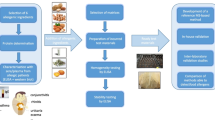
Abstract
In order to support patients’ safety, the analytical methods should be able to quantify the allergenic proteins in food products. Analytical performance of the currently used ELISA methods is not always appropriate, particularly in case of processed foods. A possible way to investigate the sources of analytical errors is the utilisation of model food matrices that mimic the technology and behaviour of real food products. Consequently, factors affecting the analytical results were identified, their contribution to the whole analytical error was determined, and the underlying phenomena were interpreted in this study. Heat-treated model matrices incurred with gliadin, milk, egg, or soy proteins were produced and analysed with commercially available ELISA test kits. The data were evaluated with statistical tools. Results clearly show that the type of the food matrix, the level of processing, and the analytical methods (i.e. the type of the ELISA kit) are the three main factors that play a significant role in the uncertainty. The developed laboratory methodology seems to be suitable for generalising the investigation of other allergens, methods, matrices, and the scientific background of the phenomena.





Similar content being viewed by others
References
Taylor SL, Hefle SL (2001) Food allergies and other food sensitivities. Food Tech 55(9):68–83
Kerbach S, Alldrick AJ, Crevel RWR, Dömötör L, DunnGalvin A, Mills ENC, Pfaff S, Poms RE, Popping B, Tömösközi S (2009) Managing food allergens in the food supply chain—viewed from different stakeholder perspectives. Qual Assur Saf Crop 1:50–60
Sicherer SH, Sampson H (2010) Food allergy. J Allergy Clin Immun 125(2):116–125
Breitender H (2008) Can any protein become an allergen? Rev Fr Allergol 48:135–138
Besler M, Steinhart H, Paschke A (2001) Stability of food allergens and allergenicity of processed foods. J Chromatogr B 756:207–228
Gerrard JA (2002) Protein–protein crosslinking in food: methods, consequences, applications. Trends Food Sci Tech 13:391–399
Sathe SK, Teuber SS, Roux KH (2005) Effects of food processing on the stability of food allergens. Biotechnol Adv 23:423–429
Thomas K, Herouet-Guicheney C, Ladics G, Bannon G, Cockburn A, Crevel R, Fitzpatrick J, Mills C, Privalle L, Vieths S (2007) Evaluating the effect of food processing on the potential human allergenicity of novel proteins: international workshop report. Food Chem Toxicol 45:1116–1122
Gendel SM (2012) Comparison of international food allergen labeling regulations. Regul Toxicol Pharm 63(2):279–285
EU Regulation No 1169/2011 of the European Parliament and of the Council. Off J EU 304:18-63
Taylor SL, Baumert JL, Kruizinga AG, Remington BC, Crevel RWR, Brooke-Taylor S, Allen KJ, Houben G (2014) Establishment of reference doses for residues of allergenic foods: report of the VITAL expert panel. Food Chem Toxicol 63:9–17
Mattarozzi M, Milioli M, Bignardi C, Elviri L, Corradini C, Careri M (2014) Investigation of different sample pre-treatment routes for liquid chromatography tandem mass spectrometry detection of caseins and ovalbumin in fortified red wine. Food Control 38:82–87
Azarnia S, Boye JI, Mongeon V, Sabik H (2013) Detection of ovalbumin in eggwhite, whole egg and incurred pasta using LC–ESI-MS/MS and ELISA. Food Res Int 52:526–534
Heick J, Fischer M, Popping B (2011) First screening method for the simultaneous detection of seven allergens by liquid chromatography mass spectrometry. J Chromatogr A 1218(7):938–943
Köppel R, van Velsen-Zimmerli F, Bucher T (2012) Two quantitative hexaplex real-time PCR systems for the detection and quantification of DNA from twelve allergens in food. Eur Food Res Technol 235(5):843–852
Koeberl M, Clarke D, Lopata AL (2014) Next generation of food allergen quantification using mass spectrometry systems. J Proteome Res 13:3499–3509
Prado M, Boix A, von Holst C (2013) Development of a real-time PCR method for the simultaneous detection of mackerel and horse mackerel. Food Control 34:19–23
Bignardi C, Mattarozzi M, Penna A, Sidoli S, Elviri L, Careri M, Mangia A (2013) A rapid size-exclusion solid-phase extraction step for enhanced sensitivity in multi-allergen determination in dark chocolate and biscuits by liquid chromatography–tandem mass spectrometry. Food Anal Method 6:1144–1152
Johnson PE, Baumgartner S, Aldick T, Bessant C, Giosafatto V, Heick J, Mamone G, O’Connor G, Poms R, Popping B, Reuter A, Ulberth F, Watson A, Monaci L, Mills ENC (2011) Current perspectives and recommendations for the development of mass spectrometry methods for the determination of allergens in foods. J AOAC Int 94(4):1026–1033
Wieser H, Koehler P (2009) Is the calculation of the gluten content by multiplying the prolamin content by a factor of 2 valid? Eur Food Res Technol 229:9–13
Cucu T, Devreese B, Kerkaert B, Rogge M, Vercruysse L, de Meulenaer B (2012) ELISA-based detection of soybean proteins: a comparative study using antibodies against modified and native proteins. Food Anal Method 5:1121–1130
