Abstract
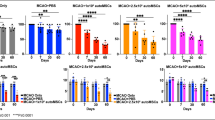
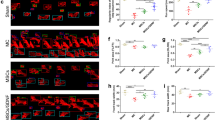
A novel type of non-tumorigenic pluripotent stem cell, the Muse cell (multi-lineage, differentiating stress enduring cell), resides in the connective tissue and in cultured mesenchymal stem cells (MSCs) and is reported to differentiate into multiple cell types according to the microenvironment to repair tissue damage. We examined the efficiency of Muse cells in a mouse intracerebral hemorrhage (ICH) model. Seventy μl of cardiac blood was stereotactically injected into the left putamen of immunodeficient mice. Five days later, 2 × 105 of human bone marrow MSC-derived Muse cells (n = 6) or cells other than Muse cells in MSCs (non-Muse, n = 6) or the same volume of PBS (n = 11) was injected into the ICH cavity. Water maze and motor function tests were implemented for 68 days, and immunohistochemistry for NeuN, MAP2 and GFAP was done. The Muse group showed impressive recovery: Recovery was seen in the water maze after day 19, and motor functions after 5 days was compared with the other two groups, with a significant statistical difference (p < 0.05). The survival rate of the engrafted cells in the Muse group was significantly higher than in the non-Muse group (p < 0.05) at day 69, and those cells showed positivity for NeuN (~57%) and MAP-2 (~41.6%). Muse cells could remain in the ICH brain, differentiate into neural-lineage cells and restore functions without inducing them into neuronal cells by gene introduction and cytokine treatment prior to transplantation. A simple collection of Muse cells and their supply to the brain in naïve state facilitates regenerative therapy in ICH.



Similar content being viewed by others
References
Amariglio N et al (2009) Donor-derived brain tumor following neural stem cell transplantation in an ataxia telangiectasia patient. PLoS Med 6:e1000029. doi:10.1371/journal.pmed.1000029
Belayev L et al (2003) Experimental intracerebral hemorrhage in the mouse: histological, behavioral, and hemodynamic characterization of a double-injection model. Stroke 34:2221–2227. doi:10.1161/01.STR.0000088061.06656.1E
Chen J, Li Y, Wang L, Zhang Z, Lu D, Lu M, Chopp M (2001) Therapeutic benefit of intravenous administration of bone marrow stromal cells after cerebral ischemia in rats. Stroke 32:1005–1011
Chen J et al (2012) Transplantation of adipose-derived stem cells is associated with neural differentiation and functional improvement in a rat model of intracerebral hemorrhage. CNS Neurosci Ther 18:847–854. doi:10.1111/j.1755-5949.2012.00382.x
Deinsberger W, Vogel J, Kuschinsky W, Auer LM, Boker DK (1996) Experimental intracerebral hemorrhage: description of a double injection model in rats. Neurol Res 18:475–477
Feng M et al (2011) Serial 18F-FDG PET demonstrates benefit of human mesenchymal stem cells in treatment of intracerebral hematoma: a translational study in a primate model. J Nucl Med 52:90–97. doi:10.2967/jnumed.110.080325
Flaherty ML et al (2005) Racial variations in location and risk of intracerebral hemorrhage. Stroke 36:934–937. doi:10.1161/01.STR.0000160756.72109.95
Hickenbottom SL, Grotta JC, Strong R, Denner LA, Aronowski J (1999) Nuclear factor-kappaB and cell death after experimental intracerebral hemorrhage in rats. Stroke 30:2472–2477 (discussion 2477–2478)
Honmou O et al (2011) Intravenous administration of auto serum-expanded autologous mesenchymal stem cells in stroke. Brain 134:1790–1807. doi:10.1093/brain/awr063
