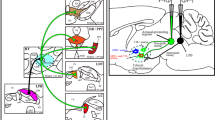
Abstract
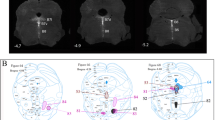
Previous studies have shown that: (1) raphe neurons respond to acoustic and vestibular stimuli, some with a latency of 10–15 ms; (2) alterations of the raphe nuclei alter the acoustic startle reflex; (3) the dorsal raphe nucleus (DRN) is the major source of serotonergic neurons; and (4) approximately 57% of the DRN neurons are nonserotonergic. In the present study, cholera toxin subunit-B (CTB) was injected into cat cochleas, and the brain tissue was examined after a survival period of 5–7 days. Aside from neurons which were known to project to the inner ear, i.e., olivocochlear and vestibular efferent neurons, a surprising new finding was made that somata of a subpopulation of DRN neurons were intensely labeled with CTB. These CTB-labeled neurons were densely distributed in a dorsomedian part of the DRN with some in a surrounding area outside the DRN. The present results suggest that a novel raphe-labyrinthine projection may exist. A future study of anterograde labeling with injections of a tracer in the DRN will be needed to establish the existence of a raphe-labyrinthine projection more thoroughly. A raphe-labyrinthine descending input, together with an ascending input from the inner ear to the DRN through intervening neurons, such as the juxta-acousticofloccular raphe neurons (JAFRNs) described by Ye and Kim, may mediate a brain stem reflex whereby a salient multisensory (including auditory and vestibular) stimulus may alter the sensitivity of the inner ear. As a mammal responds to a biologically important auditory-vestibular multisensory event, the raphe projections to the inner ear and other auditory and vestibular structures may enhance the mammal's ability to localize and recognize the sound and respond properly. The raphe-labyrinthine projection may also modulate the inner ear's sensitivity as a function of the sleep–wake arousal state of an organism on a slower time course.



Similar content being viewed by others
References
Adams JC, Warr WB (1976) Origins of axons in the cat's acoustic striae determined by injection of horseradish peroxidase into severed tracts. J Comp Neurol 170:107–122
Allen GF, Cechetto DF (1994) Serotoninergic and nonserotoninergic neurons in the medullary raphe system have axon collateral projections to autonomic and somatic cell groups in the medulla and spinal cord. J Comp Neurol 350:357-366
Arnesen AR, Osen KK (1984) Fibre population of the vestibulocochlear anastomosis in the cat. Acta Otolaryngol (Stockh) 98:255–269
Basbaum AI, Fields HL (1984) Endogenous pain control systems: brainstem spinal pathways and endorphin circuitry. Ann Rev Neurosci 7:309–338
Brodal A (1981) Neurological anatomy in relation to clinical medicine. Oxford University Press, New York
Buttner-Ennever JA, Cohen B, Pause M, Fries W (1988) Raphe nucleus of the pons containing omnipause neurons of the oculomotor system in the monkey, and its homologue in man. J Comp Neurol 267:307-321
Campbell MJ, Lewis DA, Foote SL, Morrison JH (1987) Distribution of choline acetyltransferase-, serotonin-, dopamine beta-hydroxylase-, tyrosine hydroxylase-immunoreactive axons in monkey primary auditory cortex. J Comp Neurol 261:209-220
Charara A, Parent A (1998) Chemoarchitecture of the primate dorsal raphe nucleus. J Chem Neuroanat 72:111–127
Dahlstrom A, Fuxe K (1964) Evidence for the existence of monoamine-containing neurons in the central nervous system. I. Demonstration of monoamines in the cell bodies of the brain stem neurons. Acta Physiol Scand (Suppl 232) 62:1–55
Davis M, Sheard MH (1974) Habituation and sensitization of the rat startle response: effect of raphe lesions. Physiol Behav 12:425–431
Davis M, Astrachan DI, Kass E (1980) Excitatory and inhibitory effects of serotonin on sensorimotor reactivity measured with acoustic startle. Science 209:521–523
Dechesne C, Raymond J, Sans A (1984) The efferent vestibular system in the cat: a horseradish peroxidase and fluorescent retrograde tracers study. Neuroscience 11:893–901
Ebert U, Ostwald J (1992) Serotonin modulates auditory information processing in the cochlear nucleus of the rat. Neurosci Lett 145:51–54
