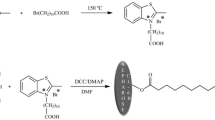
Abstract
Immobilized metal affinity chromatography (IMAC) has been widely used for the specific separation of biopolymers. However, leakage of metal ions from IMAC adsorbents is of concern in IMAC. In this study, we designed a novel tridenate bis(5-methyltetrazolium)amine (BMTA) to reduce the leakage of metal ions by improving the affinity to immobilized metal ions. The ligand was bonded onto silica via three-step reaction to prepare a high-performance IMAC stationary phase. The chromatographic behaviors of ribonuclease A, cytochrome c, and lysozyme on the Cu(II)-, Ni(II)-, and Zn(II)-chelated stationary phase were investigated with respect to pH effect and elution with an imidazole gradient. The retention times of these three proteins increased by increasing the pH of the mobile phase but decreased by increasing the concentration of the competitive displacer. The retaining strength of the three proteins on the chelated stationary phase were in the order Cu(II) > Ni(II) > Zn(II). The behavior of these three proteins was consistent with the properties of a typical IMAC. The BMTA ligand exhibited a much stronger affinity for Cu(II) and Ni(II) than iminodiacetic acid (IDA), which is often regarded as a standard tridentate IMAC ligand. Quantum mechanical calculations at the B3LYP/6-31G level were used to image the coordination mode of the protein-metal ions-BMTA complex. In addition, a fused histidine-tagged cecropin b-human epidermal growth factor (CB-EGF) from Escherichia coli crude extract was purified by the Ni(II)-chelated stationary phase, and the purity of the CB-EGF was determined to be at least 90 %. These results suggest that the BMTA ligand may have potential applications in the preparation of therapeutics.

A novel ligand of tridenate bis(5-methyltetrazolium)amine (BMTA) was designed to reduce the leakage of metal ions from the column in immobolized metal affinity chromatography (IMAC).










Similar content being viewed by others
References
Porath J, Carlsson JAN, Olsson I, Belfrage G. Metal chelate affinity chromatography, a new approach to protein fractionation. Nature. 1975;258:598–9.
Bibi NS, Singh NK, Dsouza RN, Aasim M, Fernández-Lahore M. Synthesis and performance of megaporous immobilized metal-ion affinity cryogels for recombinant protein capture and purification. J Chromatogr A. 2013;1272:145–9.
Mirza MR, Rainer M, Messner CB, Güzel Y, Schemeth D, Stasyk T, et al. A new type of metal chelate affinity chromatography using trivalent lanthanide ions for phosphopeptide enrichment. Analyst. 2013;138:2995–3004.
Smith CL, Stauber JL, Wilson MR, Jolley DF. The use of immobilized metal affinity chromatography (IMAC) to compare expression of copper-binding proteins in control and copper-exposed marine microalgae. Anal Bioanal Chem. 2014;406:305–15.
Block H, Maertens B, Priestersbach A, Brinker N, Kubicek J, Fabis R, et al. Immobilized-metal affinity chromatography (IMAC): a review. Methods Enzymol. 2009;463:439–73.
Kowalska E, Bartnicki F, Pels K, Strzalka W. The impact of immobilized metal affinity chromatography (IMAC) resins on DNA aptamer selection. Anal Bioanal Chem. 2014;406:5495–9.
Jiang W, Hearn MTW. Protein interaction with immobilized metal ion affinity ligands under high ionic strength conditions. Anal Biochem. 1996;242:45–54.
Gaberc-Porekar V, Menart V. Perspectives of immobilized-metal affinity chromatography. J Biochem Bioph Methods. 2001;49:335–60.
Hage DS, Anguizola JA, Bi C, Li R, Matsuda R, Papastavros E, et al. Pharmaceutical and biomedical applications of affinity chromatography: recent trends and developments. J Pharm Biomed Anal. 2012;69:93–105.
Pfaunmiller E, Paulemond M, Dupper C, Hage D. Affinity monolith chromatography: a review of principles and recent analytical applications. Anal Bioanal Chem. 2013;405:2133–45.
