Abstract
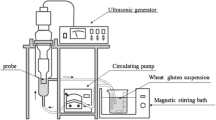
In this study, the effect of sweeping frequency ultrasound (SFU) treatment on the degree of hydrolysis of zein and the angiotensin I-converting enzyme (ACE)-inhibitory activity of its hydrolysates were investigated. The mechanism through which ultrasonic pretreatment releases peptides with ACE-inhibitory activity was also studied by fluorescence, circular dichroism (CD), scanning electron microscopy (SEM), and atomic force microscopy (AFM). Compared to the control, SFU and fixed frequency ultrasound (FFU) increased the degree of zein hydrolysis by approximately 11.5 %. Sweeping frequency ultrasound pretreatment increased ACE-inhibitory activity of zein hydrolysates by 12.3–116.7 % over the control. At 40 ± 2 kHz, SFU-treated zein hydrolysates had 42.9 % ACE-inhibitory activity, representing an increase of 116.7 % over the control. The fluorescence intensity of SFU- and FFU-treated zein was weaker than in untreated zein, indicating that more Phe, Trp, and Tyr residues were exposed outside the polypeptide chains. CD spectra show that SFU treatment resulted in increase in the α-helix content by 3.4 %, and β-sheet, β-turns, and random coils content by 24.4 %. Analyses of microstructure by SEM and AFM revealed that ultrasonic pretreatment ruptured the fine meshwork structure of zein resulting in the appearance of several micro-holes. We conclude that the SFU pretreatment for 40 ± 2 kHz can remarkably raise the degree of zein hydrolysis and ACE-inhibitory activity of the hydrolysates by altering the second structure of zein and rupturing the smooth surface of protein.






Similar content being viewed by others
References
Hernandez-Izquierdo JM, Krochta VM (2008) Thermoplastic processing of proteins for film formation—a review. J Food Sci 73:R30–R39
Liu J, Lee WW (2004) Plasticized prolamine composition. US2004086595A1
Chiue H, Iwami K, Kusano T, Ibuki F (1994) Decreased antioxidative activity of maize zein in response to deamidation rate. Biosci Biotech Biochem 58:198–199
Zhu L, Chen J, Tang X, Xiong Y (2008) Reducing, radical scavenging, and chelation properties of in vitro digests of alcalase-treated zein hydrolysate. J Agric Food Chem 56:2714–2721
Guo H, Shi Y (2009) A novel zein-based dry coating tablet design for zero-order release. Int J Pharm 370:81–86
Hurtado-Lopez P, Murdan S (2005) Formulation and characterization of zein microspheres as delivery vehicles. J Drug Deliv Sci Technol 15:267–272
Liu X, Sun Q, Wang H, Zhang L, Wang J (2005) Microspheres of corn protein, zein, for an ivermectin drug delivery system. Biomaterials 26:109–115
Zhong Q, Jin M (2009) Nanoscalar structures of spray-dried zein microcapsules and in vitro release kinetics of the encapsulated lysozyme as affected by formulations. J Agric Food Chem 57:3886–3894
McGowan BA, Padua GW, Lee SY (2005) Formulation of corn zein chewing gum and evaluation of sensory properties by the time-intensity method. J Food Sci 70:S475–S481
Sessa DJ, Selling GW, Willett JL, Palmquist DE (2006) Viscosity control of zein processing with sodium dodecyl sulfate. Ind Crops Prod 23:15–22
Wang Y, Padua GW (2003) Tensile properties of extruded zein sheets and extrusion blown films. Macromol Mater Eng 288:886–893
Hurtado-Lopez P, Murdan S (2006) Zein microspheres as drug/antigen carriers: a study of their degradation and erosion, in the presence and absence of enzymes. J Microencapsul 23:303–314
Wang H, Gong S, Wang J (2008) Mechanical improvement of zein protein as scaffold for bone tissue engineering. Mater Sci Technol 24:1045–1052
Wang Q, Xian WJ, Li SF, Liu C, Padua GW (2008) Topography and biocompatibility of patterned hydrophobic/hydrophilic zein layers. Acta Biomater 4:844–851
Shukla R, Cheryan M (2001) Zein: the industrial protein from corn. Ind Crop Prod 13:171–192
Ariyoshi Y (1993) Angiotensin-converting enzyme inhibitors derived from food proteins. Trends Food Sci Technol 4:139–144
Rees ED, Singer SJ (1956) A preliminary study of the properties of proteins in some nonaqueous solvents. Arch Biochem Biophys 63:144–159
Jia J, Ma H, Zhao W, Wang Z, Tian W, Luo L, He R (2010) The use of ultrasound for enzymatic preparation of ACE-inhibitory peptides from wheat germ protein. Food Chem 119:336–342
Khanal SK, Grewell D, Sung S, van Leeuwen J (2007) Ultrasound applications in wastewater sludge pretreatment: a review. Crit Rev Environ Sci Technol 37:277–313
