Abstract
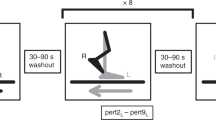
This study investigated the effects of walking in an elastic force field (FF) for varying durations (49–1,629 strides) on: (a) the magnitude and duration of aftereffects, (b) performance in the FF on the next day. On day 1 in the FF, subjects (n = 17) showed an initial large error in peak toe velocity during swing (9–61% above baseline) that was largely reduced within the first 40 strides. After FF removal, subjects (16/17) showed aftereffects: (1) reduction in toe velocity (9–38% below baseline), (2) increase in hamstrings muscle activation. While the magnitude of aftereffects did not correlate to FF exposure duration, aftereffects duration did (p < 0.05). During FF exposure on day 2, initial toe velocity error was smaller than on day 1 (13/17 subjects, p < 0.001) regardless of day 1 exposure duration. This was associated with an earlier effective onset in hamstring activation. These results suggest that during walking, even short daily exposures to a FF (≥49 strides) lead to significant retention of the new movement parameters.




Similar content being viewed by others
References
Bailey CH, Kandel ER (1993) Structural changes accompanying memory storage. Annu Rev Physiol 55:397–426
Bailey CH, Bartsch D, Kandel ER (1996) Toward a molecular definition of long-term memory storage. Proc Natl Acad Sci USA 93:13445–13452
Blanchette A, Bouyer LJ (2009) Timing-specific transfer of adapted muscle activity after walking in an elastic force field. J Neurophysiol. 102:568–577
Brashers-Krug T, Shadmehr R, Bizzi E (1996) Consolidation in human motor memory. Nature 382:252–255
Caithness G, Osu R, Bays P, Chase H, Klassen J, Kawato M, Wolpert DM, Flanagan JR (2004) Failure to consolidate the consolidation theory of learning for sensorimotor adaptation tasks. J Neurosci 24:8662–8671
Choi JT, Bastian AJ (2007) Adaptation reveals independent control networks for human walking. Nat Neurosci 10:1055–1062
Dietz V, Zijlstra W, Duysens J (1994) Human neuronal interlimb coordination during split-belt locomotion. Exp Brain Res 101:513–520
Doyon J, Benali H (2005) Reorganization and plasticity in the adult brain during learning of motor skills. Curr Opin Neurobiol 15:161–167
Dudai Y (2004) The neurobiology of consolidations, or, how stable is the engram? Annu Rev Psychol 55:51–86
Ebbinghaus H (1913) Memory: a contribution to experimental psychology. Dover Publications, New York
Emken JL, Reinkensmeyer DJ (2005) Robot-enhanced motor learning: accelerating internal model formation during locomotion by transient dynamic amplification. IEEE Trans Neural Syst Rehabil Eng 13:33–39
Fortin K, Blanchette A, McFadyen BJ, Bouyer LJ (2007) Effects of exposure duration to an elastic force field during human walking. Soc Neurosci (abstract)
Freriks B, Hermens H, Disselhorst-Klug C, Rau G (1999) The recommendations for sensors and sensor placement procedures for surface electromyography. In: Hermens H, Freriks L, Merletti R, Hagg G, Stegeman D, Blok J, Rau G, Enschede Disselhorst-KlugsC (eds) SENIAM 8: European recommendations for surface electromyography. Roessingh Research and Development BV, The Netherlands, pp 15–53
Gordon KE, Ferris DP (2007) Learning to walk with a robotic ankle exoskeleton. J Biomech 40:2636–2644
Gordon CR, Fletcher WA, Melvill JG, Block EW (1995) Adaptive plasticity in the control of locomotor trajectory. Exp Brain Res 102:540–545
Hebb DO (1949) The organization of behavior: a neurophsychological theory. Wiley, New York
Hinder MR, Milner TE (2005) Novel strategies in feedforward adaptation to a position-dependent perturbation. Exp Brain Res 165:239–249
Joiner WM, Smith MA (2008) Long-term retention explained by a model of short-term learning in the adaptive control of reaching. J Neurophysiol 100:2948–2955