Hua Y, Keep RF, Hoff JT, Xi G (2007) Brain injury after intracerebral hemorrhage: the role of thrombin and iron. Stroke 38:759–762. doi:10.1161/01.STR.0000247868.97078.10
Inagawa T, Ohbayashi N, Takechi A, Shibukawa M, Yahara K (2003) Primary intracerebral hemorrhage in Izumo City, Japan: incidence rates and outcome in relation to the site of hemorrhage. Neurosurgery 53:1283–1297 (discussion 1297–1288)
Jeong SW, Chu K, Jung KH, Kim SU, Kim M, Roh JK (2003) Human neural stem cell transplantation promotes functional recovery in rats with experimental intracerebral hemorrhage. Stroke 34:2258–2263. doi:10.1161/01.STR.0000083698.20199.1F
Karp JM, Leng Teo GS (2009) Mesenchymal stem cell homing: the devil is in the details. Cell Stem Cell 4:206–216. doi:10.1016/j.stem.2009.02.001
Kuroda Y et al (2010) Unique multipotent cells in adult human mesenchymal cell populations. Proc Natl Acad Sci USA 107:8639–8643. doi:10.1073/pnas.0911647107
Kuroda Y, Wakao S, Kitada M, Murakami T, Nojima M, Dezawa M (2013) Isolation, culture and evaluation of multilineage-differentiating stress-enduring (Muse) cells. Nat Protoc 8:1391–1415. doi:10.1038/nprot.2013.076
Lee HJ, Lim IJ, Lee MC, Kim SU (2010) Human neural stem cells genetically modified to overexpress brain-derived neurotrophic factor promote functional recovery and neuroprotection in a mouse stroke model. J Neurosci Res 88:3282–3294. doi:10.1002/jnr.22474
Li ZM, Zhang ZT, Guo CJ, Geng FY, Qiang F, Wang LX (2013) Autologous bone marrow mononuclear cell implantation for intracerebral hemorrhage-a prospective clinical observation. Clin Neurol Neurosurg 115:72–76. doi:10.1016/j.clineuro.2012.04.030
Liang H et al (2013) Transplantation of bone marrow stromal cells enhances nerve regeneration of the corticospinal tract and improves recovery of neurological functions in a collagenase-induced rat model of intracerebral hemorrhage. Mol Cells 36:17–24. doi:10.1007/s10059-013-2306-9
Lu A, Tang Y, Ran R, Ardizzone TL, Wagner KR, Sharp FR (2006) Brain genomics of intracerebral hemorrhage. J Cereb Blood Flow Metab 26:230–252. doi:10.1038/sj.jcbfm.9600183
Mayne M, Fotheringham J, Yan HJ, Power C, Del Bigio MR, Peeling J, Geiger JD (2001) Adenosine A2A receptor activation reduces proinflammatory events and decreases cell death following intracerebral hemorrhage. Ann Neurol 49:727–735
Mimura T, Dezawa M, Kanno H, Yamamoto I (2005) Behavioral and histological evaluation of a focal cerebral infarction rat model transplanted with neurons induced from bone marrow stromal cells. J Neuropathol Exp Neurol 64:1108–1117
Nagai A et al (2007) Multilineage potential of stable human mesenchymal stem cell line derived from fetal marrow. PLoS ONE 2:e1272. doi:10.1371/journal.pone.0001272
Parr AM, Tator CH, Keating A (2007) Bone marrow-derived mesenchymal stromal cells for the repair of central nervous system injury. Bone Marrow Transplant 40:609–619. doi:10.1038/sj.bmt.1705757
Power C et al (2003) Intracerebral hemorrhage induces macrophage activation and matrix metalloproteinases. Ann Neurol 53:731–742. doi:10.1002/ana.10553
Qin J et al (2013a) Functional recovery after transplantation of induced pluripotent stem cells in a rat hemorrhagic stroke model. Neurosci Lett 554:70–75. doi:10.1016/j.neulet.2013.08.047
Qin J et al (2013b) Transplantation of human neuro-epithelial-like stem cells derived from induced pluripotent stem cells improves neurological function in rats with experimental intracerebral hemorrhage. Neurosci Lett 548:95–100. doi:10.1016/j.neulet.2013.05.007
