Abstract
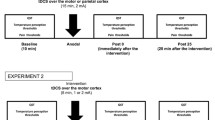
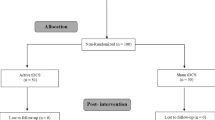
Transcranial direct current stimulation (tDCS) is a non-invasive neuromodulatory technique that can affect human pain perception. Placebo effects are present in most treatments and could therefore also interact with treatment effects in tDCS. The present study investigated whether short-term tDCS reduced heat pain intensity, stress, blood pressure and increased heat pain thresholds in healthy volunteers when controlling for placebo effects. Seventy-five (37 females) participants were randomized into three groups: (1) active tDCS group receiving anodal tDCS (2 mA) for 7 min to the primary motor cortex (M1), (2) placebo group receiving the tDCS electrode montage but only active tDCS stimulation for 30 s and (3) natural history group that got no tDCS montage but the same pain stimulation as the active tDCS and the placebo group. Heat pain was induced by a PC-controlled thermode attached to the left forearm. Pain intensity was significantly lower in the active tDCS group when examining change scores (pretest–posttest) for the 47 °C condition. The placebo group displayed lower pain compared with the natural history group, displaying a significant placebo effect. In the 43 and 45 °C conditions, the effect of tDCS could not be separated from placebo effects. The results revealed no effects on pain thresholds. There was a tendency that active tDCS reduced stress and systolic blood pressure, however, not significant. In sum, tDCS had an analgesic effect on high-intensity pain, but the effect of tDCS could not be separated from placebo effects for medium and low pain.




Similar content being viewed by others
References
Alonzo A, Brassil J, Taylor JL, Martin D, Loo CK (2012) Daily transcranial direct current stimulation (tDCS) leads to greater increases in cortical excitability than second daily transcranial direct current stimulation. Brain Stimul 5:208–213. doi:10.1016/j.brs.2011.04.006
Antal A, Brepohl N, Poreisz C, Boros K, Csifcsak G, Paulus W (2008) Transcranial direct current stimulation over somatosensory cortex decreases experimentally induced acute pain perception. Clin J Pain 24:56–63
Aslaksen PM, Flaten MA (2008) The roles of physiological and subjective stress in the effectiveness of a placebo on experimentally induced pain. Psychosom Med 70:811–818. doi:10.1097/PSY.001361818105ed
Aslaksen PM, Bystad M, Vambheim SM, Flaten MA (2011) Gender differences in placebo analgesia: event-related potentials and emotional modulation. Psychosom Med 73:193–199. doi:10.1097/PSY.0b013e3182080d73
Benedetti F, Rainero I, Pollo A (2003) New insights into placebo analgesia. Curr Opin Anaesthesiol 16:515–519
Boggio PS, Zaghi S, Lopes M, Fregni F (2008) Modulatory effects of anodal transcranial direct current stimulation on perception and pain thresholds in healthy volunteers. Eur J Neurol 15:1124–1130. doi:10.1111/j.1468-1331.2008.02270.x
Borckardt JJ, Bikson M, Frohman H et al (2012) A pilot study of the tolerability and effects of high-definition transcranial direct current stimulation (HD-tDCS) on pain perception. J Pain 13:112–120. doi:10.1016/j.jpain.2011.07.001
Csifcsak G, Antal A, Hillers F et al (2009) Modulatory effects of transcranial direct current stimulation on laser-evoked potentials. Pain Med 10:122–132. doi:10.1111/j.1526-4637.2008.00508.x
DosSantos MF, Love TM, Martikainen IK et al (2012) Immediate effects of tDCS on the mu-opioid system of a chronic pain patient. Front Psychiatry 3:93. doi:10.3389/fpsyt.2012.00093
Enck P, Benedetti F, Schedlowski M (2008) New insights into the placebo and nocebo responses. Neuron 59:195–206
Enck P, Bingel U, Schedlowski M, Rief W (2013) The placebo response in medicine: minimize, maximize or personalize? Nat Rev Drug Discov 12:191–204. doi:10.1038/nrd3923
Fields HL, Levine JD (1984) Placebo analgesia—a role for endorphins. Trends Neurosci 7:271–273. doi:10.1016/s0166-2236(84)80193-9