Yao XP, Fu ZJ, Zhao YG, Wang L, Fang LY, Shen HY. Use of tetraethylenepentamine-functional Fe3O4 magnetic polymers for matrix solid phase dispersion extraction and preconcentration of Cr(VI) in water samples at ultratrace levels. Talanta. 2012;97:124–30.
Chaouk H, Hearn MTW. New ligand, N-(2-pyridylmethyl)aminoacetate, for use in the immobilized metal ion affinity chromatographic separation of proteins. J Chromatogr A. 1999;852:105–15.
Petzold M, Coghlan CJ, Hearn MTW. Studies with an immobilized metal affinity chromatography cassette system involving binuclear triazacyclononane-derived ligands: automation of batch adsorption measurements with tagged recombinant proteins. J Chromatogr A. 2014;1351:61–9.
Mooney JT, Fredericks D, Hearn MTW. Use of phage display methods to identify heptapeptide sequences for use as affinity purification ‘tags’ with novel chelating ligands in immobilized metal ion affinity chromatography. J Chromatogr A. 2011;1218:92–9.
Mooney JT, Fredericks D, Christensen T, Hearn MTW. Removal of cleavage slow points from affinity tags used in the IMAC purification of recombinant proteins. J Biotechnol. 2014;9:1023–32.
Mooney JT, Fredericks D, Hearn MTW. Application of an IMAC cassette for the purification of N-terminally tagged proteins. Sep Purif Technol. 2013;120:265–74.
Ueda EKM, Gout PW, Morganti L. Current and prospective applications of metal ion-protein binding. J Chromatogr A. 2003;988:1–23.
Kagedal L. In: Janson JC, Ryden L, editors. Protein purification. New York: Wiley; 1989. p. 227–51.
Porath J. IMAC-Immobilized metal ion affinity based chromatography. Trends Anal Chem. 1988;7:254–9.
Codd R, Gu J, Ejje N, Lifa T. New applications of immobilized metal ion affinity chromatography in chemical biology. In: Inorganic chemical biology. Wiley, 2014. 1–35.
Lei GH, Liu LT, Xiong XH, Wei YM, Zheng XH. New α-amino phenylalanine tetrazole ligand for immobilized metal affinity chromatography of proteins. J Sep Sci. 2008;31:3002–8.
Sheng RL, Wang PF, Gao YH, Wu Y, Liu WM, Ma JJ, et al. Colorimetric test kit for Cu2+ detection. Org Lett. 2008;10:5015–8.
Demko ZP, Sharpless KB. An expedient route to the tetrazole analogues of α-amino acids. Org Lett. 2004;4:2525–7.
Mehyou Z, Lobinski R, Hagège A. One-step coating of silica capillaries for selective protein retention by Cu(II)-IDA IMAC. Talanta. 2011;87:168–73.
Morley JS. Polypeptides. XI. Tetrazole analogues of the C-terminal tetrapeptide amide sequence of the gastrins. J Chem Soc. 1969;5:809–13.
Zhang Y, Chen YN, Wang CZ, Wei YM. Immobilization of 5-aminopyridine-2-tetrazole on cross-linked polystyrene for the preparation of a new adsorbent to remove heavy metal ions from aqueous solution. J Hazard Mater. 2014;76:129–37.
Jin XL, Yu C, Li YF, Qi YX, Yang LQ, Zhao GH, et al. Preparation of novel nano-adsorbent based on organic–inorganic hybrid and their adsorption for heavy metals and organic pollutants presented in water environment. J Hazard Mater. 2011;186:1672–80.
Jiang W, Chen XB, Pan BC, Zhang QX, Teng L, Chen YF, et al. Spherical polystyrene supported chitosan thin film of fast kinetics and high capacity for copper removal. J Hazard Mater. 2014;276:295–301.
Lei GH, Xiong XH, Wei YM, Zheng XH, Zheng JB. Novel tetrazole-functionalized ion exchanger for weak cation-exchange chromatography of proteins. J Chromatogr A. 2008;1187:197–204.
Belew M, Porath J. Immobilized metal ion affinity chromatography: effect of solute structure, ligand density, and salt concentration on the retention of peptides. J Chromatogr A. 1990;516:333–54.
Wu CY, Suen SY, Chen SC, Tzeng JH. Analysis of protein adsorption on regenerated cellulose-based immobilized copper ion affinity membranes. J Chromatogr A. 2003;996:53–70.
