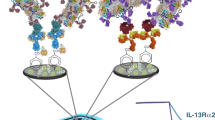
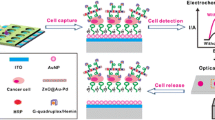
Abstract
Metastasis is responsible for about 90% of cancer-associated deaths. In the context of solid tumors, the low oxygen concentration in the tumor microenvironment (hypoxia) is one of the key factors contributing to metastasis. Tumor cells adapt to these conditions by overexpressing certain proteins such as programmed death ligand 1 (PD-L1) and hypoxia-inducible factor 1 alpha (HIF-1α). However, the determination of these tumor hypoxia markers that can be used to follow-up tumor progression and improve the efficiency of therapies has been scarcely addressed using electrochemical biosensors. In this work, we report the first electrochemical bioplatform for the determination of PD-L1 as well as the first one allowing its simultaneous determination with HIF-1α. The target proteins were captured and enzymatically labeled on magnetic microbeads and amperometric detection was undertaken on the surface of screen-printed dual carbon electrodes using the hydrogen peroxide/peroxidase/hydroquinone system. Sandwich immunoassays were implemented for both the HIF-1α and PD-L1 sensors and the analytical characteristics were evaluated providing LOD values of 86 and 279 pg mL−1 for the amperometric determination of PD-L1 and HIF-1α standards, respectively. The developed electrochemical immunoplatforms are competitive versus the only electrochemical immunosensor reported for the determination of HIF-1α and the “gold standard” ELISA methodology for the single determination of both proteins in terms of assay time, compatibility with the simultaneous determination of both proteins making their use suitable for untrained users at the point of attention. The dual amperometric immunosensor was applied to the simultaneous determination of HIF-1α and PD-L1 in cancer cell lysates. The analyses lasted only 2 h and just 0.5 μg of the sample was required.
Graphical abstract







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
International Agency for Research on Cancer, World Health Organization, Press Release N° 292, 15 December 2020. https://www.iarc.fr/wp-content/uploads/2020/12/pr292_E.pdf. Accessed 20 Dec 2020.
Tawadros AIF, Khalafalla MMM. Expression of programmed death-ligand 1 and hypoxia-inducible factor-1α proteins in endometrial carcinoma. J Cancer Res Ther. 2018;14:1063–9.
Barsoum IB, Smallwood CA, Siemens DR, Graham CH. A mechanism of hypoxia-mediated escape from adaptive immunity in cancer cells. Cancer Res. 2014;74:665–74.
Noman MZ, Desantis G, Janji B, Hasmim M, Karray S, Dessen P, et al. PD-L1 is a novel direct target of HIF-1α, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J Exp Med. 2014;211:781–90.
Li Y, Patel SP, Roszik J, Qin Y. Hypoxia-driven immunosuppressive metabolites in the tumor microenvironment: new approaches for combinational immunotherapy. Front Immunol. 2018;9:1591. https://doi.org/10.3389/fimmu.2018.01591.
Cubillos-Zapata C, Balbás-García C, Avendaño-Ortiz J, Toledano V, Torres M, Almendros I, et al. Age-dependent hypoxia-induced PD-L1 upregulation in patients with obstructive sleep apnoea. Respirology. 2019;24:684–92.
Wu Y, Chen W, Xu ZP, Gu W. PD-L1 distribution and perspective for cancer immunotherapy—blockade, knockdown, or inhibition. Front Immunol. 2019;10:2022. https://doi.org/10.3389/fimmu.2019.02022.
Chen TC, Wu CT, Wang CP, Hsu W-L, Yang T-L, Lou P-J, et al. Associations among pretreatment tumor necrosis and the expression of HIF-1alpha and PD-L1 in advanced oral squamous cell carcinoma and the prognostic impact thereof. Oral Oncol. 2015;51:1004–10.
Chang YL, Yang CY, Lin MW, Wu CT, Yang PC. High co-expression of PD-L1 and HIF-1alpha correlates with tumour necrosis in pulmonary pleomorphic carcinoma. Eur J Cancer. 2016;60:125–35.
Dogan HT, Kiran M, Bilgin B, Kiliçarslan A, Sendur MAN, Yalçin B, et al. Prognostic significance of the programmed death ligand 1 expression in clear cell renal cell carcinoma and correlation with the tumor microenvironment and hypoxiainducible factor expression. Diagn Pathol. 2018;13:60. https://doi.org/10.1186/s13000-018-0742-8.
