Abstract
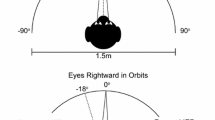
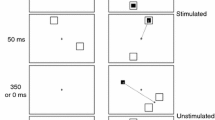
Mammals with foveas (or analogous retinal specializations) frequently shift gaze without moving the head, and their behavior contrasts sharply with “afoveate” mammals, in which eye and head movements are strongly coupled. The ability to move the eyes without moving the head could reflect a gating mechanism that blocks a default eye-head synergy when an attempted head movement would be energetically wasteful. Based upon such considerations of efficiency, we predicted that for saccades to targets lying within the ocular motor range, the tendency to generate a head movement would depend upon a subject’s expectations regarding future directions of gaze. We tested this hypothesis in two experiments with normal human subjects instructed to fixate sequences of lighted targets on a semicircular array. In the target direction experiment, we determined whether subjects were more likely to move the head during a small gaze shift if they expected that they would be momentarily required to make a second, larger shift in the same direction. Adding the onward-directed target increased significantly the distribution of final head positions (customary head orientation range, CHOR) observed during fixation of the primary target from 16.6±4.9° to 25.2±7.8°. The difference reflected an increase in the probability, and possibly the amplitude, of head movements. In the target duration experiment, we determined whether head movements were potentiated when subjects expected that gaze would be held in the vicinity of the target for a longer period of time. Prolonging fixation increased CHOR significantly from 53.7±18.8° to 63.2±15.9°. Larger head movements were evoked for any given target eccentricity, due to a narrowing in the gap between the x-intercepts of the head amplitude:target eccentricity relationship. The results are consistent with the idea that foveate mammals use knowledge of future gaze direction to influence the coupling of saccadic commands to premotor circuitry of the head. While the circuits ultimately mediating the coupling may lie within the brainstem, our results suggest that the cerebrum plays a supervisory role, since it is a likely seat of expectation regarding target behavior. Eye-head coupling may reflect separate gating and scaling mechanisms, and changes in head movement tendencies may reflect parametric modulation of either mechanism.








Similar content being viewed by others
References
Andre-Deshays C, Revel M, Berthoz A (1991) Eye-head coupling in humans. II. Phasic components. Exp Brain Res 84:359–366
Bard C, Fleury M, Paillard J (1992) Different patterns in aiming accuracy for head-movers and non-head movers. In: Berthoz A, Vidal PP, Graf W (eds) The head-neck sensory motor system. Oxford University Press, New York, pp 582–586
Bizzi E, Kalil R, Morasso P (1972) Two modes of active eye-head coordination in monkeys. Brain Res 40:45–48
Collewijn H (1977) Eye and head movements in freely moving rabbits. J Physiol (Lond) 266:471–498
Corneil BD, Munoz DP (1999) Human eye-head gaze shifts in a distractor task. II. Reduced threshold for initiation of early head movements. J Neurophysiol 82:1406–1421
Corneil BD, Olivier E, Munoz DP (2002a) Neck muscle responses to stimulation of monkey superior colliculus. I. Topography and manipulation of stimulation parameters. J Neurophysiol 88:1980–2002
Corneil BD, Olivier E, Munoz DP (2002b) Neck muscle responses to stimulation of monkey superior colliculus. II. Gaze shift initiation and volitional head movements. J Neurophysiol 88:2000–2018
Freedman E (2001) Interactions between eye and head control signals can account for movement kinematics. Biol Cybern 84:453–462
Freedman EG, Sparks DL (1997) Activity of cells in the deeper layers of the superior colliculus of the rhesus monkey: Evidence for a gaze displacement command. J Neurophysiol 78:1669–1690
Freedman EG, Stanford TR, Sparks DL (1996) Combined eye-head gaze shifts produced by electrical stimulation of the superior colliculus in rhesus monkeys. J Neurophysiol 76:927–952
Fuller J (1980) Linkage of eye and head movements in the alert rabbit. Brain Res 194:219–222
Fuller JH (1981) Eye and head movements during vestibular stimulation in the alert rabbit. Brain Res 205:363–381
Fuller JH (1992a) Comparison of head movement strategies among mammals. In: Berthoz A, Graf W, Vidal PP (eds) The head-neck sensory motor system. Oxford University Press, New York, pp 101–112
Fuller JH (1992b) Head movement propensity. Exp Brain Res 92:152–164
Galiana H, Guitton D (1992) Central organization and modeling of eye-head coordination during orienting gaze shifts. Ann NY Acad Sci 656:452–471
Gilchrist I, Brown V, Findlay J (1997) Saccades without eye movements. Nature 390:130–131
