Abstract
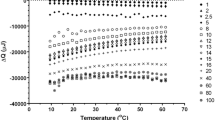
Interaction forces that caused the formation of amylose recrystallite and led to the enthalpy change of the recrystallite were studied. Results obtained from differential scanning calorimetry (DSC) in combination with molecular simulation showed that the amylose recrystallite was formed and the volume of amylose fractions decreased as the short-term retrogradation occurred. This volume decrease indicated that a disordered conformation of amylose fractions was transformed to an ordered one. It was further confirmed that the formation of ordered configuration and the enthalpy change that occurred in DSC measurement for amylose recrystallite were related to a decrease in bonded interaction energy and an increase in Van der Waals attraction and hydrogen bonds formed.


Similar content being viewed by others
References
Khanna S, Tester RF (2006) Food Hydrocoll 5:567–576
Miles MJ, Morris VJ, Orford PD, Ring SG (1985) Carbohydr Res 135:271–281
Ring SR, Colonna P, I’Anson KJ, Kalichevsky MT, Miles MJ, Morris VJ, Orford PD (1987) Carbohydr Res 162:277–293
Gudmundsson M, Eliasson AC (1990) Carbohydr Polym 13:295–315
Eerlingen RC, Crombez M, Delcour JA (1993) Cereal Chem 70:339–344
Tsuge H, Hishida M, Iwasaki H, Watanabe S, Goshima G (1990) Starch/Stärke 42:213–216
Miles MJ, Morris VJ, Ring SG (1985) Carbohydr Res 135:257–269
Smits ALM, Ruhnau FC, Vliegenthart JFG, van Soest JJG (1998) Starch/Stärke 50:478–483
Silverio J, Svensson E, Eliasson AC, Olofsson G (1996) J Therm Anal 47:1179–1200
Eerlingen RC, Jacobs H, Delcour JA (1994) Cereal Chem 71:351–355
Russell PL (1987) J Cereal Sci 6:147–158
Shamai K, Bianco-Peled H, Shimoni E (2003) Carbohydr Polym 54:363–369
Richardson G, Kidman S, Langton M, Hermansson AM (2004) Carbohydr Polym 58:7–13
Slade L, Levine H (1987) In: Stivala SS, Crescenzi V, Dea ICM (eds) Recent developments in industrial polysaccharides. Gorton and Breach Science, New York, pp 387–430
Jacobson MR, BeMiller JN (1998) Cereal Chem 75:22–29
Jang JK, Pyun YR (1997) Starch/Stärke 49:272–277
Longton J, LeGrys GA (1981) Starch/Stärke 33:410–414
Ogura I, Yamamoto T (1995) Polymer 36:1375–1381
Yousef FO, Zughul MB, Badwan AA (2007) J Incl Phenom Macro Chem 57:519–523
Farmer BL, Chapman BR, Dudis DS, Adams WW (1993) Polymer 34:1588–1601
Xie YH, Soh AK (2005) Mater Lett 59:971–975
Tang MC, Copeland L (2007) Carbohydr Polym 70:1–7
Acknowledgments
This study was financially supported by Program of International Technology Cooperation (No. 2007DFA31120), Key Program of Ministry Education (No. 109080), Programs of State Key Laboratory of Food Science and Technology, Jiangnan University (Nos. SKLF-TS-200810 and SKLF-MB-200804), and Nature Science Foundation of Jiangsu Province (BK2008003).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Tian, Y., Li, Y., Jin, Z. et al. A novel molecular simulation method for evaluating the endothermic transition of amylose recrystallite. Eur Food Res Technol 229, 853–858 (2009). https://doi.org/10.1007/s00217-009-1124-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-009-1124-y