Guo R, Li Y, Wang Z, Bai H, Duan J, Wang S, et al. Hypoxia-inducible factor-1α and nuclear factor-κB play important roles in regulating programmed cell death ligand 1 expression by epidermal growth factor receptor mutants in non-small-cell lung cancer cells. Cancer Sci. 2019;110:1665–75.
Koh YW, Lee SJ, Han J-H, Haam S, Jung J, Lee HW. PD-L1 protein expression in non-small-cell lung cancer and its relationship with the hypoxia-related signaling pathways: a study based on immunohistochemistry and RNA sequencing data. Lung Cancer. 2019;129:41–7.
Zhao Y, Wang X, Wu W, Long H, Huang J, Wang Z, et al. EZH2 regulates PD-L1 expression via HIF-1a in non-small cell lung cancer cells. Biochem Biophys Res Commun. 2019;517:201–9.
Zhou L, Cha G, Chen L, Yang C, Xu D, Ge M. HIF1α/PD-L1 axis mediates hypoxia-induced cell apoptosis and tumor progression in follicular thyroid carcinoma. Onco Targets Ther. 2019;12:6461–70.
Wen Q, Han T, Wang Z, Jiang S. Role and mechanism of programmed death-ligand 1 in hypoxia-induced liver cancer immune escape (review). Oncol Lett. 2020;19:2595–601.
Schott DS, Pizon M, Pachmann U, Pachmann K. Sensitive detection of PD-L1 expression on circulating epithelial tumor cells (CETCs) could be a potential biomarker to select patients for treatment with PD-1/PD-L1 inhibitors in early and metastatic solid tumors. Oncotarget. 2017;8(42):72755–72.
Kythreotou A, Siddique A, Mauri FA, Bower M, Pinato DJ. PD-L1. J Clin Pathol. 2018;71:189–94.
Oh KS, Mahalingam M. PD-L1 detection—pearls and pitfalls associated with current methodologies focusing on entities relevant to dermatopathology. Am J Dermatopathol. 2019;41:539–65.
Stepula E, König M, Wang X-P, Levermann J, Schimming T, Kasimir-Bauer S, et al. Localization of PD-L1 on single cancer cells by iSERS microscopy with Au/Au core/satellite nanoparticles. J Biophotonics. 2019:e201960034. https://doi.org/10.1002/jbio.201960034.
Costantini A, Kamga PT, Dumenil C, Chinet T, Emile J-F, Leprieur EG. Plasma biomarkers and immune checkpoint inhibitors in non-small cell lung cancer: new tools for better patient selection? Cancers. 2019;11(9):1269. https://doi.org/10.3390/cancers11091269.
Ilie M, Hofman V, Dietel M, Soria J-C, Hofman P. Assessment of the PD-L1 status by immunohistochemistry: challenges and perspectives for therapeutic strategies in lung cancer patients. Virchows Arch. 2016;468:511–25.
Grosu HB, Arriola A, Stewart J, Ma J, Bassett R, Hernandez M, et al. PD-L1 detection in histology specimens and matched pleural fluid cell blocks of patients with NSCLC. Respirology. 2019;24:1198–203.
Hsieh T-C, Wu JM. Tumor PD-L1 induction by resveratrol/piceatannol may function as a search, enhance, and engage (“SEE”) signal to facilitate the elimination of “cold, non-responsive” low PD-L1-expressing tumors by PD-L1 blockade. Int J Mol Sci. 2019;20(23):5969. https://doi.org/10.3390/ijms20235969.
Janning M, Kobus F, Babayan A, Wikman H, Velthaus J-L, Bergmann S, et al. Determination of PD-L1 expression in circulating tumor cells of NSCLC patients and correlation with response to PD-1/PD-L1 inhibitors. Cancers. 2019;11(6):835. https://doi.org/10.3390/cancers11060835.
Shibel PEE, Abdelhamid HS, Soliman SAM, Gabal SM. Investigation of immunohistochemical expression of programmed death-ligand 1 (PD-L1) in female mammary carcinoma and its correlation with the extent of stromal tumour infiltrating lymphocytes. J Clin Diagn Res. 2019;13(9):EC11–7.