Haley R, Fruchtl M, Brune EM, Ataai M, Henry R, Beitle R. A redesigned escherichia coli triosephosphate isomerase restores growth properties in a bacterial strain useful for immobilized metal affinity chromatography (IMAC). J Biotechnol. 2014;188:48–52.
Leonidas DD, Maiti TK, Samanta A, Dasgupta S, Pathak T, Zographos SE, et al. The binding of 3'-N-piperidine-4-carboxyl-3'-deoxy-ara-uridine to ribonuclease A in the crystal. Bioorg Med Chem. 2006;14:6055–64.
Di ZM, Chen GL, Lei JD, Li R, Li HR. Synthesis of metal chelate affinity chromatographic medium and studies of chromatographic characteristics. Chin J Chromatogr. 1998;6:297–300.
Geng XD. Guide to theory of modern separation science. Beijing: Higher Education Press; 2001. p. 54–108.
Geng XD, Regnier FE. Retention model for proteins in reversed-phase liquid chromatography. J Chromatogr A. 1984;296:15–30.
Ko YM, Chen CI, Shieh CJ, Liu YC. Simultaneous purification and immobilization of d-hydantoinase on the immobilized metal affinity membrane via coordination bonds. Biochem Eng J. 2012;61:20–7.
Tsai YH, Wang MY, Suen SY. Purification of hepatocyte growth factor using polyvinyldiene fluoride based immobilized metal affinity membranes: equilibrium adsorption study. J Chromatogr B. 2002;766:133–43.
Li R, Wang Y, Chen GL, Mei S, Wang XG, Zheng JB, et al. Effects of immobilized metal ion on retention behaviors of proteins in metal chelate affinity chromatography. J Chin Chem Soc. 2011;58:215–21.
Sharma S, Agarwal GP. Interactions of proteins with immobilized metal ions: a comparative analysis using various isotherm models. Anal Biochem. 2001;288:126–40.
Chen WY, Wu CF, Liu CC. Interactions of imidazole and proteins with immobilized Cu(II) ions: effects of structure, salt concentration, and pH in affinity and binding capacity. J Colloid Interface Sci. 1996;180:135–43.
Geng XD, Ke CY, Chen G, Liu P, Wang F, Zhang HQ, et al. On-line separation of native proteins by two-dimensional liquid chromatography using a single column. J Chromatogr A. 2009;1216:3553–62.
Rassi Z, Horváth C. Metal chelate-interaction chromatography of proteins with iminodiacetic acid-bonded stationary phases on silica support. J Chromatogr A. 1986;359:241–53.
Raghavachari K. Perspective on Density functional thermochemistry. III. The role of exact exchange. Theor Chem Acc. 2000;103:361–3.
De Meyer T, Hemelsoet K, Van der Schueren L, Pauwels E, De Clerck K, Van Speybroeck V. Investigating the halochromic properties of azo dyes in an aqueous environment by using a combined experimental and theoretical approach. Chem Eur J. 2012;18:8120–9.
Improta R. Quantum mechanical calculations unveil the structure and properties of the absorbing and emitting excited electronic states of guanine quadruplex. Chem Eur J. 2014;20:8106–15.
Wan Y, Shen WR, Han LP. Prokaryotic expression, purification and fermentation of cecropin B and epidermal growth factor fusion protein. Chin J Biol. 2009;22:1075–9.
Chang Q, Liang F, Chang JH. Multiple injection method of protein separation on liquid chromatography. Chem World. 2006;47:81–7.
Acknowledgments
This work was supported by the Natural Science Foundation of China (Nos. 21275115, 21475104, and 21575114), the Industry Development Project by the Science and Technology Department of Shaanxi Province (No. 2016GY-214), and the Changjiang Scholars and Innovative Research Team in University (IRT-15R55).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 525 kb)
Rights and permissions
About this article
Cite this article
Bo, C., Wang, C. & Wei, Y. Novel bis(5-methyltetrazolium)amine ligand-bonded stationary phase with reduced leakage of metal ions in immobilized metal affinity chromatography of proteins. Anal Bioanal Chem 408, 7595–7605 (2016). https://doi.org/10.1007/s00216-016-9826-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9826-z