Qureshi AI et al (2003) Apoptosis as a form of cell death in intracerebral hemorrhage. Neurosurgery 52:1041–1047 (discussion 1047–1048)
Ries C, Egea V, Karow M, Kolb H, Jochum M, Neth P (2007) MMP-2, MT1-MMP, and TIMP-2 are essential for the invasive capacity of human mesenchymal stem cells: differential regulation by inflammatory cytokines. Blood 109:4055–4063. doi:10.1182/blood-2006-10-051060
Ronning OM, Guldvog B, Stavem K (2001) The benefit of an acute stroke unit in patients with intracranial haemorrhage: a controlled trial. J Neurol Neurosurg Psychiatry 70:631–634
Sacco S, Marini C, Toni D, Olivieri L, Carolei A (2009) Incidence and 10-year survival of intracerebral hemorrhage in a population-based registry. Stroke 40:394–399. doi:10.1161/STROKEAHA.108.523209
Seyfried DM, Han Y, Yang D, Ding J, Savant-Bhonsale S, Shukairy MS, Chopp M (2008) Mannitol enhances delivery of marrow stromal cells to the brain after experimental intracerebral hemorrhage. Brain Res 1224:12–19. doi:10.1016/j.brainres.2008.05.080
Shimamura N, Munakata A, Naraoka M, Nakano T, Ohkuma H (2011) Decompressive hemi-craniectomy is not necessary to rescue supratentorial hypertensive intracerebral hemorrhage patients: consecutive single-center experience. Acta Neurochir Suppl 111:415–419. doi:10.1007/978-3-7091-0693-8_71
Uchida H et al (2016) Transplantation of unique subpopulation of fibroblasts, muse cells, ameliorates experimental stroke possibly via robust neuronal differentiation. Stem Cells 34:160–173. doi:10.1002/stem.2206
Wakao S, Kuroda Y, Ogura F, Shigemoto T, Dezawa M (2012) Regenerative effects of mesenchymal stem cells: contribution of muse cells, a novel pluripotent stem cell type that resides in mesenchymal cells. Cells 1:1045–1060. doi:10.3390/cells1041045
Xi G, Keep RF, Hoff JT (2006) Mechanisms of brain injury after intracerebral haemorrhage. Lancet Neurol 5:53–63. doi:10.1016/S1474-4422(05)70283-0
Yamauchi T, Kuroda Y, Morita T, Shichinohe H, Houkin K, Dezawa M, Kuroda S (2015) Therapeutic effects of human multilineage-differentiating stress enduring (MUSE) cell transplantation into infarct brain of mice. PLoS ONE 10:e0116009. doi:10.1371/journal.pone.0116009
Zheng M et al (2015) Iron-induced necrotic brain cell death in rats with different aerobic capacity. Transl Stroke Res 6:215–223. doi:10.1007/s12975-015-0388-8
Acknowledgements
We thank Mark Inglin (University of Basel) for his editorial assistance.
Authors’ contributions
NS took part in conception and design, financial support, data analysis and interpretation and manuscript writing. KK and LW performed collection and/or assembly of data. MN carried out data analysis and interpretation. HU and SW made provision for study material. MD took part in conception and design, and the final approval of manuscript. HO was involved administrative support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We have no conflict of interest.
Availability of data and material
The datasets during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
This work was supported by Grants-in-Aid for Scientific Research of Japan, grant number 26462149 for Norihito Shimamura.
Rights and permissions
About this article
Cite this article
Shimamura, N., Kakuta, K., Wang, L. et al. Neuro-regeneration therapy using human Muse cells is highly effective in a mouse intracerebral hemorrhage model. Exp Brain Res 235, 565–572 (2017). https://doi.org/10.1007/s00221-016-4818-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-016-4818-y