Abstract
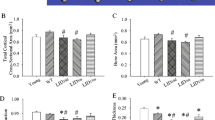
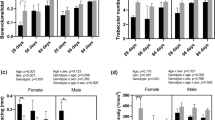
Transcriptional regulation of the postnatal skeleton is incompletely understood. Here, we determined the consequence of loss of early growth response gene 1 (EGR-1) on bone properties. Analyses were performed on both the microscopic and molecular levels utilizing micro-computed tomography (micro-CT) and Fourier transform infrared imaging (FTIRI), respectively. Mice deficient in EGR-1 (Egr-1 −/−) were studied and compared to sex- and age-matched wild-type (wt) control animals. Femoral trabecular bone in male Egr-1 −/− mice demonstrated osteopenic characteristics marked by reductions in both bone volume fraction (BV/TV) and bone mineral density (BMD). Morphological analysis revealed fewer trabeculae in these animals. In contrast, female Egr-1 −/− animals had thinner trabeculae, but BV/TV and BMD were not significantly reduced. Analysis of femoral cortical bone at the mid-diaphysis did not show significant osteopenic characteristics but detected changes in cross-sectional geometry in both male and female Egr-1 −/− animals. Functionally, this resulted in decreased resistance to three-point bending as indicated by a reduction in maximum load, failure load, and stiffness. Assessment of compositional bone properties, including mineral-to-matrix ratio, carbonate-to-phosphate ratio, crystallinity, and cross-linking, in femurs by FTIRI did not show any significant differences or an appreciable trend between Egr-1 −/− and wt mice of either sex. Unexpectedly, rib bone from Egr-1 −/− animals displayed distinct osteopenic traits that were particularly pronounced in female mice. This study provides genetic evidence that both sex and skeletal site are critical determinants of EGR-1 activity in vivo and that its site-specific action may contribute to the mechanical properties of bone.





Similar content being viewed by others
References
Wagner EF (2010) Bone development and inflammatory disease is regulated by AP-1 (Fos/Jun). Ann Rheum Dis 69(Suppl 1):i86–i88
Wagner EF (2002) Functions of AP1 (Fos/Jun) in bone development. Ann Rheum Dis 61(Suppl 2):ii40–ii42
Karsenty G (2008) Transcriptional control of skeletogenesis. Annu Rev Genomics Hum Genet 9:183–196
Kawamata A, Izu Y, Yokoyama H, Amagasa T, Wagner EF, Nakashima K, Ezura Y, Hayata T, Noda M (2008) JunD suppresses bone formation and contributes to low bone mass induced by estrogen depletion. J Cell Biochem 103:1037–1045
Wang ZQ, Ovitt C, Grigoriadis AE, Mohle-Steinlein U, Ruther U, Wagner EF (1992) Bone and haematopoietic defects in mice lacking c-fos. Nature 360:741–745
Milbrandt J (1987) A nerve growth factor-induced gene encodes a possible transcriptional regulatory factor. Science 238:797–799
Lau LF, Nathans D (1987) Expression of a set of growth-related immediate early genes in BALB/c 3T3 cells: coordinate regulation with c-fos or c-myc. Proc Natl Acad Sci USA 84:1182–1186
Lemaire P, Revelant O, Bravo R, Charnay P (1988) Two mouse genes encoding potential transcription factors with identical DNA-binding domains are activated by growth factors in cultured cells. Proc Natl Acad Sci USA 85:4691–4695
Lim RW, Varnum BC, Herschman HR (1987) Cloning of tetradecanoyl phorbol ester-induced “primary response” sequences and their expression in density-arrested Swiss 3T3 cells and a TPA non-proliferative variant. Oncogene 1:263–270
Christy B, Nathans D (1989) DNA binding site of the growth factor-inducible protein Zif268. Proc Natl Acad Sci USA 86:8737–8741
Gashler A, Sukhatme VP (1995) Early growth response protein 1 (Egr-1): prototype of a zinc-finger family of transcription factors. Prog Nucleic Acid Res Mol Biol 50:191–224
Swirnoff AH, Milbrandt J (1995) DNA-binding specificity of NGFI-A and related zinc finger transcription factors. Mol Cell Biol 15:2275–2287
Gururajan M, Simmons A, Dasu T, Spear BT, Calulot C, Robertson DA, Wiest DL, Monroe JG, Bondada S (2008) Early growth response genes regulate B cell development, proliferation, and immune response. J Immunol 181:4590–4602