Bubendorf L, Conde E, Cappuzzo F, Langfort R, Schildhaus H-U, Votruba J, et al. A noninterventional, multinational study to assess PD-L1 expression in cytological and histological lung cancer specimens. Cancer Cytopathol. 2020;128:928–38.
Park H, Bang J-H, Nam A-R, Park JE, Jin MH, Bang Y-J, et al. Prognostic implications of soluble programmed death-ligand 1 and its dynamics during chemotherapy in unresectable pancreatic cancer. Sci Rep. 2019;9:11131. https://doi.org/10.1038/s41598-019-47330-1.
Li Y, Cui X, Yang Y-J, Chen Q-Q, Zhong L, Zhang T, et al. Serum sPD-1 and sPD-L1 as biomarkers for evaluating the efficacy of neoadjuvant chemotherapy in triple-negative breast cancer patients. Clin Breast Cancer. 2019;19(5):326–32.
Dai X, Pi G, Yang S-L, Chen GG, Liu L-P, Dong H.-H. Association of PD-L1 and HIF-1α coexpression with poor prognosis in hepatocellular carcinoma. Transl Oncol 2018;11:559–566.
Chen B, Li L, Li M, Wang X. HIF1A expression correlates with increased tumor immune and stromal signatures and aggressive phenotypes in human cancers. Cell Oncol. 2020;43:877–88.
Baraket A, Lee M, Zine N, Sigaud M, Bausells J, Errachid A. A fully integrated electrochemical biosensor platform fabrication process for cytokines detection. Biosens Bioelectron. 2017;93:170–5.
Aydin EB, Aydin M, Sezgintürk MK. The development of an ultra-sensitive electrochemical immunosensor using a PPyr-NHS functionalized disposable ITO sheet for the detection of interleukin 6 in real human serums. New J Chem. 2020;44:14228–38.
Cortina ME, Melli LJ, Roberti M, Mass M, Longinotti G, Tropea S, et al. Electrochemical magnetic microbeads-based biosensor for point-of-care serodiagnosis of infectious diseases. Biosens Bioelectron. 2016;80:24–33.
Kudr J, Klejdus B, Adam V, Zitka O. Magnetic solids in electrochemical analysis. TrAC Trends Anal Chem. 2018;98:104–13.
Pastucha M, Farka Z, Lacina K, Mikušová Z, Skládal P. Magnetic nanoparticles for smart electrochemical immunoassays: a review on recent developments. Microchim Acta. 2019;186:312. https://doi.org/10.1007/s00604-019-3410-0.
Pingarrón JM, Campuzano S, González-Cortés A, Yáñez-Sedeño P. Electrochemical immunosensors for clinical diagnostics. In: Wandelt K, editor. Encyclopedia of interfacial chemistry: surface science and electrochemistry: Elsevier; 2018. p. 156–65.
Hussain KK, Gurudatt NG, Mir TA, Shim Y-B. Amperometric sensing of HIF1α expressed in cancer cells and the effect of hypoxic mimicking agents. Biosens Bioelectron. 2016;83:312–8.
Muñoz-San Martín C, Gamella M, Pedrero M, Montero-Calle A, Barderas R, Campuzano S, et al. Magnetic beads-based electrochemical immunosensing of HIF-1α, a biomarker of tumoral hypoxia. Sensors Actuators B Chem. 2020;307:127623. https://doi.org/10.1016/j.snb.2019.127623.
Torrente-Rodríguez RM, Ruiz-Valdepeñas Montiel V, Campuzano S, Pedrero M, Farchado M, Vargas E, et al. Electrochemical sensor for rapid determination of fibroblast growth factor receptor 4 in raw cancer cell lysates. PLoS One. 2017;12:e0175056. https://doi.org/10.1371/journal.pone.0175056.
Valverde A, Povedano E, Ruiz-Valdepeñas Montiel V, Yáñez-Sedeño P, Garranzo-Asensio M, Barderas R, et al. Electrochemical immunosensor for IL-13 receptor α2 determination and discrimination of metastatic colon cancer cells. Biosens Bioelectron. 2018;117:766–72.
Xue H, Bei Y, Zhan Z, Chen X, Xu X, Fu YV. Utilizing biotinylated proteins expressed in yeast to visualize DNA–protein interactions at the single-molecule level. Front Microbiol. 2017;8:2062. https://doi.org/10.3389/fmicb.2017.02062.
