Abstract
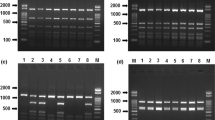
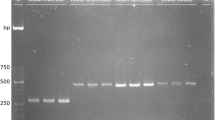
In recent decades, the authentication of fishery products has relied mainly on DNA analysis of mitochondrial genes; however, these methods cannot distinguish hybrids from their respective maternal species. As an alternative for wild or farmed hybrid fish authentication, we developed assays based on the amplification of a parvalbumin intron by exon-primed intron-crossing PCR. Parvalbumins, the major class of fish allergens, are encoded by nuclear genes and are present in high concentrations in the light muscle of many fish species. Amplicons of analysed fish species were characterized by sequencing (tunas), single strand conformation polymorphism (SSCP) (scombrid, catfish, tilapia, and snapper species) and restriction fragment length polymorphism (RFLP) (catfish) analyses. The SSCP method differentiated catfish, tilapia, snapper and scombrid species except tunas. Tunas of the genus Thunnus had an unexpected low variability of intron sequences, which prevented their differentiation by sequencing or SSCP. For study of hybrid catfish, RFLP analysis with Ban I endonuclease was used to construct specific DNA fragment profiles. Isoelectric focusing (IEF) of sarcoplasmic proteins was a rapid screening method to identify catfish, tilapia and snapper species because of their specific protein patterns. The heat-stable, anodic protein bands of these patterns presumably belong to parvalbumins, the major class of fish allergens. PCR and IEF techniques for analysing parvalbumins can be used as routine methods to control the labelling of fish products with the exception of tuna products.





Similar content being viewed by others
References
FAO (2012) The state of world fisheries and aquaculture. FAO, Rome
D’Amico P, Armani A, Castigliego L, Sheng G, Gianfaldoni G, Guidi A (2014) Seafood traceability issues in Chinese food business activities in the light of European provisions. Food Control 35:7–13
Jacquet JL, Pauly D (2008) Trade secrets: renaming and mislabeling of seafood. Mar Policy 32:309–318
Galal-Khallaf A, Ardura A, Mohammed-Geba K, Borrell YJ, Garcia-Vazquez E (2014) DNA barcoding reveals a high level of mislabeling in Egyptian fish fillets. Food Control 46:441–445
Hanner R, Becker S, Ivanova N, Steinke D (2011) FISH-BOL and seafood identification: geographically dispersed case studies reveal systemic market substitution across Canada. Mitochondrial DNA 22(Suppl 1):106–122
Na-Nakorn U, Brummett RE (2009) Use and exchange of aquatic genetic resources for food and aquaculture: Clarias catfish. Rev Aquac 1:214–223
Bartley D, Rana K, Immink A (2001) The use of inter-specific hybrids in aquaculture and fisheries. Rev Fish Biol Fish 10:325–337
Hashimoto DT, do Prado FD, Senhorini JA, Foresti F, Porto-Foresti F (2013) Detection of post-F1 hybrids in broodstock using molecular markers: approaches for genetic management in aquaculture. Aquac Res 44:876–884
Scribner KT, Page KS, Bartron ML (2001) Hybridization in freshwater fishes: a review of case studies and cytonuclear methods of biological inference. Rev Fish Biol Fish 10:293–323
Ardura A, Planes S, Garzia-Vazquez E (2013) Applications of DNA barcoding to fish landings: authentication and diversity assessment. ZooKeys 365:49–65
Barbuto M, Galimberti A, Ferri E, Labra M, Malandra R, Galli P, Casiraghi M (2010) DNA barcoding reveals fraudulent substitutions in shark seafood products: the Italian case of “palombo” (Mustelus spp.). Food Res Int 43:376–381
Hellberg RS, Morrissey MT (2011) Advances in DNA-based techniques for the detection of seafood species substitution on the commercial market. J Lab Autom 16:308–321
Handy SM, Deeds JR, Ivanova NV, Hebert PD, Hanner RH, Ormos A, Weigt LA, Moore MM, Yancy HF (2011) A single-laboratory validated method for the generation of DNA barcodes for the identification of fish for regulatory compliance. J AOAC Int 94:201–210
