Abstract
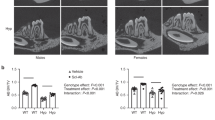
Hypophosphatemia is an X-linked dominant disorder resulting from a mutation in the PHEX gene. While osteoblast-specific expression of the PHEX transgene has been reported to decrease the phosphate wasting associated with the disease in male hypophosphatemic (HYP) mice, there are reports that the mineralization defect is only partially corrected in young animals. To test the hypothesis that osteoblast-specific expression of the PHEX gene for a longer time would correct the mineralization defect, this study examined the bones of 9-month-old male and female HYP mice and their wild-type controls with or without expression of the transgene under a collagen type I promoter. Serum phosphate levels, alkaline phosphatase activity, and FGF23 levels were also measured. Mineral analyses based on wide-angle X-ray diffraction, Fourier transform-infrared (FT-IR) spectroscopy, and FT-IR imaging confirmed the decreased mineral content and increased mineral crystal size in male HYP humerii compared to wild-type males and females with or without the transgene and in female HYP mice with or without the transgene. There was a significant increase in mineral content and a decrease in crystallinity in the HYP males’ bones with the transgene, compared to those without. Of interest, expression of the transgene in wild-type animals significantly increased the mineral content in both males and females without having a detectable effect on crystallinity or carbonate content. In contrast to the bones, based on micro-computed tomography and FT-IR imaging, at 9 months there were no significant differences between the HYP and the WT teeth, precluding analysis of the effect of the transgene.







Similar content being viewed by others
References
Baum M, Syal A, Quigley R, Seikaly M (2006) Role of prostaglandins in the pathogenesis of X-linked hypophosphatemia. Pediatr Nephrol 21:1067–1074
Holm IA, Huang X, Kunkel LM (1997) Mutational analysis of the PEX gene in patients with X-linked hypophosphatemic rickets. Am J Hum Genet 60:790–797
Beck L, Soumounou Y, Martel J, Krishnamurthy G, Gauthier C, Goodyer CG, Tenenhouse HS (1997) Pex/PEX Tissue distribution and evidence for a deletion in the 3′ region of the Pex gene in X-linked hypophosphatemic mice. J Clin Invest 99:1200–1209
Glorieux FH, Holick MF, Scriver CR, DeLuca HF (1973) X-linked hypophosphataemic rickets: inadequate therapeutic response to 1, 25-dihydroxycholecalciferol. Lancet 2:287–289
Eicher EM, Southard JL, Scriver C, Glorieux FH (1976) Hypophosphatemia: mouse model for human familial hypophosphatemic (vitamin D-resistant) rickets. Proc Natl Acad Sci USA 73:4667–4671
Rowe PS, Oudet CL, Francis F, Sinding C, Pannetier S, Econs MJ, Strom TM, Meitinger T, Garabedian M, David A, Macher MA, Questiaux E, Popowska E, Pronicka E, Read AP, Mokrzycki A, Glorieux FH, Drezner MK, Hanauer A, Lehrach H, Goulding JN, O’Riordan JL (1997) Distribution of mutations in the PEX gene in families with X-linked hypophosphataemic rickets (HYP). Hum Mol Genet 6:539–549
Du L, Desbarats M, Viel J, Glorieux FH, Cawthorn C, Ecarot B (1996) cDNA cloning of the murine Pex gene implicated in X-linked hypophosphatemia and evidence for expression in bone. Genomics 36:22–28
Guo R, Quarles LD (1997) Cloning and sequencing of human PEX from a bone cDNA library: evidence for its developmental stage-specific regulation in osteoblasts. J Bone Miner Res 12:1009–1017
Boskey AL, Gilder H, Neufeld E, Ecarot B, Glorieux FH (1991) Phospholipid changes in the bones of the hypophosphatemic mouse. Bone 12:345–351
Abe K, Ooshima T, Masatomi Y, Sobue S, Moriwaki Y (1989) Microscopic and crystallographic examinations of the teeth of the X-linked hypophosphatemic mouse. J Dent Res 68:1519–1524
Abe K, Ooshima T, Sobue S, Moriwaki Y (1989) The crystallinity of human deciduous teeth in hypophosphataemic vitamin D-resistant rickets. Arch Oral Biol 34:365–372
Erben RG, Mayer D, Weber K, Jonsson K, Juppner H, Lanske B (2005) Overexpression of human PHEX under the human beta-actin promoter does not fully rescue the Hyp mouse phenotype. J Bone Miner Res 20:1149–1160
Bai X, Miao D, Panda D, Grady S, McKee MD, Goltzman D, Karaplis AC (2002) Partial rescue of the Hyp phenotype by osteoblast-targeted PHEX (phosphate-regulating gene with homologies to endopeptidases on the X chromosome) expression. Mol Endocrinol 16:2913–2925
