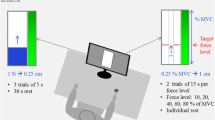
Abstract
The purpose of this investigation was to examine the relationship between strength and the magnitude and time sequential structure of force variability. Young and old adults produced isometric force via index finger abduction to a visually presented target corresponding to a constant force level of 5 or 25% maximal voluntary contraction (MVC). Cluster analysis was used to divide subjects into groups based on age and strength. The variability of older adults was greater and showed more time dependent structure than their younger counterparts. The force output of weaker subjects was also more variable and had a stronger sequential structure. Indeed, when MVC was controlled for there was no significant age effect on force variability. The relationship between strength and variability remained significant, however, when chronological age was controlled for. The findings revealed that the established age-related changes in force variability are more fundamentally due to the association between strength and force variability and provide a further challenge to using chronological age as a marker of the biological aging process in studies of motor control.






Similar content being viewed by others
References
Arking R (1998) Biology of aging. Sinauer Associates, Inc., Sunderland
Austad SN (2001) Concepts and theories of aging. In: EJ Masoro, Austad SN (eds) Handbook of the biology of aging. Academic, Amsterdam, pp 3–18
Barry BK, Carson RG (2004) The consequence of resistance training for movement control in older adults. J Gerontol A Biol Sci 59:M730–M745
Bosco C, Komi PV (1980) Influence of the aging on the mechanical behavior of leg extensor muscles. E J Appl Physiol 45:209–219
Busse EW (1969) Theories of aging. In: EW Busse, E Pfeiffer (eds) Behavior and adaptation in late life. Little, Brown, Boston, pp 11–31
Christou EA, Carlton LG (2001) Old adults exhibit greater motor output variability than young adults only during rapid discrete isometric contractions. J Gerontol A Bio Sci Med Sci 56:B524–B532
Christou EA, Tracy BL (2005) Aging and variability in motor output. In: Davids K, Bennet S, Newell KM (eds) Movement system variability. Human Kinetics, Champaign, pp 199–218
Christou EA, Grossman M, Carlton LG (2002) Modeling variability of force during isometric contractions of the quadriceps femoris. J Mot Behav 34:67–81
De Luca CJ, LeFever RS, McCue MP, Xenakis AP (1982) Control scheme governing concurrently active human motor units during voluntary contractions. J Physiol 329:129–142
Doherty TJ (2003) Invited review: aging and sarcopenia. J Appl Physiol 95:1717–1727
Doherty TJ, Brown WF (1993) The estimated numbers and relative sizes of thenar muscle motor units as selected by multiple point stimulation in young and older adults. Muscle Nerve 16:355–366
Enoka RM, Christou EA, Hunter SK et al (2003) Mechanisms that contribute to differences in motor performance between young and old adults. J Electromyogr Kines 13:1–12
Erim Z, Beg MF, Burke DT, De Luca CJ (1999) Effects of aging on motor-unit control properties. J Neurophysiol 82:2081–2019
Fried LP, Ferrucci L, Darer J, Williamson JD, Anderson G (2004) Untangling the concepts of disability, frailty and comorbidity: Implications for improved targeting and care. J Gerontol Med Sci 59:255–263
Fuglevand AJ, Winter D, Patla AE (1993) Models of recruitment and rate coding organization in motor-unit pools. J Neurophysiol 70:2470–2488
Galganski ME, Fuglevand AJ, Enoka RM (1993) Reduced control of motor output in a human hand muscle of elderly subjects during submaximal contractions. J Neurophysiol 69:2108–2115
Hamliton AFdC, Jones KE, Wolpert DM (2005) The scaling of motor noise with muscle strength and motor unit number. Exp Brain Res 157:417–430
Hughes VA, Frontera WR, Wood M et al (2001) Longitudinal muscle strength changes in older adults: Influence of muscle mass, physical activity, and health. J Gerontol Bio Sci Med Sci 56:B209–B217
Jones KE, Hamilton AFdC, Wolpert DM (2002) Sources of signal-dependent noise during isometric force production. J Neurophysiol 88:533–544