Fregni F, Pascual-Leone A (2007) Technology insight: noninvasive brain stimulation in neurology—perspectives on the therapeutic potential of rTMS and tDCS. Nat Clin Pract Neurol 3:383–393. doi:10.1038/ncpneuro0530
Fregni F, Boggio PS, Lima MC et al (2006a) A sham-controlled, phase II trial of transcranial direct current stimulation for the treatment of central pain in traumatic spinal cord injury. Pain 122:197–209. doi:10.1016/j.pain.2006.02.023
Fregni F, Gimenes R, Valle AC et al (2006b) A randomized, sham-controlled, proof of principle study of transcranial direct current stimulation for the treatment of pain in fibromyalgia. Arthritis Rheum 54:3988–3998. doi:10.1002/art.22195
Garcia-Larrea L, Peyron R, Mertens P et al (1999) Electrical stimulation of motor cortex for pain control: a combined PET-scan and electrophysiological study. Pain 83:259–273
Hansen N, Obermann M, Poitz F et al (2011) Modulation of human trigeminal and extracranial nociceptive processing by transcranial direct current stimulation of the motor cortex. Cephalalgia 31:661–670. doi:10.1177/0333102410390394
Jurgens TP, Schulte A, Klein T, May A (2012) Transcranial direct current stimulation does neither modulate results of a quantitative sensory testing protocol nor ratings of suprathreshold heat stimuli in healthy volunteers. Eur J Pain 16:1251–1263. doi:10.1002/j.1532-2149.2012.00135.x
Lang N, Siebner HR, Ward NS et al (2005) How does transcranial DC stimulation of the primary motor cortex alter regional neuronal activity in the human brain? Eur J Neurosci 22:495–504. doi:10.1111/j.1460-9568.2005.04233.x
Lyby PS, Aslaksen PM, Flaten MA (2011) Variability in placebo analgesia and the role of fear of pain—an ERP study. Pain 152:2405–2412. doi:10.1016/j.pain.2011.07.010
Mackay C, Cox T, Burrows G, Lazzerini T (1978) Inventory for measurement of self-reported stress and arousal. Brit J Soc Clin Psychol 17:283–284
Muthen B, Brown HC (2009) Estimating drug effects in the presence of placebo response: causal inference using growth mixture modeling. Stat Med 28:3363–3385. doi:10.1002/sim.3721
Mylius V, Borckardt JJ, Lefaucheur JP (2012a) Noninvasive cortical modulation of experimental pain. Pain 153:1350–1363. doi:10.1016/j.pain.2012.04.009
Mylius V, Jung M, Menzler K et al (2012b) Effects of transcranial direct current stimulation on pain perception and working memory. Eur J Pain 16:974–982. doi:10.1002/j.1532-2149.2011.00105.x
Nitsche MA, Paulus W (2000) Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol 527:633–639. doi:10.1111/j.1469-7793.2000.t01-1-00633.x
Nitsche MA, Paulus W (2001) Sustained excitability elevations induced by transcranial DC motor cortex stimulation in humans. Neurology 57:1899–1901
O’Connell NE, Wand BM, Marston L, Spencer S, DeSouza LH (2010) Non-invasive brain stimulation techniques for chronic pain. Cochrane Database Syst Rev. doi:10.1002/14651858.CD008208.pub2
O’Connell NE, Cossar J, Marston L, Wand BM, Bunce D, Moseley GL, De Souza LH (2012) Rethinking clinical trials of transcranial direct current stimulation: participant and assessor blinding is inadequate at intensities of 2 mA. PLoS One 7:e47514. doi:10.1371/journal.pone.0047514
Rhudy JL, Meagher MW (2000) Fear and anxiety: divergent effects on human pain thresholds. Pain 84:65–75
Varelmann D, Pancaro C, Cappiello EC, Camann WR (2010) Nocebo-induced hyperalgesia during local anesthetic injection. Anesth Analg 110:868–870. doi:10.1213/ANE.0b013e3181cc5727
Wager TD, Rilling JK, Smith EE et al (2004) Placebo-induced changes in fMRI in the anticipation and experience of pain. Science 303:1162–1167
Acknowledgments
The present study was funded by the University of Tromsø and by a grant from the Norwegian Fibromyalgia Association to Per M. Aslaksen.
Conflict of interest
The authors have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aslaksen, P.M., Vasylenko, O. & Fagerlund, A.J. The effect of transcranial direct current stimulation on experimentally induced heat pain. Exp Brain Res 232, 1865–1873 (2014). https://doi.org/10.1007/s00221-014-3878-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-014-3878-0