Fields HL, Heinricher MM, Mason P (1991) Neurotransmitters in nociceptive modulatory circuits. Annu Rev Neurosci 14:219–245
Gil-Loyzaga P, Bartolome MV, Vicente-Torres MA (1997) Serotonergic innervation of the organ of Corti of the cat cochlea. Neuroreport 8:3519–3522
Heym J, Trulson ME, Jacobs BL (1982) Raphe unit activity in freely moving cats: effects of phasic auditory and visual stimuli. Brain Res 232:29–39
Jacobs BL, Azmitia EC (1992) Structure and function of the brain serotonin system. Physiol Rev 72:165–229
Kalen P, Karlson M, Wiklund L (1985) Possible excitatory amino acid afferents to nucleus raphe dorsalis of the rat investigated with retrograde wheat germ agglutinin and d-[3H]aspartate tracing. Brain Res 360:285–297
Kawasaki T, Sato Y (1981) Afferent projections to the caudal part of the dorsal nucleus of the raphe in cats. Brain Res 211:439–444
Kishimoto T, Sasa M, Takaori S (1991) Inhibition of lateral vestibular nucleus neurons by 5-hydroxytryptamine derived from the dorsal raphe nucleus. Brain Res 553:229–237
Klepper A, Herbert H (1991) Distribution and origin of noradrenergic and serotonergic axons in the cochlear nucleus and inferior colliculus of the rat. Brain Res 557:190–201
Le Moal M, Olds M (1979) Unit responses to auditory input in the dorsal and median raphe nuclei of the rat. Physiol Behav 22:11–15
Leung CG, Mason P (1996) Spectral analysis of arterial blood pressure and raphe magnus neuronal activity in anesthetized rats. Am J Physiol 271:483-489
Leung CG, Mason P (1999) Physiological properties of raphe magnus neurons during sleep and waking. J Neurophysiol 81:584-595
Licata F, Li Volsi G, Maugeri G, Sananagelo F (1995) Neuronal responses in vestibular nuclei to dorsal raphe electrical activation. J Vest Res 5:137–145
Li YQ, Takada M, Mizuno N (1993) The sites of origin of serotoninergic afferent fibers in the trigeminal motor, facial and hypoglossal nuclei in the rat. Neurosci Res 17:307-313
Marriage J, Barnes NM (1995) Is central hyperacusis a symptom of 5-hydroxytryptamine (5-HT) dysfunction? J Laryngol Otol 109:915–921
Marsh RA, Fuzzessery ZM, Grose CD, Wenstrup JJ (2002) Projection to the inferior colliculus from the basal nucleus of the amygdala. J Neurosci 22:10449–10460
Morin LP, Meyer-Bernstein EL (1999) The ascending serotonergic system in the hamster: comparison with projections of the dorsal and median raphe nuclei. Neuroscience 91:81-105
Nieuwenhuys R (1985) Chemoarchitecture of the brain. Springer, Berlin
Puizillout JJ, Gaudin-Chazal G, Sayadi A, Vigier D (1981) Serotoninergic mechanisms and sleep. J Physiol (Paris) 77:415-424
Rasmussen K, Strecker R, Jacobs BL (1986) Single unit response of noradrenergic, serotonergic and dopaminergic neurons in freely moving cats to simple sensory stimuli. Brain Res 369:336–340
Rasmussen K, Kallman MJ, Helton DR (1997) Serotonin-1A antagonists attenuate the effect of nicotine withdrawal on the auditory startle response. Synapse 27:145–152
Rigdon GC, Weatherspoon JK (1992) 5-Hydroxytryptamine-1a receptor agonists block repulse inhibition of acoustic startle reflex. J Pharmacol Exp Ther 263:486–493
Simpson KL, Fisher TM, Waterhouse BD, Lin RC (1998) Projection patterns from the raphe nuclear complex to the ependymal wall of the ventricular system in the rat. J Comp Neurol 399:61-72
Steinbusch HWM (1981) Distribution of serotonin-immunoreactivity in the central nervous system of the rat—cell bodies and terminals. Neuroscience 6:557–618
Taber E, Brodal A, Walberg F (1960) The raphe nuclei of the brain stem in the cat. I. Normal topography and cytoarchitecture and general discussion. J Comp Neurol 114:161–187
Thompson AM, Thompson GC (1995) Light microscopic evidence of serotoninergic projections to olivocochlear neurons in the bushy baby (Otolemur garnettii). Brain Res 695:263–266
Thompson GC, Thompson AM, Garret KA, Britton BH (1994) Serotonin and serotonin receptors in the central auditory system. Otolaryngol Head Neck Surg 110:93–102
Thompson AM, Moore KR, Thompson GC (1995) Distribution and origin of serotoninergic afferents to guinea pig cochlear nucleus. J Comp Neurol 351:104–116
