Abstract
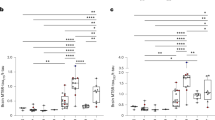
Limited cleavage promotes the aggregation propensity of protein tau in neurodegenerative tauopathies. Cathepsin S (CatS) is overexpressed in brains of patients suffering from tauopathies such as Alzheimer’s disease (AD). Furthermore, CatS serum levels correlate with survival in the elderly. The current study investigates whether limited cleavage by CatS promotes tau aggregation, and whether CatS serum levels may correlate with disease severity in tauopathies. Oligomer formation of fluorescently labeled protein tau was monitored by single particle fluorescence spectroscopy after coincubation with CatS. Tau cleavage patterns were investigated by SDS-PAGE. For serum analyses, samples were collected from 42 patients with probable progressive supranuclear palsy (PSP) according to NINDS-PSP criteria. Disease severity was assessed by PSP rating scale (PSP-RS), PSP staging system (PSP-S) and Schwab and England Activities of Daily Living (SEADL). CatS, cystatin C (CysC) and interleukin 6 (IL-6) serum levels were determined by ELISA, ECLIA and turbidimetry, respectively. SDS-PAGE demonstrated a distinct cleavage pattern of protein tau after coincubation with CatS. Furthermore, tau oligomer formation was increased 2.4-fold (p < 0.05) after limited cleavage. Serum CatS and CysC levels did not correlate with disease severity in PSP. Of note, IL-6 correlated with PSP-S (r = 0.41; 95% CI 0.11–0.65; p = 0.008), SEADL (r = −0.37; 95% CI −0.61 to −0.06; p = 0.017) and the history and gait/midline subdomains of the PSP-RS. While CatS facilitates tau aggregation in vitro, serum levels of CatS appear not to correlate with disease severity. The observed correlation of IL-6 with disease severity warrants further investigation of inflammatory markers in PSP.

Similar content being viewed by others
References
Bader B, Nubling G, Mehle A, Nobile S, Kretzschmar H, Giese A (2011) Single particle analysis of tau oligomer formation induced by metal ions and organic solvents. Biochem Biophys Res Commun 411:190–196. doi:10.1016/j.bbrc.2011.06.135
Bieschke J, Giese A, Schulz-Schaeffer W, Zerr I, Poser S, Eigen M, Kretzschmar H (2000) Ultrasensitive detection of pathological prion protein aggregates by dual-color scanning for intensely fluorescent targets. Proc Natl Acad Sci USA 97:5468–5473
Chen SY et al (2012) Sequence variants of interleukin 6 (IL-6) are significantly associated with a decreased risk of late-onset Alzheimer’s disease. J Neuroinflammation 9:21. doi:10.1186/1742-2094-9-21
Flannery T et al (2006) Cathepsin S expression: an independent prognostic factor in glioblastoma tumours—a pilot study. Int J Cancer 119:854–860. doi:10.1002/ijc.21911
Gamblin TC et al (2003) Caspase cleavage of tau: linking amyloid and neurofibrillary tangles in Alzheimer’s disease. Proc Natl Acad Sci USA 100:10032–10037. doi:10.1073/pnas.1630428100
Gerhard A, Trender-Gerhard I, Turkheimer F, Quinn NP, Bhatia KP, Brooks DJ (2006) In vivo imaging of microglial activation with [11C](R)-PK11195 PET in progressive supranuclear palsy. Mov Disord 21:89–93. doi:10.1002/mds.20668
Gerson J et al (2016) Tau oligomers derived from traumatic brain injury cause cognitive impairment and accelerate onset of pathology in Htau mice. J Neurotrauma 33:2034–2043. doi:10.1089/neu.2015.4262
Gerson JE, Sengupta U, Kayed R (2017) Tau oligomers as pathogenic seeds: preparation and propagation in vitro and in vivo methods. Mol Biol 1523:141–157. doi:10.1007/978-1-4939-6598-4_9
Golbe LI, Ohman-Strickland PA (2007) A clinical rating scale for progressive supranuclear palsy. Brain 130:1552–1565. doi:10.1093/brain/awm032
