Abstract
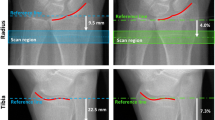
This study evaluates in vivo methods for calculating cortical thickness (Ct.Th) with respect to sensitivity to tissue-level changes in mineralization and the ability to predict whole-bone mechanical properties. Distal radial and tibial images obtained from normal volunteers using high-resolution peripheral quantitative computed tomography (HR-pQCT) were segmented using three thresholds including the manufacturer default and ±5% in terms of equivalent mineral density. Ct.Th was determined in two ways: using a direct three-dimensional (3D) method and using an annular method, where cortical bone volume is divided by periosteal surface area. D comp (mg HA/cm3) was calculated based on the mean density-calibrated linear attenuation values of the cortical compartment. Finite element analysis was performed to evaluate the predictive ability of the annular and direct Ct.Th methods. Using the direct 3D method, a ±5% change in threshold resulted in a 2% mean difference in Ct.Th for both the radius and tibia. An average difference of 5% was found using the annular method. The change in threshold produced changes in D comp ranging 0.50-1.56% for both the tibia and radius. Annular Ct.Th correlated more strongly with whole-bone apparent modulus (R 2 = 0.64 vs. R 2 = 0.41). Both thickness calculation methods and threshold selection have a direct impact on cortical parameters derived from HR-pQCT images. Indirectly, these results suggest that moderate changes in tissue-level mineralization can affect cortical measures. Furthermore, while the direct 3D Ct.Th method is less sensitive to threshold effects, both methods are moderate predictors of mechanical strength, with the annular method being the stronger correlate.







Similar content being viewed by others
References
Miller PD (2006) Guidelines for the diagnosis of osteoporosis: T-scores vs fractures. Rev Endocr Metab Disord 7:15
Benhamou C-L (2007) Effects of osteoporosis medications on bone quality. Joint Bone Spine 74:39–47
Eswaran SK, Gupta A, Adams MF, Keaveny TM (2006) Cortical and trabecular load sharing in the human vertebral body. J Bone Miner Res 21:307–314
Pistoia W, van Rietbergen B, Lochmuller EM, Lill CA, Eckstein F, Ruegsegger P (2002) Estimation of distal radius failure load with micro-finite element analysis models based on three-dimensional peripheral quantitative computed tomography images. Bone 30:842–848
Boutroy S, Bouxsein ML, Munoz F, Delmas PD (2005) In vivo assessment of trabecular bone microarchitecture by high-resolution peripheral quantitative computed tomography. J Clin Endocrinol Metab 90:6508–6515
Khosla S, Riggs BL, Atkinson EJ, Oberg AL, McDaniel LJ, Holets M, Peterson JM, Melton LJ 3rd (2006) Effects of sex and age on bone microstructure at the ultradistal radius: a population-based noninvasive in vivo assessment. J Bone Miner Res 21:124–131
MacNeil JA, Boyd SK (2007) Accuracy of high-resolution peripheral quantitative computed tomography for measurement of bone quality. Med Eng Phys. doi:10.1016/j.medengphy.2006.11.002
Hildebrand T, Laib A, Muller R, Dequeker J, Ruegsegger P (1999) Direct three-dimensional morphometric analysis of human cancellous bone: microstructural data from spine, femur, iliac crest, and calcaneus. J Bone Miner Res 14:1167–1174
Laib A, Hauselmann HJ, Ruegsegger P (1998) In vivo high resolution 3D-QCT of the human forearm. Technol Health Care 6:329–337
Hildebrand T, Ruegsegger P (1997) A new method for the model-independent assessment of thickness in three-dimensional images. J Microsc 185:67–75
Muller RRP (1995) Three-dimensional finite element modelling of non-invasively assessed trabecular bone structures. Med Eng Phys 17:126–133
Van Rietbergen BWH, Huiskes R, Odgaard A (1995) A new method to determine trabecular bone elastic properties and loading using micromechanical finite-element models. J Biomech 28:69–81
Van Rietbergen BOA, Kabel J, Huiskes R (1996) Direct mechanics assessment of elastic symmetries and properties of trabecular bone architecture. J Biomech 29:1653–1657
Boivin GY, Chavassieux PM, Santora AC, Yates J, Meunier PJ (2000) Alendronate increases bone strength by increasing the mean degree of mineralization of bone tissue in osteoporotic women. Bone 27:687–694
Waarsing JH, Day JS, Weinans H (2004) An improved segmentation method for in vivo microCT imaging. J Bone Miner Res 19:1640–1650
Burghardt AJ, Kazakia GJ, Majumdar S (2007) A local adaptive threshold strategy for high resolution peripheral quantitative computed tomography of trabecular bone. Ann Biomed Eng 35(10):1678–1686
Khosla SML, Achenbach SJ, Oberg AL, Riggs BL (2006) Hormonal and biochemical determinants of trabecular microstructure at the ultradistal radius in women and men. J Clin Endocrinol Metab 91:885–891
Borah BDT, Ritman EL, Jorgensen SM, Liu S, Chmielewski PA, Phipps RJ, Zhou XSJ, Turner RT (2006) Long-term risedronate treatment normalizes mineralization and continues to preserve trabecular architecture: sequential triple biopsy studies with micro-computed tomography. Bone 39:345–352
Zoehrer RRP, Paschalis EP, Hofstaetter JG, Durchschlag E, Fratzl P, Phipps RKK (2006) Effects of 3- and 5-year treatment with risedronate on bone mineralization density distribution in triple biopsies of the iliac crest in postmenopausal women. J Bone Miner Res 21:1106–1112
Acknowledgments
The authors acknowledge funding from NIH RO1 AG17762 (S. M.). Furthermore, they thank Dr. Andres Laib from Scanco Medical for detailed technical insight and Jingyi Yu at the University of California-San Francisco for assistance with patient scanning.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Davis, K.A., Burghardt, A.J., Link, T.M. et al. The Effects of Geometric and Threshold Definitions on Cortical Bone Metrics Assessed by In Vivo High-Resolution Peripheral Quantitative Computed Tomography. Calcif Tissue Int 81, 364–371 (2007). https://doi.org/10.1007/s00223-007-9076-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-007-9076-3