Abstract
We developed and evaluated a candidate serum reference material to help improve clinical routine measurement, and to provide traceability of the measurement results. D8-Homocystine, dithiothreitol, and acetonitrile were used as an internal standard, the reducing agent, and the protein precipitating agent, respectively. A triple quadrupole mass spectrometer with an electrospray ionization source was used for monitoring the transitions (m/z 140.0 → 94.0, 136.0 → 90.0) in multiple-reaction-monitoring mode. We used a calibration model relying on bracketing and gravimetric measurements to give SI-traceability and higher accuracy to serum value assignments. The method was evaluated for accuracy using NIST Standard Reference Material SRM1955. The results of the three concentrations (1, 2, and 3) of total homocysteine in human serum samples were determined by an isotope-dilution liquid chromatography-tandem mass spectrometry method; tHcy 1 is 28.8 ± 1.1 μmol/L, tHcy 2 is 17.93 ± 0.57 μmol/L, and tHcy 3 is 14.38 ± 0.46 μmol/L.

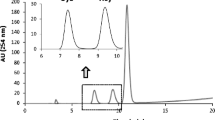
The workflow diagram.


Similar content being viewed by others
References
Palazhy S, Kamath P, Vasudevan DM. Elevated oxidative stress among coronary artery disease patients on statin therapy: a cross sectional study. Indian Heart J. 2015;67(3):227–32.
Verdoia M, Schaffer A, Pergolini P, et al. Homocysteine levels influence platelet reactivity in coronary artery disease patients treated with acetylsalicylic acid. J Cardiovasc Pharmacol. 2015;66(1):35–40.
Kumakura H, Fujita K, Kanai H, et al. High-sensitivity C-reactive protein, lipoprotein(a) and homocysteine are risk factors for coronary artery disease in Japanese patients with peripheral arterial disease. J Atheroscler Thromb. 2015;22(4):344–54.
Shenoy V, Mehendale V, Prabhu K, et al. Correlation of serum homocysteine levels with the severity of coronary artery disease. Indian J Clin Biochem. 2014;29(3):339–44.
Alvarez B, Yugueros X, Fernández E, et al. Relationship between plasma homocysteine and the morphological and immunohistochemical study of carotid plaques in patients with carotid stenosis over 70%. Ann Vasc Surg. 2012;26(4):500–5.
Choi JW, Lee MH, Fujii T, et al. Association of the urine homocysteine/creatinine ratio to proinflammatory cytokine, natural anticoagulant, and nitric oxide levels in cerebrovascular disease. Ann Clin Lab Sci. 2014;44(4):461–5.
Mouravas H, Verettas D, Kazakos K, et al. Homocysteine and its relationship to deep venous thrombosis in patients undergoing total knee or hip arthroplasty. Hippokratia. 2010;14(3):185–8.
Hoţoleanu C, Porojan-Iuga M, Rusu ML, et al. Hyperhomocysteinemia: clinical and therapeutical involvement in venous thrombosis. Rom J Intern Med. 2007;45(2):159–64.
Amores-Sánchez MI, Medina MA. Methods for the determination of plasma total homocysteine: a review. Clin Chem Lab Med. 2000;38(3):199–204.
Rasmussen K, Møller J. Total homocysteine measurement in clinical practice. Ann Clin Biochem. 2000;37(Pt 5):627–48.
Ubbink JB. Assay methods for the measurement of total homocyst(e)ine in plasma. Semin Thromb Hemost. 2000;26(3):233–41.
Magera MJ, Lacey JM, Casetta B, et al. Method for the determination of total homocysteine in plasma and urine by stable isotope dilution and electrospray tandem mass spectrometry. Clin Chem. 1999;45(9):1517–22.
Satterfield MB, Sniegoski LT, Welch MJ, et al. Comparison of isotope dilution mass spectrometry methods for the determination of total homocysteine in plasma and serum. Anal Chem. 2003;75:4631–8.
Nelson BC, Pfeiffer CM, Sniegoski LT, et al. Development and evaluation of an isotope dilution LC/MS method for the determination of total homocysteine in human plasma. Anal Chem. 2003;75:775–84.
Nelson BC, Satterfield MB, Sniegoski LT, et al. Simultaneous quantification of homocysteine and folate in human serum or plasma using liquid chromatography/tandem mass spectrometry. Anal Chem. 2005;77:3586–93.
Satterfield MB, Sniegoski LT, Sharpless KE, et al. Development of a new standard reference material: SRM 1955 (homocysteine and folate in human serum). Anal Bioanal Chem. 2006;385:612–22.
Ducros V, Belva-Besnet H, Casetta B, et al. A robust liquid chromatography tandem mass spectrometry method for total plasma homocysteine determination in clinical practice. Clin Chem Lab Med. 2006;44(8):987–90.
World Health Organization. Laboratory biosafety manual—third edition. WHO, 2004.
Clinical and Laboratory Standards Institute. EP30-A characterization and qualification of commutable reference materials for laboratory medicine; approved guideline. CLSI, 2015.
Acknowledgements
This work is financially supported by the National Natural Science Foundation of China (No. 21275134) and National Key Technology Research and Development Program of the Ministry of Science and Technology of China (2013BAK10B01, 2011FY130100, 2011BAI02B05, 2011AA02A111).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Ethics Committee of Beijing Aerospace General Hospital and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent was obtained from all individual participants included in the study.
This article does not contain any studies with animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Liu, Y., Song, D., Xu, B. et al. Development of a matrix-based candidate reference material of total homocysteine in human serum. Anal Bioanal Chem 409, 3329–3335 (2017). https://doi.org/10.1007/s00216-017-0272-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-017-0272-3