Gormley JA et al (2011) The role of Cathepsin S as a marker of prognosis and predictor of chemotherapy benefit in adjuvant CRC: a pilot study. Br J Cancer 105:1487–1494. doi:10.1038/bjc.2011.408
Jobs E et al (2011) Association between serum cathepsin S and mortality in older adults. JAMA 306:1113–1121. doi:10.1001/jama.2011.1246
Khlistunova I et al (2006) Inducible expression of Tau repeat domain in cell models of tauopathy: aggregation is toxic to cells but can be reversed by inhibitor drugs. J Biol Chem 281:1205–1214. doi:10.1074/jbc.M507753200
Lemere CA, Munger JS, Shi GP, Natkin L, Haass C, Chapman HA, Selkoe DJ (1995) The lysosomal cysteine protease, cathepsin S, is increased in Alzheimer’s disease and Down syndrome brain. An immunocytochemical study. Am J Pathol 146:848–860
Litvan I et al (1996) Accuracy of clinical criteria for the diagnosis of progressive supranuclear palsy (Steele-Richardson–Olszewski syndrome). Neurology 46:922–930
Lv BJ, Lindholt JS, Cheng X, Wang J, Shi GP (2012) Plasma cathepsin S and cystatin C levels and risk of abdominal aortic aneurysm: a randomized population-based study. PLoS One 7:e41813. doi:10.1371/journal.pone.0041813
Munger JS et al (1995) Lysosomal processing of amyloid precursor protein to A beta peptides: a distinct role for cathepsin S. Biochem J 311(Pt 1):299–305
Nilsson E, Bodolea C, Gordh T, Larsson A (2013) Cerebrospinal fluid cathepsin B and S. Neurol Sci 34:445–448. doi:10.1007/s10072-012-1022-0
Nubling G, Bader B, Levin J, Hildebrandt J, Kretzschmar H, Giese A (2012a) Synergistic influence of phosphorylation and metal ions on tau oligomer formation and coaggregation with alpha-synuclein at the single molecule level. Mol Neurodegener 7:35. doi:10.1186/1750-1326-7-35
Nubling G, Levin J, Bader B, Israel L, Botzel K, Lorenzl S, Giese A (2012b) Limited cleavage of tau with matrix-metalloproteinase MMP-9, but not MMP-3, enhances tau oligomer formation. Exp Neurol 237:470–476. doi:10.1016/j.expneurol.2012.07.018
Park SK, Kim K, Page GP, Allison DB, Weindruch R, Prolla TA (2009) Gene expression profiling of aging in multiple mouse strains: identification of aging biomarkers and impact of dietary antioxidants. Aging Cell 8:484–495. doi:10.1111/j.1474-9726.2009.00496.x
Polyakova O et al (2009) Proteolysis of prion protein by cathepsin S generates a soluble beta-structured intermediate oligomeric form, with potential implications for neurotoxic mechanisms. Eur Biophys J 38:209–218. doi:10.1007/s00249-008-0371-3
Schwab (1969) Projection technique for evaluating surgery in Parkinson’s Disease. Paper presented at the Third Symposium on Parkinson’s Disease, Edinburgh, Scotland
Wang M, Jia J (2010) The interleukin-6 gene -572C/G promoter polymorphism modifies Alzheimer’s risk in APOE epsilon 4 carriers. Neurosci Lett 482:260–263. doi:10.1016/j.neulet.2010.07.051
Wang YP, Biernat J, Pickhardt M, Mandelkow E, Mandelkow EM (2007) Stepwise proteolysis liberates tau fragments that nucleate the Alzheimer-like aggregation of full-length tau in a neuronal cell model. Proc Natl Acad Sci USA 104:10252–10257. doi:10.1073/pnas.0703676104
Acknowledgements
This study was funded by the Friedrich-Baur-Foundation (Grant No. 25/13). The authors would like to thank Ms. Sabine Paul for excellent technical assistance. The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nübling, G., Schuberth, M., Feldmer, K. et al. Cathepsin S increases tau oligomer formation through limited cleavage, but only IL-6, not cathespin S serum levels correlate with disease severity in the neurodegenerative tauopathy progressive supranuclear palsy. Exp Brain Res 235, 2407–2412 (2017). https://doi.org/10.1007/s00221-017-4978-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-017-4978-4