Galimberti A, De Mattia F, Losa A, Bruni I, Federici S, Casiraghi M, Martellos S, Labra M (2013) DNA barcoding as a new tool for food traceability. Food Res Int 50:55–63
Wong EH-K, Hanner RH (2008) DNA barcoding detects market substitution in North American seafood. Food Res Int 41:828–837
Lago FC, Alonso M, Vieites JM, Espiñeira M (2014) Fish and seafood authenticity-species identification. In: Boziaris IS (ed) Seafood processing: technology, quality and safety. Wiley Blackwell, Chichester, pp 419–452
Altinelataman C, Kündiger R, Cakli S, Rehbein H (2009) Comparison of IEF patterns of sarcoplasmic proteins of fish from North Atlantic and Aegean Sea. Food Control 20:980–985
Berrini A, Tepedino V, Borromeo V, Secchi C (2006) Identification of freshwater fish commercially labelled “perch” by isoelectric focusing and two-dimensional electrophoresis. Food Chem 96:163–168
Tepedino V, Ababouch M, Ferri M, Berrini A (2008) Commercial fish species identification with isoelectric focusing: application to breaded fish products, vol 904. FAO Fisheries and Aquaculture Report, Rome, pp 115–118
Schiefenhövel K, Rehbein H (2013) Differentiation of Sparidae species by DNA sequence analysis, PCR-SSCP and IEF of sarcoplasmic proteins. Food Chem 138:154–160
Griffiths AM, Sotelo CG, Mendes R, Pérez-Martín RI, Schröder U, Shorten M, Silva HA, Verrez-Bagnis V, Mariani S (2014) Current methods for seafood authenticity testing in Europe: is there a need for harmonisation? Food Control 45:95–100
Carrera M, Cañas B, Piñeiro C, Vázquez J, Gallardo JM (2006) Identification of commercial hake and grenadier species by proteomic analysis of the parvalbumin fraction. Proteomics 6:5278–5287
Attardi G (1985) Animal mitochondrial DNA: an extreme example of genetic economy. Int Rev Cytol 93:93–145
Civera T (2003) Species identification and safety of fish products. Vet Res Commun 27:481–489
Avise JC (2009) Phylogeography: retrospect and prospect. J Biogeogr 36(1):3–15
Bottero MT, Dalmasso A (2011) Animal species identification in food products: evolution of biomolecular methods. Vet J 190(1):34–38
Antoniou A, Magoulas A (2014) Application of mitochondrial DNA in stock identification. Stock identification methods, 2nd edn. Academic Press, San Diego, pp 257–295
Rehbein H (2013) Differentiation of fish species by PCR-based DNA analysis of nuclear genes. Eur Food Res Technol 236:979–990
Abdullah A, Rehbein H (2014) Authentication of raw and processed tuna from Indonesian markets using DNA barcoding, nuclear gene and characater-based approach. Eur Food Res Technol 239:695–706
Cawthorn D, Steinman H, Witthuhn R (2011) Establishment of a mitocondrial DNA sequence database for the identification of fish species commercially available in South Africa. Mol Ecol Resour 11:979–991
Viñas J, Tudela S (2009) A validated methodology for genetic identification of tuna species (genus Thunnus). PLoS ONE 4(10):e7606
Rasmussen RS, Morrissey MT (2008) DNA-based methods for the identification of Commercial Fish and Seafood Species. Compr Rev Food Sci Food Saf 7:280–295
Lopata A, Potter P (2000) Allergy and other adverse reactions to seafood. Allergy Clin Immunol Int 12:271–281
Pascual C, Reche M, Fiandor A, Valbuena T, Cuevas T, Esteban M (2008) Fish allergy in childhood. Pediatr Allergy Immunol 19:573–579
Mills EC, Mackie AR, Burney P, Beyer K, Frewer L, Madsen C et al (2007) The prevalence, cost and basis of food allergy across Europe. Allergy 62(7):717–722
Rencova E, Kostelnikova D, Tremlova B (2013) Detection of allergenic parvalbumin of Atlantic and Pacific herrings in fish products by PCR. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 30:1679–1683
Lim DL, Neo KH, Yi FC, Chua KY, Goh DL, Shek LP, Giam YC, Van Bever HP, Lee BW (2008) Parvalbumin–the major tropical fish allergen. Pediatr Allergy Immunol 19:399–407
Sharp MF, Lopata AL (2014) Fish allergy: in review. Clin Rev Allergy Immunol 46(3):258–271
Nwaru BI, Hickstein L, Panesar SS, Muraro A, Werfel T, Cardona V et al (2014) The epidemiology of food allergy in Europe: a systematic review and meta-analysis. Allergy 69(1):62–75