Kallman DA, Plato CC, Tobin JD (1990) The role of muscle loss in the age-related decline in grip strength: Cross-sectional and longitudinal perspectives. J Gerontol Med Sci 45:M82–M88
Keen DA, Yue GH, Enoka RM (1994) Training-related enhancement in the control of motor output in elderly humans. J App Physiol 77:2648–2658
Kornatz KW, Christou EA, Enoka RM (2005) Practice reduces motor unit discharge variability in a hand muscle and improves manual dexterity in old adults. J Appl Physiol 98:2072–2080
Laidlaw DH, Bilodeau M, Enoka RM (2000) Steadiness is reduced and motor unit discharge is more variable in old adults. Muscle Nerve 23:600–612
Lipsitz LA (2002) Dynamics of stability: the physiologic basis of functional health and frailty. J Gerontol A Bio Sci Med Sci 57:B115–B125
Milner-Brown HS, Stein RB, Yemm R (1973) Changes in firing rate of human motor units during linearly changing voluntary contractions. J Physiol (Lond) 230:371–390
Moritz CT, Barry BK, Proscoe MA, Enoka RM (2005) Discharge rate variability influences the variation in force fluctuations across the working range of a hand muscle. J Neurophysiol 93:2449–2459
Petrofsky JS, Burse RL, Lind AR (1975) Comparison of women and men to isometric exercise. J Appl Physiol 38:863–868
Pincus SM (1991) Approximate entropy as a measure of system complexity. Proc Nat Acad Sci 88:2297–2301
Reuter-Lorenz PA (2002) New visions of the aging mind and brain. Tr Cog Sci 6:394–400
Rowe JW, Kahn RL (1987) Human aging: usual and successful. Science 237:143–149
Semmler JG, Nordstrom MA (1998) Motor unit discharge and force tremor in skill-and strength-trained individuals. Exp Brain Res 119:27–38
Shinohara M, Keenen KG, Enoka RM (2005) Fluctuations in motor output during steady contractions are weakly related across contraction types and between hands. Muscle Nerve 31:741–750
Shock NW, Norris AH (1970) Neuromuscular coordination as a factor in age changes in muscular exercise. Med Sport 4:92–99
Slifkin AB, Newell KM (1999) Noise, information transmission and force variability. J Exp Psychol Hum Percep Perf 25:837–851
Slifkin AB, Newell KM (2000) Variability and noise in continuous force production. J Mot Behav 32:141–150
Sosnoff JJ and Newell KM (2006) Aging, visual intermittency and variability in isometric force output. J Gerontol Psychol Sci 61:P117–P124
Taylor AM, Christou EA, Enoka RM (2003) Multiple features of motor-unit activity influence force fluctuations during isometric contractions. J Neurophysiol 90:1350–1361
Tracy BL, Enoka RM (2000) Older adults are less steady during submaximal isometric contractions with the knee extensors muscles. J Appl Physiol 92:1004–2000
Tracy BL, Byrnes WC, Enoka RM (2004) Strength training reduces force fluctuations during anisometric contractions of the quadriceps femoris muscles in old adults. J Appl Physiol 96:1530–1540
Tracy BL, Maluf KS, Stephenson JL, Hunter SK, Enoka RM (2005) Variability of motor unit discharge and force fluctuations across a range of muscle forces in older adults. Muscle Nerve 32:533–540
Vaillancourt DE, Newell KM (2003) Aging and the time and frequency structure of force output variability. J Appl Physiol 94:903–912
Vaillancourt DE, Larsson L, Newell KM (2003) Effects of aging on force variability, single motor unit discharge patterns, and the structure of 10, 20, and Hz EMG activity. Neurobiol Aging 24:25–35
Vandervoort AA, Kramer JF, Wharram ER (1990) Eccentric knee strength of elderly females. J Gerontol Biol Sci 45:B125–B128
Wohlwill JF (1970) The age variable in psychological research. Psych Rev 77:49–64
Yan JH (1999) Tai Chi practice reduces movement force variability for seniors. J Gerontol Med Sci 54:M629–M634
Yao W, Fuglevand RJ, Enoka RM (2000) Motor-unit synchronization increases EMG amplitude and decreases force steadiness of stimulated contractions. J Neurophysiol 83:441–452
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sosnoff, J.J., Newell, K.M. Are age-related increases in force variability due to decrements in strength?. Exp Brain Res 174, 86–94 (2006). https://doi.org/10.1007/s00221-006-0422-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-006-0422-x