Goossens H, Van Opstal A (1997) Human eye-head coordination in two dimensions under different sensorimotor conditions. Exp Brain Res 114:542–560
Gresty M (1975) Eye, head and body movements of the guinea pig in response to optokinetic stimulation and sinusoidal oscillation in yaw. Pflugers Arch 353:201–214
Guitton D, Munoz DP, Galiana HL (1990) Gaze control in the cat: studies and modeling of the coupling between orienting eye and head movements in different behavioral tasks. J Neurophysiol 64:509–531
Hollands M, Patla A, Vickers J (2002) “Look where you’re going!”: gaze behaviour associated with maintaining and changing the direction of locomotion. Exp Brain Res 143:221–230
Isa T, Sasaki S (2002) Brainstem control of head movements during orienting; organization of the premotor circuits. Prog Neurobiol 66:205–241
Isoda M, Tanji AJ (2002) Cellular activity in the supplementary eye field during sequential performance of multiple saccades. J Neurophysiol 88:3541–3545
Kowler E, Pizlo Z, Zhu G, Erkelens C, Steinman R, Collewijn H (1992) Coordination of head and eyes during the performance of natural (and unnatural) visual tasks. In: Berthoz A, Graf W, Vidal P (eds) The Head-Neck Sensory Motor System. Oxford University Press, Oxford
Land M (1992) Predictable eye-head coordination during driving. Nature 359:318–320
Laurutis V, Robinson D (1986) The vestibulo-ocular reflex during human saccadic eye movements. J Physiol 373:209–233
Lee C (1999) Eye and head coordination in reading: roles of head movement and cognitive control. Vision Res 39:3761–3768
Martinez-Trujillo J, Wang H, Crawford D (2003) Electrical stimulation of the supplementary eye fields in the head-free macaque evokes kinematically normal gaze shifts. J Neurophysiol 89:2961–2974
Pelz J, Hayhoe M, Loeber R (2001) The coordination of eye, head, and hand movements in a natural task. Exp Brain Res 139:266–277
Phillips JO, Ling L, Fuchs AF, Siebold C, Plorde JJ (1995) Rapid horizontal gaze movement in the monkey. J Neurophysiol 73:1632–1652
Pierrot-Deseilligny C, Ploner C, Muri R, Gaymard B, Rivaud-Pechoux S (2002) Effects of cortical lesions on saccadic eye movements in humans. Ann NY Acad Sci 956:216–229
Robinson F, Phillips J, Fuchs A (1994) Coordination of gaze shifts in primates: brainstem inputs to neck and extraocular motoneuron pools. J Comp Neurol 346:43–62
Ron S, Berthoz A, Gur S (1994) Model of coupled or dissociated eye-head coordination. J Vestib Res 4:383–390
Roucoux A, Guitton D, Crommelinck M (1980) Stimulation of the superior colliculus in the alert cat. II. Eye and head movements evoked when the head is unrestrained. Exp Brain Res 39:75–85
Silverman B (1986) Density Estimation for Statistics and Data Analysis. Chapman and Hall, New York
Smeets J, Hayhoe M, Ballard D (1996) Goal-directed arm movements change eye-head coordination. Exp Brain Res 109:434–440
Sparks D, Freedman E, Chen L, Gandhi N (2001) Cortical and subcortical contributions to coordinated eye and head movements. Vision Res 41:3295–3305
Stahl J (1999) Amplitude of human head movements associated with horizontal saccades. Exp Brain Res 126:41–54
Stahl J (2000) Plasticity of head movement tendencies. Soc Neurosci Abstr 26:8.7
Stahl J (2001a) Eye-head coordination and the variation of eye movement accuracy with orbital eccentricity. Exp Brain Res 136:200–210
Stahl J (2001b) Knowledge of future gaze direction influences probability of saccade-associated head movements. Soc Neurosci Abstr 27:405.420
Stahl J (2002) Knowledge of future target position influences saccade-associated head movements. Ann NY Acad Sci 956:418–420
Steinman R, Kowler E, Collewijn H (1990) New directions for oculomotor research. Vision Res 30:1845–1864
Tomlinson R (1990) Combined eye-head gaze shifts in the primate. III. Contributions to the accuracy of gaze saccades. J Neurophysiol 64:1873–1891
Tu TA, Keating GE (2000) Electrical stimulation of the frontal eye field in a monkey produces combined eye and head movements. J Neurophysiol 84:1103–1106
Tweed D, Glenn B, Vilis T (1995) Eye-head coordination during large gaze shifts. J Neurophysiol 73:766–779
van Alphen A, Stahl J, Koekkoek S, De Zeeuw C (2001) The dynamic characteristics of the mouse vestibulo-ocular and optokinetic response. Brain Res 890:296–305
Zangemeister W, Stark L (1982a) Types of gaze movement: Variable interactions of eye and head movements. Exp Neurol 77:563–577
Zangemeister WH, Stark L (1982b) Gaze latency: Variable interactions of head and eye latency. Exp Neurol 75:389–406
Acknowledgements
This work was supported by a grant from the Department of Veterans Affairs.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oommen, B.S., Smith, R.M. & Stahl, J.S. The influence of future gaze orientation upon eye-head coupling during saccades. Exp Brain Res 155, 9–18 (2004). https://doi.org/10.1007/s00221-003-1694-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-003-1694-z