Kadaba MP, Wootten ME, Gainey J, Cochran GV (1985) Repeatability of phasic muscle activity: performance of surface and intramuscular wire electrodes in gait analysis. J Orthop Res 3:350–359
Kadaba MP, Ramakrishnan HK, Wootten ME, Gainey J, Gorton G, Cochran GV (1989) Repeatability of kinematic, kinetic, and electromyographic data in normal adult gait. J Orthop Res 7:849–860
Kandel ER (2001) The molecular biology of memory storage: a dialogue between genes and synapses. Science 294:1030–1038
Krakauer JW, Shadmehr R (2006) Consolidation of motor memory. Trends Neurosci 29:58–64
Krueger WCF (1929) The effect of overlearning on retention. J Exp Psychol 12:71–78
Lackner JR, DiZio P (1994) Rapid adaptation to Coriolis force perturbations of arm trajectory. J Neurophysiol 72:299–313
Lackner JR, DiZio P (2000) Aspects of body self-calibration. Trends Cogn Sci 4:279–288
Lam T, Anderschitz M, Dietz V (2006) Contribution of feedback and feedforward strategies to locomotor adaptations. J Neurophysiol 95:766–773
Layne CS, McDonald PV, Bloomberg JJ (1997) Neuromuscular activation patterns during treadmill walking after space flight. Exp Brain Res 113:104–116
Luh CW (1922) The conditions of retention. Psychol Monogr 31:1–87
Noble JW, Prentice SD (2006) Adaptation to unilateral change in lower limb mechanical properties during human walking. Exp Brain Res 169:482–495
Patla AE (1996) Neurobiomechanical bases for the control of human locomotion. In: Bronstein A, Brandt T, Wolloncott MH (eds) Clinical aspects of balance and gait disorders. Arnold, London, pp 19–40
Patterson RM, Stegink Jansen CW, Hogan HA, Nassif MD (2001) Material properties of thera-band tubing. Phys Ther 81:1437–1445
Prokop T, Berger W, Zijlstra W, Dietz V (1995) Adaptational and learning processes during human split-belt locomotion: interaction between central mechanisms and afferent input. Exp Brain Res 106:449–456
Reisman DS, Block HJ, Bastian AJ (2005) Interlimb coordination during locomotion: what can be adapted and stored? J Neurophysiol 94:2403–2415
Savion-Lemieux T, Penhune VB (2005) The effects of practice and delay on motor skill learning and retention. Exp Brain Res 161:423–431
Seeds NW, Williams BL, Bickford PC (1995) Tissue plasminogen activator induction in Purkinje neurons after cerebellar motor learning. Science 270:1992–1994
Shadmehr R, Mussa-Ivaldi FA (1994) Adaptive representation of dynamics during learning of a motor task. J Neurosci 14:3208–3224
Smith MA, Ghazizadeh A, Shadmehr R (2006) Interacting adaptive processes with different timescales underlie short-term motor learning. PLoS Biol 4:e179
Stickgold R, Walker MP (2007) Sleep-dependent memory consolidation and reconsolidation. Sleep Med 8:331–343
Thoroughman KA, Shadmehr R (1999) Electromyographic correlates of learning an internal model of reaching movements. J Neurosci 19:8573–8588
Winter DA, Yack HJ (1987) EMG profiles during normal walking: stride-to-stride and inter-subject variability. Electroenceph Clin Neurophysiol 67:402–411
Yang JF, Winter DA (1984) Electromyographic amplitude normalization methods: improving their sensitivity as diagnostic tools in gait analysis. Arch Phys Med Rehab 65:517–521
Ackowledgments
The authors would like to thank subjects for participating in this project, M. Denis Côté for statistical support, M. Simon Lebel for data collection and M. Guy St-Vincent for his technical support. Financial support was provided by an operating grant from the Canadian Institutes of Health Research (CIHR). Karine Fortin and Laurent J. Bouyer were supported by the Fonds de la Recherche en Santé du Québec (FRSQ).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fortin, K., Blanchette, A., McFadyen, B.J. et al. Effects of walking in a force field for varying durations on aftereffects and on next day performance. Exp Brain Res 199, 145–155 (2009). https://doi.org/10.1007/s00221-009-1989-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-009-1989-9