Sharma GM (2012) Immunoreactivity and detection of wheat proteins by commercial ELISA kits. J AOAC Int 95(2):364–371
Bugyi Z, Török K, Hajas L, Adonyi Z, Popping B, Tömösközi S (2013) Comparative study of commercially available gluten ELISA kits using an incurred reference material. Qual Assur Saf Crop 5(1):79–87
van Eckert R, Bond J, Rawson P, Klein CL, Stern M, Jordan TW (2010) Reactivity of gluten detecting monoclonal antibodies to a gliadin reference material. J Cereal Sci 51(2):198–204
Bugyi Z, Nagy J, Török K, Hajas L, Tömösközi S (2010) Towards development of incurred materials for quality assurance purposes in the analysis of food allergens. Anal Chim Acta 672(1–2):25–29
Bugyi Z, Török K, Hajas L, Adonyi Z, Poms RE, Popping B, Diaz-Amigo C, Kerbach S, Tömösközi S (2012) Development of incurred reference material for improving conditions of gluten quantification. J AOAC Int 95(2):382–387
Monaci L, Brohée M, Tregoat V, van Hengel A (2011) Influence of baking time and matrix effects on the detection of milk allergens in cookie model food system by ELISA. Food Chem 127(2):669–675
Platteau C, De Loose M, De Meulenaer B, Taverniers I (2011) Detection of Allergenic Ingredients Using real-time PCR: a case study on hazelnut (Corylus avellena) and soy (Glycine max). J Agric Food Chem 59:10803–10814
Lacorn M, Immer U (2011) Allergen determination in food: reference materials and traceability of results. Accredit Qual Assur 16:449–452
Diaz-Amigo C, Popping B (2013) Accuracy of ELISA detection methods for gluten and reference materials: a realistic assessment. J Agric Food Chem 61(24):5681–5688
Siegel M, Mutschler A, Boernsen B, Pietsch K, Waiblinger H (2013) Food matrix standards for the quantification of allergenic food ingredients using real-time PCR. Eur Food Res Technol 237:185–197
Phillips MM, Sharpless KE, Wise SA (2013) Standard reference materials for food analysis. Anal Bioanal Chem 405:4325–4335
van Eckert R, Berghofer E, Ciclitira PJ, Chirdo F, Denery-Papini S, Ellis HJ, Ferranti P, Goodwin P, Immer U, Mamone G, Mendez E, Mothes T, Novalin S, Osman A, Rumbo M, Stern M, Thorell L, Whim A, Wieser H (2006) Towards a new gliadin reference material–isolation and characterisation. J Cereal Sci 43(3):331–341
Dumont V, Kerbach S, Poms R, Johnson P, Mills C, Popping B, Tömösközi S, Delahaut P (2010) Development of milk and egg incurred reference materials for the validation of food allergen detection methods. Qual Assur Saf Crop 2(4):208–215
Török K, Hajas L, Bugyi Z, Balázs G, Tömösközi S (2014) Investigation of the effects of food processing and matrix components on the analytical results of ELISA using an incurred gliadin reference material candidate. Acta Aliment Hung. doi:10.1556/AAlim.2014.0018
Török K, Horváth V, Horváth Á, Hajas L, Bugyi Z, Tömösközi S (2014) Investigation of incurred single- and multi-component model food matrices for determination of food proteins triggering allergy and coeliac disease. Eur Food Res Technol 239:923–932
Gomaa A, Ribereau S, Boye JI (2012) Detection of allergens in a multiple allergen matrix and study of the impact of thermal processing. J Nutr Food Sci 9:1–6
Khuda S, Slate A, Pereira M, Al-Taher F, Jackson L, Diaz-Amigo C, Bigley EC, Whitaker T, Williams K (2012) Effect of processing on recovery and variability associated with immunochemical analytical methods for multiple allergens in a single matrix: dark chocolate. J Agric Food Chem 60:4204–4211
Khuda S, Slate A, Pereira M, Al-Taher F, Jackson L, Diaz-Amigo C, Bigley EC, Whitaker T, Williams K (2012) Effect of processing on recovery and variability associated with immunochemical analytical methods for multiple allergens in a single matrix: sugar cookies. J Agric Food Chem 60:4195–4203
Acknowledgments
This research is related to the scientific goals of MoniQA Association and the Hungarian national project “Development of quality-orientated, harmonised educational and R + D+I strategy and operational model at the Budapest University of Technology and Economics” (ÚMFT TÁMOP-4.2.1/B-09/1/KMR-2010-0002). This research is also connected to the scientific program of the “Improvement and optimisation of the nutritional value and technological properties of gluten-free products—study on the effect of newly developed food additives and alternative crops” project (TÉT_10-1-2011-0731).
Conflict of interest
The authors declare that they have no conflict of interest.
Compliance with Ethical standards
This article does not contain any studies with human participants or animals performed by any of the authors. Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Török, K., Hajas, L., Horváth, V. et al. Identification of the factors affecting the analytical results of food allergen ELISA methods. Eur Food Res Technol 241, 127–136 (2015). https://doi.org/10.1007/s00217-015-2441-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-015-2441-y