Liu GM, Huang YY, Cai QF, Weng L, Su WJ, Cao MJ (2010) Effect of in vitro gastrointestinal digestion of freshwater fish parvalbumins [J]. J Fish China 7:020
Elsayed S, Bennich H (1975) The primary structure of allergen M from cod. Scand J Immunol 4:203–208
Lindstrøm C, van Dô T, Hordvik I, Endresen C, Elsayed S (1996) Cloning of two distinct cDNAs encoding parvalbumin, the major allergen of Atlantic salmon (Salmo salar). Scand J Immunol 44:335–344
Swoboda I, Bugajska-Schretter A, Valenta R, Spitzauer S (2002) Recombinant fish parvalbumins: candidates for diagnosis and treatment of fish allergy. Allergy 57(Suppl 72):94–96
Van Do T, Hordvik I, Endresen C, Elsayed S (2003) The major allergen (parvalbumin) of codfish is encoded by at least two isotypic genes: cDNA cloning, expression and antibody binding of the recombinant allergens. Mol Immunol 39:595–602
Carrera M, Cañas B, Gallardo J (2012) Rapid direct detection of the major fish allergen, parvalbumin, by selected MS/MS ion monitoring mass spectrometry. J Proteomics 75:3211–3220
Kuehn A, Swoboda I, Arumugam K, Hilger C, Hentges F (2014) Fish allergens at a glance: variable allergenicity of parvalbumins, the major fish allergens. Front Immunol 5:179
Focant B, Mélot F, Collin S, Chikou A, Vandewalle P, Huriaux F (1999) Muscle parvalbumin isoforms of Clarias gariepinus, Heterobranchus longifilis, and Chrysichthys auratus: isolation characterization and expression during development. J Fish Biol 54:832–851
Arif S, A-u Hasnain (2010) A major cross-reactive fish allergen with exceptional stability: parvalbumin. Afr J Food Sci 4:109–114
Kawase S, Ushio H, Ohshima T, Yamanaka H, Fukuda H (2001) Preparation of monoclonal antibodies against tuna parvalbumin. Fish Sci 67:559–561
Kuehn A, Scheuermann T, Hilger C, Hentges F (2010) Important variations in parvalbumin content in common fish species: a factor possibly contributing to variable allergenicity. Int Arch Allergy Immunol 153:359–366
Lim DL, Neo KH, Goh DL, Shek LP, Lee BW (2005) Missing parvalbumin: implications in diagnostic testing for tuna allergy. J Allergy Clin Immunol 115:874–875
Rosmilah M, Shahnaz M, Meinir J, Masita A, Noormalin A, Jamaluddin M (2013) Identification of parvalbumin and two new thermolabile major allergens of Thunnus tonggol using a proteomics approach. Int Arch Allergy Immunol 162:299–309
Shiomi K, Hamada Y, Sekiguchi K, Shimakura K, Nagashima Y (1999) Two classes of allergens, parvalbumins and higher molecular weight substances, in Japanese Eel and Bigeye Tuna. Fish Sci 65:943–948
Lee SJ, Ju CC, Chu SL, Chien MS, Chan TH, Liao WL (2007) Molecular cloning, expression and phylogenetic analyses of parvalbumin in tilapia, Oreochromis mossambicus. J Exp Zool A Ecol Genet Physiol 307:51–61
Rosmilah M, Shahnaz M, Masita A, Noormalin A, Jamaludin M (2005) Identification of major allergens of two species of local snappers: Lutjanus argentimaculatus (merah/red snapper) and Lutjanus johnii (jenahak/golden snapper). Trop Biomed 22:171–177
Ebo DG, Kuehn A, Bridts CH, Hilger C, Hentges F, Stevens WJ (2010) Monosensitivity to pangasius and tilapia caused by allergens other than parvalbumin. J Investig Allergol Clin Immunol 20:84–88
Rehbein H (2008) New fish on the german market: consumer protection against fraud by identification of species. Journal für Verbraucherschutz und Lebensmittelsicherheit 3:49–53
Manthey M, Hilge V, Rehbein H (1988) Sensory and chemical evaluation of three catfish species (Silurus glanis, Ictalurus punctatus, Clarias gariepinus) from intensive culture. Arch FischWiss 38:215–227
Rehbein H, Näumann G, Stumme B (2011) Differenzierung von Pangasius (Pangasius hypophthalmus) und Asiatischem Rotflossenwels (Hemibagrus wyckioides) durch Protein- und DNA-Analyse [Differentiation between Pangasius (Pangasius hypophthalmus) and Asian Redtail catfish (Hemibagrus wyckioides) by protein-and DANN analysis]. Inf Fischereiforsch 58:13–19
