Abstract
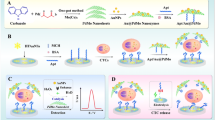
In this work, we developed a photoelectrochemical assay for circulating tumor cells (CTCs) detection based on hexagonal carbon-nitrogen tubes (HCNT) as visible light-sensitive materials. The MCF-7 cell was selected as the model CTC and was captured through specific recognition between epithelial cell adhesion molecules (EpCAM) on the cell surface and anti-EpCAM antibodies. Anti-EpCAM antibody-modified magnetic nanoparticles were used to enrich and separate MCF-7 cells from samples. The detection signal was amplified by Ag2S nanocrystals, which can compete with HCNTs for absorbing visible light, leading to a decrease of photocurrent intensity. The linear range of the assay for MCF-7 cells is from 10 to 5000 cells mL−1, with a detection limit of 3 cells mL−1 (S/D = 3). The assay has good selectivity for MCF-7 detection over HeLa cells. The assay was successfully applied for the detection of MCF-7 in human whole blood, which indicates the potential for clinical application.
Graphical abstract






Similar content being viewed by others
References
Haber DA, Velculescu VE. Blood-based analyses of cancer: circulating tumor cells and circulating tumor DNA. Cancer Discov. 2014;4(6):650–1.
Manier S, Park J, Capelletti M, Bustoros M, Freeman SS, Ha G, et al. Whole-exome sequencing of cell-free DNA and circulating tumor cells in multiple myeloma. Nat Commun. 2018;9(1):1691.
Davis AA, Zhang Q, Gerratana L, Shah AN, Zhan Y, Qiang W, et al. Association of a novel circulating tumor DNA next-generating sequencing platform with circulating tumor cells (CTCs) and CTC clusters in metastatic breast cancer. Breast Cancer Res. 2019;21(1):137.
Nie LJ, Li FL, Huang XL, Aguilar ZP, Wang YA, Xiong YH, et al. Folic acid targeting for efficient isolation and detection of ovarian cancer CTCs from human whole blood based on two-step binding strategy. ACS Appl Mater Interfaces. 2018;10(16):14055–62.
Wu XX, Xia YZ, Huang YJ, Li J, Ruan HM, Chen TX, et al. Improved SERS-active nanoparticles with various shapes for CTC detection without enrichment process with supersensitivity and high specificity. ACS Appl Mater Interfaces. 2016;8(31):19928–38.
Safarpour H, Dehghani S, Nosrati R, Zebardast N, Alibolandi M, Mokhtarzadeh A, et al. Optical and electrochemical-based nano-aptasensing approaches for the detection of circulating tumor cells (CTCs). Biosens Bioelectron. 2020;148:111833.
Zhong X, Zhang H, Zhu Y, Liang Y, Yuan Z, Li J, et al. Circulating tumor cells in cancer patients: developments and clinical applications for immunotherapy. Mol Cancer. 2020;19(1):15.
Burinaru TA, Avram M, Avram A, Marculescu C, Tincu B, Tucureanu V, et al. Detection of circulating tumor cells using microfluidics. ACS Comb Sci. 2018;20(3):107–26.
Yin JX, Deng JQ, Wang L, Du C, Zhang W, Jiang XY. Detection of circulating tumor cells by fluorescence microspheres-mediated amplification. Anal Chem. 2020;92(10):6968–76.
Cao J, Zhao X-P, Younis MR, Li Z-Q, Xia X-H, Wang C. Ultrasensitive capture, detection, and release of circulating tumor cells using a nanochannel–ion channel hybrid coupled with electrochemical detection technique. Anal Chem. 2017;89(20):10957–64.
Shen C, Liu S, Li X, Yang M. Electrochemical detection of circulating tumor cells based on DNA generated electrochemical current and rolling circle amplification. Anal Chem. 2019;91(18):11614–9.
Parker SG, Yang Y, Ciampi S, Gupta B, Kimpton K, Mansfeld FM, et al. A photoelectrochemical platform for the capture and release of rare single cells. Nat Commun. 2018;9(1):2288.
Shu J, Tang DP. Recent advances in photoelectrochemical sensing: from engineered photoactive materials to sensing devices and detection modes. Anal Chem. 2020;92(1):363–77.
Li C, Chen S, Gao X, Zhang W, Wang Y. Fabrication, characterization and photoelectrochemical properties of CdS/CdSe nanofilm co-sensitized ZnO nanorod arrays on Zn foil subs. J Colloid Interface Sci. 2021;588:269–82.
Eley C, Li T, Liao FL, Fairclough SM, Smith JM, Smith G, et al. Nanojunction-mediated photocatalytic enhancement in heterostructured CdS/ZnO, CdSe/ZnO, and CdTe/ZnO nanocrystals. Angew Chem Int Ed. 2014;53(30):7838–42.
Khan AH, Bertrand GHV, Teitelboim A, Sekhar MC, Polovitsyn A, Brescia R, et al. CdSe/CdS/CdTe core/barrier/crown nanoplatelets: synthesis, optoelectronic properties, and multiphoton fluorescence upconversion. ACS Nano. 2020;14(4):4206–15.