Liu S, Guo R, Tu Q, Quarles LD (2002) Overexpression of Phex in osteoblasts fails to rescue the Hyp mouse phenotype. J Biol Chem 277:3686–3697
Brault BA, Meyer MH, Meyer RA Jr, Iorio RJ (1987) Mineral uptake by the femora of older female X-linked hypophosphatemic (HYP) mice but not older male HYP mice. Clin Orthop Relat Res 222:289–299
Cao JJ, Wronski TJ, Iwaniec U, Phleger L, Kurimoto P, Boudignon B, Halloran BP (2005) Aging increases stromal/osteoblastic cell-induced osteoclastogenesis and alters the osteoclast precursor pool in the mouse. J Bone Miner Res 20:1659–1668
Boskey AL, Young MF, Kilts T, Verdelis K (2005) Variation in mineral properties in normal and mutant bones and teeth. Cells Tissues Organs 181:144–153
Pavlin D, Lichtler AC, Bedalov A, Kream BE, Harrison JR, Thomas HF, Gronowicz GA, Clark SH, Woody CO, Rowe DW (1992) Differential utilization of regulatory domains within the alpha 1(I) collagen promoter in osseous and fibroblastic cells. J Cell Biol 116:227–236
Wysolmerski JJ, Broadus AE, Zhou J, Fuchs E, Milstone LM, Philbrick WM (1994) Overexpression of parathyroid hormone-related protein in the skin of transgenic mice interferes with hair follicle development. Proc Natl Acad Sci USA 91:1133–1137
Kacena MA, Troiano NW, Wilson KM, Coady CE, Horowitz MC (2004) Evaluation of two different methylmethacrylate processing, infiltration, and embedding techniques on the histological, histochemical, and immunohistochemical analysis of murine bone specimens. J Histotechnol 27:119–130
Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR (1987) Bone histomorphometry: standardization of nomenclature, symbols, and units. J Bone Miner Res 2:595–610
Boskey AL, Mendelsohn R (2005) Infrared spectroscopic characterization of mineralized tissues. Vib Spectrosc 38:107–114
Yu X, White KE (2005) FGF23 and disorders of phosphate homeostasis. Cytokine Growth Factor Rev 16:221–232
Camacho NP, Rimnac CM, Meyer RA Jr, Doty S, Boskey AL (1995) Effect of abnormal mineralization on the mechanical behavior of X-linked hypophosphatemic mice femora. Bone 17:271–278
Soener S, RA, Meyer MH, Meyer RA Jr (1988) Ovariectomy abolishes the normalization of femoral mineral content in 40-week-old female X-linked hypophosphatemic mice. Miner Electrolyte Metab 14:321–331
Onishi T, Umemura S, Shintani S, Ooshima T (2008) Phex mutation causes overexpression of FGF23 in teeth. Arch Oral Biol. 53:99–104
Rowe PS, Garrett IR, Schwarz PM, Carnes DL, Lafer EM, Mundy GR, Gutierrez GE (2005) Surface plasmon resonance (SPR) confirms that MEPE binds to PHEX via the MEPE-ASARM motif: a model for impaired mineralization in X-linked rickets (HYP). Bone 36:33–46
Rowe PS, Kumagai Y, Gutierrez G, Garrett IR, Blacher R, Rosen D, Cundy J, Navvab S, Chen D, Drezner MK, Quarles LD, Mundy GR (2004) MEPE has the properties of an osteoblastic phosphatonin and minhibin. Bone 34:303–319
Martin A, David V, Laurence JS, Schwarz PM, Lafer EM, Hedge AM, Rowe PS (2008) Degradation of MEPE, DMP1 and release of SIBLING ASARM-peptides (minhibins): ASARM-peptide(s) are directly responsible for defective mineralization in HYP. Endocrinology 149:1757–1772
Qin C, D’Souza R, Feng JQ (2007) Dentin matrix protein 1 (DMP1): new and important roles for biomineralization and phosphate homeostasis. J Dent Res 86:1134–1141
Addison WN, Nakano Y, Loisel T, Crine P, McKee MD (2008) MEPE-ASARM peptides control extracellular matrix mineralization by binding to hydroxyapatite: an inhibition regulated by PHEX cleavage of ASARM. J Bone Miner Res 23:1638–1649
Boskey AL (2007) Mineralization of bones and teeth. Elements 3:387–393
Liu S, Tang W, Zhou J, Vierthaler L, Quarles LD (2007) Distinct roles for intrinsic osteocyte abnormalities and systemic factors in regulation of FGF23 and bone mineralization in Hyp mice. Am J Physiol Endocrinol Metab 293:E1636–E1644
Acknowledgments
This work was supported by NIH Grant DE04141 (A.L.B.), Hospital for Special Surgery Musculoskeletal Repair and Regeneration Core Center Grant NIH P30-AR046121 (A.L.B.), and the Traveler’s Summer Fellowship Program (A.F.), NIH Center for Research Translation Award P50-AR054086 (T.O.C.), and the Yale Core Center for Musculoskeletal Disorders (NIH P30-AR46032).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boskey, A., Frank, A., Fujimoto, Y. et al. The PHEX Transgene Corrects Mineralization Defects in 9-Month-Old Hypophosphatemic Mice. Calcif Tissue Int 84, 126–137 (2009). https://doi.org/10.1007/s00223-008-9201-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-008-9201-y