Abstract
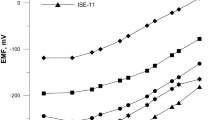
A PVC membrane electrode for copper(II) ions based on a recently synthesized naphthol-derivative Schiff’s base as membrane carrier was prepared. The sensor exhibits a Nernstian response for Cu2+ ions over a wide concentration range (5.0 × 10–6–5.0 × 10–2 mol/L) with a detection limit of 3.1 × 10–6 mol/L (0.2 μg/mL). It has a very short response time of about 5 s and can be used for ¶3 months without any divergence in potential. The proposed electrode revealed good selectivities over a wide variety of other cations including alkali, alkaline earth, transition and heavy metal ions and could be used in a pH range of 4.0–7.0. It was successfully applied to the direct determination and potentiometric titration of copper ion.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 23 February 1999 / Revised: 4 May 1999 / Accepted: 5 May 1999
Rights and permissions
About this article
Cite this article
Alizadeh, N., Ershad, S., Naeimi, H. et al. Copper(II)-selective membrane electrode based on a recently synthesized naphthol-derivative Schiff’s base. Fresenius J Anal Chem 365, 511–515 (1999). https://doi.org/10.1007/s002160051514
Issue Date:
DOI: https://doi.org/10.1007/s002160051514