Escamilla-Gómez V, Campuzano S, Pedrero M, Pingarrón JM. Development of an amperometric immunosensor for the quantification of Staphylococcus aureus using self-assembled monolayer-modified electrodes as immobilization platforms. Electroanalysis. 2007;19:1476–82.
Gadagkar SR, Call GB. Computational tools for fitting the Hill equation to dose–response curves. J Pharmacol Toxicol Methods. 2015;71:68–76.
Finkelmeier F, Canli O, Tal A, Pleli T, Trojan J, Schmidt M, et al. High levels of the soluble programmed death-ligand (sPD-L1) identify hepatocellular carcinoma patients with a poor prognosis. Eur J Cancer. 2016;59:152–9.
Wuethrich A, Rajkumar AR, Shanmugasundaram KB, Reza KK, Dey S, Howard CB, et al. Single droplet detection of immune checkpoints on a multiplexed electrohydrodynamic biosensor. Analyst. 2019;144:6914–21.
Yazdian-Robati R, Ramezani M, Khedri M, Ansari N, Abnous K, Taghdisi SM. An aptamer for recognizing the transmembrane protein PDL-1 (programmed death-ligand 1), and its application to fluorometric single cell detection of human ovarian carcinoma cells. Microchim Acta. 2017;184:4029–35.
Ou Y-C, Wen X, Johnson CA, Shae D, Ayala OD, Webb JA, et al. Multimodal multiplexed immunoimaging with nanostars to detect multiple immunomarkers and monitor response to immunotherapies. ACS Nano. 2020;14:651–63.
Zhang M, Jiang H, Zhang R, Jiang H, Xu H, Pan W, et al. Near-infrared fluorescence-labeled anti-PD-L1-mAb for tumor imaging in human colorectal cancer xenografted mice. J Cell Biochem. 2019;120:10239–47.
Liu C, Zeng X, An Z, Yang Y, Eisenbaum M, Gu X, et al. Sensitive detection of exosomal proteins via a compact surface plasmon resonance biosensor for cancer diagnosis. ACS Sens. 2018;3:1471–9.
Pang Y, Shi J, Yang X, Wang C, Sun Z, Xiao R. Personalized detection of circling exosomal PD-L1 based on Fe3O4@TiO2 isolation and SERS immunoassay. Biosens Bioelectron. 2020;148:111800. https://doi.org/10.1016/j.bios.2019.111800.
He Y, Wu Y, Wang Y, Wang X, Xing S, Li H, et al. Applying CRISPR/Cas13 to construct exosomal PD-L1 ultrasensitive biosensors for dynamic monitoring of tumor progression in immunotherapy. Adv Ther. 2020;3:2000093. https://doi.org/10.1002/adtp.202000093.
Cao Y, Wang Y, Yu X, Jiang X, Li G, Zhao J. Identification of programmed death ligand-1 positive exosomes in breast cancer based on DNA amplification-responsive metal-organic frameworks. Biosens Bioelectron. 2020;166:112452. https://doi.org/10.1016/j.bios.2020.112452.
Melanson SEF, Tanasijevic MJ, Jarolim P. Cardiac troponin assays: a view from the clinical chemistry laboratory. Circulation. 2007;116:e501–4.
Tate J, Ward G. Interferences in immunoassay. Clin Biochem Rev. 2004;25:105–20.
Grigorieva DV, Gorudko IV, Sokolov AV, Kosmachevskaya OV, Topunov AF, Buko IV, et al. Measurement of plasma hemoglobin peroxidase activity. Bull Exp Biol Med. 2013;155:118–21.
Anderson RL, Chaplin H Jr. Plasma hemoglobin concentration in umbilical cord blood. Am J Dis Child. 1963;105:19–26.
Zhao R-N, Feng Z, Zhao Y-N, Jia L-P, Ma R-N, Zhang W, et al. A sensitive electrochemical aptasensor for Mucin 1 detection based on catalytic hairpin assembly coupled with PtPdNPs peroxidase-like activity. Talanta. 2019;200:503–10.
Kurozumi S, Inoue K, Matsumoto H, Fujii T, Horiguchi J, Oyama T, et al. Clinicopathological values of PD-L1 expression in HER2-positive breast cancer. Sci Rep. 2019;9:16662. https://doi.org/10.1038/s41598-019-52944-6.