Hofer G, Grimmer C, Sukhatme VP, Sterzel RB, Rupprecht HD (1996) Transcription factor Egr-1 regulates glomerular mesangial cell proliferation. J Biol Chem 271:28306–28310
Ham J, Eilers A, Whitfield J, Neame SJ, Shah B (2000) c-Jun and the transcriptional control of neuronal apoptosis. Biochem Pharmacol 60:1015–1021
Scharnhorst V, Menke AL, Attema J, Haneveld JK, Riteco N, van Steenbrugge GJ, van der Eb AJ, Jochemsen AG (2000) EGR-1 enhances tumor growth and modulates the effect of the Wilms’ tumor 1 gene products on tumorigenicity. Oncogene 19:791–800
Biesiada E, Razandi M, Levin ER (1996) Egr-1 activates basic fibroblast growth factor transcription. Mechanistic implications for astrocyte proliferation. J Biol Chem 271:18576–18581
Perez-Castillo A, Pipaon C, Garcia I, Alemany S (1993) NGFI-A gene expression is necessary for T lymphocyte proliferation. J Biol Chem 268:19445–19450
Kaufmann K, Thiel G (2001) Epidermal growth factor and platelet-derived growth factor induce expression of Egr-1, a zinc finger transcription factor, in human malignant glioma cells. J Neurol Sci 189:83–91
Kaufmann K, Thiel G (2002) Epidermal growth factor and thrombin induced proliferation of immortalized human keratinocytes is coupled to the synthesis of Egr-1, a zinc finger transcriptional regulator. J Cell Biochem 85:381–391
Lee SL, Tourtellotte LC, Wesselschmidt RL, Milbrandt J (1995) Growth and differentiation proceeds normally in cells deficient in the immediate early gene NGFI-A. J Biol Chem 270:9971–9977
Cibelli G, Policastro V, Rossler OG, Thiel G (2002) Nitric oxide-induced programmed cell death in human neuroblastoma cells is accompanied by the synthesis of Egr-1, a zinc finger transcription factor. J Neurosci Res 67:450–460
Messmer UK, Brune B (1996) Nitric oxide-induced apoptosis: p53-dependent and p53-independent signalling pathways. Biochem J 319(Pt 1):299–305
Le-Niculescu H, Bonfoco E, Kasuya Y, Claret FX, Green DR, Karin M (1999) Withdrawal of survival factors results in activation of the JNK pathway in neuronal cells leading to Fas ligand induction and cell death. Mol Cell Biol 19:751–763
Behrens A, Sibilia M, Wagner EF (1999) Amino-terminal phosphorylation of c-Jun regulates stress-induced apoptosis and cellular proliferation. Nat Genet 21:326–329
Srivastava S, Weitzmann MN, Kimble RB, Rizzo M, Zahner M, Milbrandt J, Ross FP, Pacifici R (1998) Estrogen blocks M-CSF gene expression and osteoclast formation by regulating phosphorylation of Egr-1 and its interaction with Sp-1. J Clin Invest 102:1850–1859
Hoenderop JG, van Leeuwen JP, van der Eerden BC, Kersten FF, van der Kemp AW, Merillat AM, Waarsing JH, Rossier BC, Vallon V, Hummler E, Bindels RJ (2003) Renal Ca2+ wasting, hyperabsorption, and reduced bone thickness in mice lacking TRPV5. J Clin Invest 112:1906–1914
Gourion-Arsiquaud S, West PA, Boskey AL (2008) Fourier transform-infrared microspectroscopy and microscopic imaging. Methods Mol Biol 455:293–303
Boskey A, Pleshko Camacho N (2007) FT-IR imaging of native and tissue-engineered bone and cartilage. Biomaterials 28:2465–2478
Boskey A, Mendelsohn R (2005) Infrared analysis of bone in health and disease. J Biomed Opt 10:031102
Gourion-Arsiquaud S, Allen MR, Burr DB, Vashishth D, Tang SY, Boskey AL (2010) Bisphosphonate treatment modifies canine bone mineral and matrix properties and their heterogeneity. Bone 46:666–672
Cenci S, Weitzmann MN, Gentile MA, Aisa MC, Pacifici R (2000) M-CSF neutralization and egr-1 deficiency prevent ovariectomy-induced bone loss. J Clin Invest 105:1279–1287
Gardiner EM, Baldock PA, Thomas GP, Sims NA, Henderson NK, Hollis B, White CP, Sunn KL, Morrison NA, Walsh WR, Eisman JA (2000) Increased formation and decreased resorption of bone in mice with elevated vitamin D receptor in mature cells of the osteoblastic lineage. FASEB J 14:1908–1916
