was performed under anesthesia, and all efforts were made to minimize suffering.
Rights and permissions
About this article
Cite this article
Wu, X., Huang, Z., Wang, X. et al. Ketogenic Diet Compromises Both Cancellous and Cortical Bone Mass in Mice. Calcif Tissue Int 101, 412–421 (2017). https://doi.org/10.1007/s00223-017-0292-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-017-0292-1