Khanal SK, Montalbo M, van Leeuwen J, Srinivasan G, Grewell D (2007) Ultrasound enhanced glucose release from corn in ethanol plants. J Biotech Bioeng 98:978–985
Jambrak AR, Herceg Z, Šubarić D, Babić J, Brnčić M, Brnčić SR, Bosiljkov T, Čvek D, Tripalo B, Gelo J (2010) Ultrasound effect on physical properties of cornstarch. Carbohyd Polym 79:91–100
Koda S, Kimura T, Kondo T, Mitome H (2003) A standard method to calibrate sonochemical efficiency of an individual reaction system. Ultrason Sonochem 10:149–156
Qu W, Ma H, Jia J, He R, Luo L, Pan Z (2012) Enzymolysis kinetics and activities of ACE-inhibitory peptides from wheat germ protein prepared with SFP ultrasound-assisted processing. Ultrason Sonochem 19:1021–1026
Wu QY (2004) Study on preparation and characterization of antihypertensive fermented milk Ph.D. Thesis, Jiangsu University, Jiangsu, China
Adler-Nissen J (1986) Enzymatic hydrolysis of food proteins. Elsevier, London
Vermeirssen V, Van Camp J, Verstraete W (2002) Optimisation and validation of an angiotensin-converting enzyme inhibition assay for the screening of bioactive peptides. J Biochem Biophys Methods 51:75–87
Kelly SM, Jess TJ, Price NC (2005) How to study proteins by circular dichroism. Biochim Biophys Acta 1751:119–139
Lobley A, Whitmore L, Wallace BA (2002) DICHROWEB: an interactive website for the analysis of protein secondary structure from circular dichroism spectra. Bioinformatics 18:211–212
Seerama N, Woody RW (2000) Estimation of protein secondary structure from circular dichroism spectra: comparison of CONTIN, SELCON and DCSSTR methods with expanded reference set. Anal Biochem 287:252–260
Whitmore L, Wallace BA (2007) Protein secondary structure analyses from circular dichroism spectroscopy: methods and reference databases. Biopolymers 89:392–400
Guo Y, Liu Z, An H, Li M, Hu J (2005) Nano-structure and properties of maize zein studied by atomic force microscopy. J Cereal Sci 41:277–281
Huang Q, Li L, Fu X (2007) Ultrasound effects on the structure and chemical reactivity of cornstarch granules. Starch 59:371–378
Selling GW, Hamaker SAH, Sessa DJ (2007) Effect of solvent and temperature on secondary and tertiary structure of zein by circular dichroism. Cereal Chem 84:265–270
Cabra V, Arreguin R, Galvez A, Quirasco M, Vazquez-duhalt R, Farres A (2005) Characterization of a 19 kDa α-zein of high purity. J Agric Food Chem 53:725–729
Cabra V, Arreguin R, Vazquez-duhalt R, Farres A (2006) Effect of temperature and pH on the secondary structure and processes of oligomerization of 19 kDa α-zein. BBA Proteins Proteom 1764:1110–1118
Bugs MR, Forato LA, Bortoleto-Bugs RK, Fischer H, Mascarenhas YP, Ward RJ, Colnago LA (2004) Spectroscopic characterization and structural modeling of prolamin from maize and pearl millet. Eur Biophys J 33:335–343
Greenfield NJ (1999) Applications of circular dichroism in protein and peptide analysis. TrAC Trend Anal Chem 18:236–244
Güzey D, Gülseren İ, Bruce B, Weiss J (2006) Interfacial properties and structural conformation of thermosonicated bovine serum albumin. Food Hydrocolloid 20:669–677
Karki B, Lamsal BP, Jung S, van Leeuwen JH, Pometto AL III, Grewell D, Khanal SK (2010) Enhancing protein and sugar release from defatted soy flakes using ultrasound technology. J Food Eng 96:270–278
Villamiel M, deJong P (2000) Inactivation of Pseudomonas fluorescens and Streptococcus thermophilus in Trypticase® Soy Broth and total bacteria in milk by continuous-flow ultrasonic treatment and conventional heating. J Food Eng 45:171–179
Acknowledgments
The authors wish to extend our appreciation for the support provided by National 863 Plan of China (Hi-Tech Research and Development Program of China) (2013AA100203), the National Natural Science Foundation of China (NNSFC-31071502), the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions, Nature Science Foundation of Jiangsu Provincial Education Department (11KJB550001), and Research-Innovation Program of Postgraduate in General Universities of Jiangsu (CX10B_279Z), China.
Conflict of interest
None.
Compliance with Ethics Requirements
This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ren, X., Ma, H., Mao, S. et al. Effects of sweeping frequency ultrasound treatment on enzymatic preparations of ACE-inhibitory peptides from zein. Eur Food Res Technol 238, 435–442 (2014). https://doi.org/10.1007/s00217-013-2118-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-013-2118-3