Boskey AL, Spevak L, Paschalis E, Doty SB, McKee MD (2002) Osteopontin deficiency increases mineral content and mineral crystallinity in mouse bone. Calcif Tissue Int 71:145–154
Anderson HC, Sipe JB, Hessle L, Dhanyamraju R, Atti E, Camacho NP, Millan JL (2004) Impaired calcification around matrix vesicles of growth plate and bone in alkaline phosphatase-deficient mice. Am J Pathol 164:841–847
Camacho NP, Rimnac CM, Meyer RA Jr, Doty S, Boskey AL (1995) Effect of abnormal mineralization on the mechanical behavior of X-linked hypophosphatemic mice femora. Bone 17:271–278
Boskey AL, DiCarlo E, Paschalis E, West P, Mendelsohn R (2005) Comparison of mineral quality and quantity in iliac crest biopsies from high- and low-turnover osteoporosis: an FT-IR microspectroscopic investigation. Osteoporos Int 16:2031–2038
Misof BM, Roschger P, Tesch W, Baldock PA, Valenta A, Messmer P, Eisman JA, Boskey AL, Gardiner EM, Fratzl P, Klaushofer K (2003) Targeted overexpression of vitamin D receptor in osteoblasts increases calcium concentration without affecting structural properties of bone mineral crystals. Calcif Tissue Int 73:251–257
Lian JB, Stein GS (1992) Transcriptional control of vitamin D-regulated proteins. J Cell Biochem 49:37–45
Khachigian LM, Collins T (1998) Early growth response factor 1: a pleiotropic mediator of inducible gene expression. J Mol Med 76:613–616
Mashiba T, Hirano T, Turner CH, Forwood MR, Johnston CC, Burr DB (2000) Suppressed bone turnover by bisphosphonates increases microdamage accumulation and reduces some biomechanical properties in dog rib. J Bone Miner Res 15:613–620
Mashiba T, Mori S, Burr DB, Komatsubara S, Cao Y, Manabe T, Norimatsu H (2005) The effects of suppressed bone remodeling by bisphosphonates on microdamage accumulation and degree of mineralization in the cortical bone of dog rib. J Bone Miner Metab 23(Suppl):36–42
Hashimoto J, Yoshikawa H, Takaoka K, Shimizu N, Masuhara K, Tsuda T, Miyamoto S, Ono K (1989) Inhibitory effects of tumor necrosis factor alpha on fracture healing in rats. Bone 10:453–457
Reumann MK, Nair T, Strachna O, Boskey AL, Mayer-Kuckuk P (2010) Production of VEGF receptor 1 and 2 mRNA and protein during endochondral bone repair is differential and healing phase specific. J Appl Physiol 109:1930–1938
Everts V, de Vries TJ, Helfrich MH (2009) Osteoclast heterogeneity: lessons from osteopetrosis and inflammatory conditions. Biochim Biophys Acta 1792:757–765
Cecchini MG, Hofstetter W, Halasy J, Wetterwald A, Felix R (1997) Role of CSF-1 in bone and bone marrow development. Mol Reprod Dev 46:75–83
Wiktor-Jedrzejczak W, Urbanowska E, Aukerman SL, Pollard JW, Stanley ER, Ralph P, Ansari AA, Sell KW, Szperl M (1991) Correction by CSF-1 of defects in the osteopetrotic op/op mouse suggests local, developmental, and humoral requirements for this growth factor. Exp Hematol 19:1049–1054
Rawlinson SC, McKay IJ, Ghuman M, Wellmann C, Ryan P, Prajaneh S, Zaman G, Hughes FJ, Kingsmill VJ (2009) Adult rat bones maintain distinct regionalized expression of markers associated with their development. PLoS One 4:e8358
Varanasi SS, Olstad OK, Swan DC, Sanderson P, Gautvik VT, Reppe S, Francis RM, Gautvik KM, Datta HK (2010) Skeletal site-related variation in human trabecular bone transcriptome and signaling. PLoS One 5:e10692
Acknowledgements
We thank Kirsten Stoner for skilled execution and interpretation of mechanical bone testing, Ashley Acosta for expert data collection and analysis, and Dr. Marjolein van der Meulen for critical comments on the manuscript. This work was supported by NIH grants R01AR055294 (P. M.-K.) and P30AR046121 (A. L. B.) as well as the postdoctoral fellowship F32AR056148 (E. D.).
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have stated that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Reumann, M.K., Strachna, O., Lukashova, L. et al. Early Growth Response Gene 1 Regulates Bone Properties in Mice. Calcif Tissue Int 89, 1–9 (2011). https://doi.org/10.1007/s00223-011-9486-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-011-9486-0