Thompson AM, Thompson GC, Britton BH (1998) Serotoninergic innervation of stapedial and tensor tympani motoneurons. Brain Res 787:175–178
Trojanowski JQ, Gonatas JO, Gonatas NK (1982) Horseradish peroxidase (HRP) conjugates of cholera toxin and lectins are more sensitive retrogradely transported markers than the free HRP. Brain Res 231:33–50
Trulson ME, Jacobs BL (1979) Raphe unit activity in free moving cats: correlation with level of behavioral arousal. Brian Res 163:135–150
van de Kar LD, Lorens SA (1979) Differential serotonergic innervation of individual hypothalamic nuclei and other forebrain regions by the dorsal and median midbrain raphe nuclei. Brain Res162:45-54
Vertes PR (1991) A PHA-L analysis of ascending projections of the dorsal raphe nucleus in the rat. J Comp Neurol 313:643–668
Vetter DE, Mugnaini E (1992) Distribution and dendritic features of three groups of rat olivocochlear neurons. A study with two retrograde cholera toxin tracers. Anat Embryol (Berl) 185:1-16
Villar MJ, Vitale ML, Hokfelt T, Verhofstad AA (1988) Dorsal raphe serotoninergic branching neurons projecting both to the lateral geniculate body and superior colliculus: a combine retrograde tracing-immunohistochemical study in the rat. J Comp Neurol 277:126–140
Wan XC, Trojanowski JQ, Gonatas JO (1982) Cholera toxin and wheat germ agglutinin conjugates as neuroanatomical probes: their uptake and clearance, transganglionic and retrograde transport and sensitivity. Brain Res 243:215-224
Warr WB (1975) Olivocochlear and vestibular efferent neurons of the feline brain stem: their location, morphology and number determined by retrograde axonal transport and acetylcholinesterase histochemistry. J Comp Neurol 161:159–181
Warr WB (1992) Organization of olivocochlear efferent systems in mammals. In: Webster DB, Popper AN, Fay RR (eds) The mammalian auditory pathway: neuroanatomy. Springer, New York, pp 410–448
Warr WB, Beck-Boche JE, Ye Y, Kim DO (2002) Organization of olivocochlear neurons in the cat studied with the retrograde tracer cholera toxin-B. J Assoc Res Otolaryngol 3:457–478
Warren EH, Liberman MC (1989) Effects of contralateral sound of auditory-nerve responses. I. Contributions of cochlear efferents. Hearing Res 37:89–104
Waterhouse BD, Border B, Wahl L (1993) Topographic organization of rat locus coeruleus and dorsal raphe nuclei: distribution of cells projecting to visual system structure. J Comp Neurol 336:345–361
White SR, Fung SJ (1989) Serotonin depolarizes cat spinal motoneurons in situ and decreases motoneuron after-hyperpolarizing potentials. Brain Res 502:205–213
Wiklund L, Leger L, Persson M (1981) Monoamine cell distribution in the cat brain stem. A fluorescence histochemical study with quantification of indolaminergic and locus coeruleus cell groups. J Comp Neurol 203:613–647
Willard FH, Ho RH, Martin GF (1984) The neuronal types and the distribution of 5-hydroxytryptamine and enkephalin-like immunoreactive fibers in the dorsal cochlear nucleus of the North American opossum. Brain Res Bull 12:253–266
Yates BJ, Goto T, Bolton PS (1992) Responses of neurons in the caudal medullary raphe nuclei of the cat to stimulation of the vestibular nerve. Exp Brain Res 89:323–332
Yates BJ, Goto T, Kerman I, Bolton PS (1993) Responses of caudal medullary raphe nuclei neurons to natural vestibular stimulation. J Neurophysiol 70:938–946
Ye Y, Kim DO (2001) Connections between the dorsal raphe nucleus and a hindbrain region consisting of the cochlear nucleus and neighboring structures. Acta Otolaryngol 121:284–288
Ye Y, Machado DG, Kim DO (2000) Projection of the marginal shell of the anteroventral cochlear nucleus to olivocochlear neurons in the cat. J Comp Neurol 420:127-138
Acknowledgements
This study was supported by NIDCD-NIH grant DC00360. We thank D. Machado for making comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, D.O., Yang, X.M. & Ye, Y. A subpopulation of dorsal raphe nucleus neurons retrogradely labeled with cholera toxin-B injected into the inner ear. Exp Brain Res 153, 514–521 (2003). https://doi.org/10.1007/s00221-003-1617-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-003-1617-z