Abstract
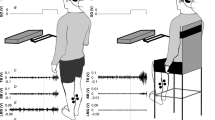
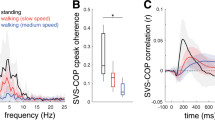
Electrical stimulation of the human vestibular nerve evokes a postural response which, unlike visually evoked sway, is unaffected by stimulus predictability. However, responses can be modified by changes in the level of background sway. Here, the effect of voluntary changes in sway magnitude upon the response to vestibular stimulation is investigated. Subjects were asked to stand either relaxed or still while stochastic vestibular stimulation (SVS) was applied to the mastoid processes (1 mA root-mean-square; 0.05–5 Hz). Calf muscle activity, ground-reaction force and sway responses were characterised in the frequency and time domains using cross-spectra and cross-correlations (CC), respectively. SVS induced coherent EMG, lateral force and sway responses. Differences in response gain between still and relaxed conditions largely reflected differences in signal power across frequencies, and peak EMG CC responses correlated strongly with background EMG changes. However, when data were normalised to account for changes in signal power, early EMG responses were almost identical between conditions, but after 232 ms, they diverged. Standing still caused heavy attenuation of the late component of the EMG response, reducing response duration by 825 ms. Similar effects were observed in force and sway, and all postural signals showed less phase lag with SVS below 2 Hz when standing still. These results demonstrate that the vestibular-evoked postural response consists of two parts: an early high-frequency component, which scales with background activity but is otherwise inflexible, and a late low-frequency component, which can be heavily attenuated by voluntary control resulting in earlier termination of the sway response.




Similar content being viewed by others
References
Amjad AM, Halliday DM, Rosenberg JR, Conway BA (1997) An extended difference of coherence test for comparing and combining several independent coherence estimates: theory and application to the study of motor units and physiological tremor. J Neurosci Methods 73:69–79
Cathers I, Day BL, Fitzpatrick RC (2005) Otolith and canal reflexes in human standing. J Physiol 563:229–234
Dakin CJ, Son GM, Inglis JT, Blouin JS (2007) Frequency response of human vestibular reflexes characterized by stochastic stimuli. J Physiol 583:1117–1127
Day BL, Cole J (2002) Vestibular-evoked postural responses in the absence of somatosensory information. Brain 125:2081–2088
Day BL, Guerraz M (2007) Feedforward versus feedback modulation of human vestibular-evoked balance responses by visual self-motion information. J Physiol 582:153–161
Day BL, Severac CA, Bartolomei L, Pastor MA, Lyon IN (1997) Human body-segment tilts induced by galvanic stimulation: a vestibularly driven balance protection mechanism. J Physiol 500(Pt 3):661–672
Fitzpatrick RC, Day BL (2004) Probing the human vestibular system with galvanic stimulation. J Appl Physiol 96:2301–2316
Fitzpatrick RC, Gorman RB, Burke D, Gandevia SC (1992) Postural proprioceptive reflexes in standing human subjects: bandwidth of response and transmission characteristics. J Physiol 458:69–83
Fitzpatrick R, Burke D, Gandevia SC (1996) Loop gain of reflexes controlling human standing measured with the use of postural and vestibular disturbances. J Neurophysiol 76:3994–4008
Gilhodes JC, Gurfinkel VS, Roll JP (1992) Role of Ia muscle spindle afferents in post-contraction and post-vibration motor effect genesis. Neurosci Lett 135:247–251
Goldberg JM, Smith CE, Fernandez C (1984) Relation between discharge regularity and responses to externally applied galvanic currents in vestibular nerve afferents of the squirrel monkey. J Neurophysiol 51:1236–1256
Guerraz M, Day BL (2005) Expectation and the vestibular control of balance. J Cogn Neurosci 17:463–469
Guerraz M, Thilo KV, Bronstein AM, Gresty MA (2001) Influence of action and expectation on visual control of posture. Brain Res Cogn Brain Res 11:259–266
Halliday DM, Rosenberg JR, Amjad AM, Breeze P, Conway BA, Farmer SF (1995) A framework for the analysis of mixed time series/point process data–theory and application to the study of physiological tremor, single motor unit discharges and electromyograms. Prog Biophys Mol Biol 64:237–278
Horak FB, Hlavacka F (2001) Somatosensory loss increases vestibulospinal sensitivity. J Neurophysiol 86:575–585
Horak FB, Moore S (1989) Lateral postural responses: the effect of stance width and perturbation amplitude. Phys Ther 69:363
Lund S, Broberg C (1983) Effects of different head positions on postural sway in man induced by a reproducible vestibular error signal. Acta Physiol Scand 117:307–309
Marsden CD, Rothwell JC, Day BL (1983) Long-latency automatic responses to muscle stretch in man: origin and function. Adv Neurol 39:509–539
Marsden JF, Castellote J, Day BL (2002) Bipedal distribution of human vestibular-evoked postural responses during asymmetrical standing. J Physiol 542:323–331
Pastor MA, Day BL, Marsden CD (1993) Vestibular induced postural responses in Parkinson’s disease. Brain 116(Pt 5):1177–1190
Pavlik AE, Inglis JT, Lauk M, Oddsson L, Collins JJ (1999) The effects of stochastic galvanic vestibular stimulation on human postural sway. Exp Brain Res 124:273–280
Acknowledgments
Thanks to David Halliday for help with data analysis, Mark Hollands and Frank Eves for equipment use, Steve Allen for technical assistance and Martin Lakie for comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reynolds, R.F. The effect of voluntary sway control on the early and late components of the vestibular-evoked postural response. Exp Brain Res 201, 133–139 (2010). https://doi.org/10.1007/s00221-009-2017-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-009-2017-9