Abstract
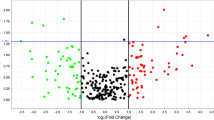
Osteoporosis is a common skeletal disorder characterized by increased risk of bone fracture (BF) due to fragility. BFs, particularly hip fracture, are a major concern in health care because of the associated morbidity and mortality, mainly in the elderly. Lately the involvement of epigenetic mechanisms in the pathophysiology of many diseases has been recognized. In this context, the identification of microRNAs (miRNAs) specific to BF should represent a substantial step forward in diagnostics and therapeutics. The present study aimed to identify specific miRNAs in osteoporotic BF patients compared to those in osteoarthritic controls. In the profiling stage, total RNA was extracted from serum, two pools were prepared, and then retro-transcribed in triplicate. Levels of 179 serum miRNAs were analyzed by real-time PCR, and 42 of them showed significance (P < 0.05), and 12 passed the false discovery rate test for multiple comparisons. Six miRNAs were selected for the replication stage and individually analyzed in sera from 15 BF patients and 12 controls. Results showed that 3 miRNAs (miR-122-5p, miR-125b-5p, and miR-21-5p) were valuable upregulated biomarkers in BF with respect to controls and, significantly, their levels were not affected by hemolysis. For miR-21-5p, the difference detected between groups was independent of age (P = 0.005) and its levels correlated to those of CTx (r = 0.76; P < 0.00001), a marker of bone resorption. In conclusion, several miRNAs may be biomarkers of BF, particularly miR-21-5p. Further studies are needed in order to better characterize the levels of these miRNAs in other bone diseases and to elucidate the mechanism involved in the association of these three miRNAs with osteoporotic BF.





Similar content being viewed by others
References
Kanis JA, McCloskey EV, Johansson H, Cooper C, Rizzoli R, Reginster JY (2013) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 24:23–57
Kanis JA, Oden A, Johnell O, De Laet C, Jonsson B, Oglesby AK (2003) The components of excess mortality after hip fracture. Bone 32:468–473
Ralston SH, Uitterlinden AG (2010) Genetics of osteoporosis. Endocr Rev 31:629–662
Hsu YH, Kiel DP (2012) Clinical review: genome-wide association studies of skeletal phenotypes: what we have learned and where we are headed. J Clin Endocrinol Metab 97:E1958–E1977
Guil S, Esteller M (2009) DNA methylomes, histone codes and miRNAs: tying it all together. Int J Biochem Cell Biol 41:87–95
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297
He L, Hannon GJ (2004) MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet 5:522–531
Brookes E, Shi Y (2014) Diverse epigenetic mechanisms of human disease. Annu Rev Genet 48:237–268
Calin GA, Dumitru CD, Shimizu M, Bichi R, Zupo S, Noch E, Aldler H, Rattan S, Keating M, Rai K, Rassenti L, Kipps T, Negrini M, Bullrich F, Croce CM (2002) Frequent deletions and down-regulation of micro- RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci USA 99:15524–15529
Kosaka N, Iguchi H, Ochiya T (2010) Circulating microRNA in body fluid: a new potential biomarker for cancer diagnosis and prognosis. Cancer Sci 101:2087–2092
Lian JB, Stein GS, van Wijnen AJ, Stein JL, Hassan MQ, Gaur T, Zhang Y (2012) MicroRNA control of bone formation and homeostasis. Nat Rev Endocrinol 8:212–227
Gluer CC (1999) Monitoring skeletal changes by radiological techniques. J Bone Miner Res 14:1952–1962
Garnero P (2014) New developments in biological markers of bone metabolism in osteoporosis. Bone 66:46–55
Kirschner MB, Kao SC, Edelman JJ, Armstrong NJ, Vallely MP, van Zandwijk N, Reid G (2011) Haemolysis during sample preparation alters microRNA content of plasma. PLoS One. 6:e24145
Pfaffl MW, Horgan GW, Dempfle L (2002) Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res 30:e36
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3:0034.1–0034.11
Blondal T, Jensby NS, Baker A, Andreasen D, Mouritzen P, Wrang TM, Dahlsveen IK (2013) Assessing sample and miRNA profile quality in serum and plasma or other biofluids. Methods 59:S1–S6
Krzeszinski JY, Wei W, Huynh H, Jin Z, Wang X, Chang TC, Xie XJ, He L, Mangala LS, Lopez-Berestein G, Sood AK, Mendell JT, Wan Y (2014) miR-34a blocks osteoporosis and bone metastasis by inhibiting osteoclastogenesis and Tgif2. Nature 512:431–435
Seibel MJ (2005) Biochemical markers of bone turnover: part I: biochemistry and variability. Clin Biochem Rev 26:97–122
Cortez MA, Calin GA (2009) MicroRNA identification in plasma and serum: a new tool to diagnose and monitor diseases. Expert Opin Biol Ther 9:703–711
Seeliger C, Karpinski K, Haug AT, Vester H, Schmitt A, Bauer JS, van Griensven M (2014) Five freely circulating miRNAs and bone tissue miRNAs are associated with osteoporotic fractures. J Bone Miner Res 29:1718–1728
Garmilla-Ezquerra P, Sanudo C, Delgado-Calle J, Perez-Nunez MI, Sumillera M, Riancho JA (2015) Analysis of the bone microRNome in osteoporotic fractures. Calcif Tissue Int 96:30–37
Mizuno Y, Tokuzawa Y, Ninomiya Y, Yagi K, Yatsuka-Kanesaki Y, Suda T, Fukuda T, Katagiri T, Kondoh Y, Amemiya T, Tashiro H, Okazaki Y (2009) miR-210 promotes osteoblastic differentiation through inhibition of AcvR1b. FEBS Lett 583:2263–2268
Li H, Wang Z, Fu Q, Zhang J (2014) Plasma miRNA levels correlate with sensitivity to bone mineral density in postmenopausal osteoporosis patients. Biomarkers 19:553–556
Sugatani T, Hruska KA (2013) Down-regulation of miR-21 biogenesis by estrogen action contributes to osteoclastic apoptosis. J Cell Biochem 114:1217–1222
Mizuno Y, Yagi K, Tokuzawa Y, Kanesaki-Yatsuka Y, Suda T, Katagiri T, Fukuda T, Maruyama M, Okuda A, Amemiya T, Kondoh Y, Tashiro H, Okazaki Y (2008) miR-125b inhibits osteoblastic differentiation by down-regulation of cell proliferation. Biochem Biophys Res Commun 368:267–272
Acknowledgments
The authors are indebted to Mrs. R. Aliaga and Dr. V. Ballester for their technical assistance in management of the patients and biochemical assays. Likewise, the authors are indebted to Drs. A. López Castel and JM Fernández-Costa, both from Valentia Biopharma (Valencia, Spain) for their excellent technical assistance regarding miRNA profiling and validation experiments. This work was supported by grant PI12/02582 from the Fondo de Investigación Sanitaria (FIS, Madrid, Spain), including funds from EU’s FEDER Program. Layla Panach is a predoctoral fellow from the Ministerio de Educación, Cultura y Deporte (Programa de Formación del Profesorado Universitario).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
The study was conducted according to the Helsinki Declaration of 2000 and was approved by the Clinical Research Ethics Committee of the Institute of Health Research, INCLIVA. All participants in the study read and signed an informed consent according to the regulations of INCLIVA.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Panach, L., Mifsut, D., Tarín, J.J. et al. Serum Circulating MicroRNAs as Biomarkers of Osteoporotic Fracture. Calcif Tissue Int 97, 495–505 (2015). https://doi.org/10.1007/s00223-015-0036-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-015-0036-z