Jung K, Lee J, Kim YM, Kim J, Kim CU, Lee MJ. Influence of defects and nanoscale strain on the photovoltaic properties of CdS/CdSe nanocomposite co-sensitized ZnO nanowire solar cells. Electrochim Acta. 2016;220:500–10.
Kumar SG, Rao KSRK. Comparison of modification strategies towards enhanced charge carrier separation and photocatalytic degradation activity of metal oxide semiconductors (TiO2, WO3 and ZnO). Appl Surf Sci. 2017;391:124–48.
Luo JJ, Liang D, Zhao D, Yang MH. Photoelectrochemical detection of circulating tumor cells based on aptamer conjugated Cu2O as signal probe. Biosens Bioelectron. 2020;151:111976.
Luo JJ, Liang D, Li XQ, Deng L, Wang ZX, Yang MH. Aptamer-based photoelectrochemical assay for the determination of MCF-7. Microchim Acta. 2020;187(5):257.
Shin SR, Zhang YS, Kim DJ, Manbohi A, Avci H, Silvestri A, et al. Aptamer-based microfluidic electrochemical biosensor for monitoring cell-secreted trace cardiac biomarkers. Anal Chem. 2016;88(20):10019–27.
Gao SX, Zheng X, Hu B, Sun MJ, Wu JH, Jiao BH, et al. Enzyme-linked, aptamer-based, competitive biolayer interferometry biosensor for palytoxin. Biosens Bioelectron. 2017;89:952–8.
Xiang WW, Lv QX, Shi HX, Xie B, Gao L. Aptamer-based biosensor for detecting carcinoembryonic antigen. Talanta. 2020;214:120716.
Zamiri R, Abbastabar Ahangar H, Zakaria A, Zamiri G, Shabani M, Singh B, et al. The structural and optical constants of Ag2S semiconductor nanostructure in the far-infrared. Chem Cent J. 2015;9(1):28.
Hou Y, Zhu L, Hao H, Zhang Z, Ding C, Zhang G, et al. A novel photoelectrochemical aptamer sensor based on rare-earth doped Bi2WO6 and Ag2S for the rapid detection of Vibrio parahaemolyticus. Microchem J. 2021;165:106132.
Zhang L, Li P, Feng L, Chen X, Jiang J, Zhang S, et al. Synergetic Ag2S and ZnS quantum dots as the sensitizer and recognition probe: a visible light-driven photoelectrochemical sensor for the “signal-on” analysis of mercury (II). J Hazard Mater. 2020;387:121715.
Ding C, Zhang C, Cheng S, Xian Y. Multivalent aptamer functionalized Ag2S nanodots/hybrid cell membrane-coated magnetic nanobioprobe for the ultrasensitive isolation and detection of circulating tumor cells. Adv Funct Mater. 2020;30(16):1909781.
Wu LL, Wen CY, Hu J, Tang M, Qi CB, Li N, et al. Nanosphere-based one-step strategy for efficient and nondestructive detection of circulating tumor cells. Biosens Bioelectron. 2017;94:219–26.
Yang DD, Liu M, Xu J, Yang C, Wang XX, Lou YB, et al. Carbon nanosphere-based fluorescence aptasensor for targeted detection of breast cancer cell MCF-7. Talanta. 2018;185:113–7.
Zheng TT, Zhang QF, Feng S, Zhu JJ, Wang Q, Wang H. Robust nonenzymatic hybrid nanoelectrocatalysts for signal amplification toward ultrasensitive electrochemical cytosensing. J Am Chem Soc. 2014;136(6):2288–91.
Luo J, Liang D, Qiu X, Yang M. Photoelectrochemical detection of breast cancer biomarker based on hexagonal carbon nitride tubes. Anal Bioanal Chem. 2019;411:6889–97.
Roshan H, Ravanan F, Sheikhi MH, Mirzaei A. High-detectivity near-infrared photodetector based on Ag2S nanocrystals. J Alloys Compd. 2021;852:156948.
Ansari JR, Singh N, Mohapatra S, Ahmad R, Saha NR, Chattopadhyay D, et al. Enhanced near infrared luminescence in Ag@Ag2S core-shell nanoparticles. Appl Surf Sci. 2019;463:573–80.
Acknowledgments
The authors are thankful for the support of this work by the Hunan Provincial Science and Technology Plan Project, China (No. 2019TP1001) and Innovation-Driven Project of Central South University (2020CX002).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no competing interests.
All experiments were in accordance with the guidelines of the National Institute of Health, China, and approved by the Institutional Ethical Committee (IEC) of the Second Xiangya Hospital affiliated with Central South University.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 235 kb)
Rights and permissions
About this article
Cite this article
Wang, Z., Luo, J., Yang, M. et al. Photoelectrochemical assay for the detection of circulating tumor cells based on aptamer-Ag2S nanocrystals for signal amplification. Anal Bioanal Chem 413, 5259–5266 (2021). https://doi.org/10.1007/s00216-021-03502-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-021-03502-5