Rehbein H (2011) Identifizierung der Welsart in Filetware durch Protein- und DNA-Analyse [Identification of the catfish species in case of fillets by Protein- and DNA-Analysis]. Inf Fischereiforsch 58:35–41
Carpenter KE, Niem VH (1999). FAO species identification guide for fishery purposes. The living marine resources of the Western Central Pacific. FAO Library
Rehbein H (2005) Identification of the fish species of raw or cold-smoked salmon and salmon caviar by single-strand conformation polymorphism (SSCP) analysis. Eur Food Res Technol 220:625–632
Downs TR, Wilfinger WW (1983) Fluorometric quantification of DNA in cells and tissue. Anal Biochem 131:538–547
Rehbein H, Kreß G (2005) Detection of short mRNA sequences in fishery products. Dtsch Lebensm-Rundsch 101:333–337
Xu YX, Zhu ZY, Lo LC, Wang CM, Lin G, Feng F, Yue GH (2006) Characterization of two parvalbumin genes and their association with growth traits in Asian seabass (Lates calcarifer). Anim Genet 37:266–268
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Schiefenhövel K, Rehbein H (2011) Identification of barramundi (Lates calcarifer) and tilapia (Oreochromis spp.) fillets by DNA- and protein-analytical methods. Journal für Verbraucherschutz und Lebensmittelsicherheit 6:203–214
Kuehn A, Hilger C, Graf T, Hentges F (2012) Protein-and DNA-based assays as complementary tools for fish allergen detection. Allergologie 35(7):343–350
Berrebi P, Retif X, Fang F, Zhang C-G (2006) Population structure and systematics of Opsariichthys bidens (Osteichthyes: Cyprinidae) in south-east China using a new nuclear marker: the introns (EPIC-PCR). Biol J Linn Soc 87:155–166
Hassan M, Lemaire C, Fauvelot C, Bonhomme F (2002) Seventeen new exon-primed intron-crossing polymerase chain reaction amplifiable introns in fish. Mol Ecol Notes 2:334–340
Rehbein H (2007) Differentiation of hake species by RFLP- and SSCP-analysis of PCR amplified cytochrome b and parvalbumin sequences. Dtsch Lebensm-Rundsch 103:511–517
Sriphairoj K, Klinbunga S, Kamonrat W, Na-Nakorn U (2010) Species identification of four economically important Pangasiid catfishes and closely related species using SSCP markers. Aquaculture 308:S47–S50
Wong LL, Peatman E, Lu J, Kucuktas H, He S, Zhou C, Na-nakorn U, Liu Z (2011) DNA barcoding of catfish: species authentication and phylogenetic assessment. PLoS ONE 6:e17812
Warner K, Timme W, Lowell B, Hirshfield M (2013) Oceana study reveals seafood fraud nationwide. Oceana Science Report. http://oceana.org/en Accessed 12 Aug 2015
Wong LL, Peatman E, Kelly L, Kucuktas H, Na-Nakorn U, Liu Z (2014) Catfish species identification using lab-on-chip PCR-RFLP. J Aquat Food Prod Technol 23:2–13
Oberst S, Abban EK, Villwock W (1996) Biochemical and immunological markers for the discrimination of three Tilapia species: T. zillii Gervais, T. guineesis Bleeker and T. dageti Thys v.d. Audenaerde (Pisces: Cichlidae) from West Africa. Aquac Res 27:235–244
Sezaki K, Watabe S, Ochiai Y, Hashimoto K (1994) Biochemical genetic evidence for a hybrid origin of spined loach, Cobitis taenia taenia, in Japan. J Fish Biol 44:683–691
Chen L, Hefle S, Taylor S, Swoboda I, Goodman R (2006) Detecting fish parvalbumin with commercial mouse monoclonal anti-frog parvalbumin IgG. J Agric Food Chem 54:5577–5582
Acknowledgments
The authors thank the Max Rubner-Institute for supporting this research with materials for analysis and laboratory facilities. One of us (A.A) gratefully acknowledges a research Grant from Deutscher Akademischer Austausch Dienst (DAAD), Germany.
Conflict of interest
The authors declare no conflict of interest.
Compliance with Ethics Requirement
This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Abdullah, A., Rehbein, H. Authentication of closely related scombrid, catfish and tilapia species by PCR-based analysis and isoelectric focusing of parvalbumin. Eur Food Res Technol 241, 497–511 (2015). https://doi.org/10.1007/s00217-015-2479-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-015-2479-x