Grenga I, Donahue RN, Lepone L, Bame J, Schlom J, Farsaci B. PD-L1 and MHC-I expression in 19 human tumor cell lines and modulation by interferon-gamma treatment. J Immunother Cancer. 2014;2:P102. https://doi.org/10.1186/2051-1426-2-S3-P102.
Wang HB, Yao H, Li CS, Liang LX, Zhang Y, Chen YX, et al. Rise of PD-L1 expression during metastasis of colorectal cancer: implications for immunotherapy. J Dig Dis. 2017;18:574–81.
Shan T, Chen S, Wu T, Yang Y, Li S, Chen X. PD-L1 expression in colon cancer and its relationship with clinical prognosis. Int J Clin Exp Pathol. 2019;12:1764–9.
Kerr KM, Nicolson MC. Non–small cell lung cancer, PD-L1, and the pathologist. Arch Pathol Lab Med. 2016;140:249–54.
Rom-Jurek E-M, Kirchhammer N, Ugocsai P, Ortmann O, Wege AK, Brockhoff G. Regulation of programmed death ligand 1 (PD-L1) expression in breast cancer cell lines in vitro and in immunodeficient and humanized tumor mice. Int J Mol Sci. 2018;19(2):563. https://doi.org/10.3390/ijms19020563.
Prasad J, Goswami B, Gowda SH, Gupta N, Kumar S, Agarwal K, et al. Does hypoxia-inducible factor-1a (HIF-1a) C1772T polymorphism predict short-term prognosis in patients with oral squamous cell carcinoma (OSCC). J Oral Pathol Med. 2018;47:660–4.
Acknowledgements
The financial support of PID2019-103899RB-I00 (Spanish Ministerio de Ciencia e Innovación), RTI2018-096135-B-I00 (Spanish Ministerio de Ciencia, Innovación y Universidades) Research Projects, PI17CIII/00045 and PI20CIII/00019 Grants from the AES-ISCIII Program co-founded by FEDER funds, and the TRANSNANOAVANSENS-CM Program from the Comunidad de Madrid (Grant S2018/NMT-4349) are gratefully acknowledged. C.M.-S.M. acknowledges a predoctoral contract from Universidad Complutense de Madrid. A.M-C. is supported by a FPU predoctoral contract supported by the Spanish Ministerio de Educación, Cultura y Deporte.
Funding
Spanish Ministerio de Ciencia e Innovación (PID2019-103899RB-I00), Spanish Ministerio de Ciencia, Innovación y Universidades (RTI2018–096135-B-I00), AES-ISCIII Program co-founded by FEDER funds (PI17CIII/00045 and PI20CIII/00019 Grants), TRANSNANOAVANSENS-CM Program from the Comunidad de Madrid (Grant S2018/NMT-4349).
Author information
Authors and Affiliations
Contributions
Cristina Muñoz-San Martín: conceptualization, investigation, writing, review, and editing—original draft. Maria Gamella: conceptualization, investigation, writing, review, and editing—original draft. María Pedrero: conceptualization, investigation, writing, review, and editing—original draft. Ana Montero-Calle: investigation, resources, review, and editing—original draft. Víctor Pérez-Ginés: investigation. Rodrigo Barderas: conceptualization, supervision, resources, review, and editing—original draft and funding acquisition. José M. Pingarrón: Review and editing—original draft. Susana Campuzano: conceptualization, supervision, writing, review, and editing—original draft and funding acquisition.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the Ethics Committee (Institutional Review Board) of the Hospital Universitari de Sant Joan de Reus (project code: CEIM 014/2017).
Consent to participate
All patients signed a written informed consent according to the declaration of Helsinki.
Consent for publication
All authors consent to the publication of the manuscript in Analytical and Bioanalytical Chemistry, should the article be accepted by the Editor-in-chief upon completion of the refereeing process.
Conflicts of interest
The authors declare no competing interests.
Disclaimer
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Published in the topical collection celebrating ABCs 20th Anniversary.
Supplementary information
ESM 1
(PDF 728 kb)
Rights and permissions
About this article
Cite this article
Muñoz-San Martín, C., Gamella, M., Pedrero, M. et al. Anticipating metastasis through electrochemical immunosensing of tumor hypoxia biomarkers. Anal Bioanal Chem 414, 399–412 (2022). https://doi.org/10.1007/s00216-021-03240-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-021-03240-8