Abstract
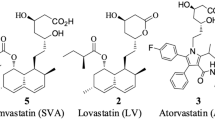
The combination of ultrahigh-resolution mass spectrometry imaging (UHRMSI) and ultrahigh-performance liquid chromatography coupled with tandem mass spectrometry (UHPLC/MS/MS) was used for the identification and the spatial localization of atorvastatin (AT) and its metabolites in rat tissues. Ultrahigh-resolution and high mass accuracy measurements on a matrix-assisted laser desorption/ionization (MALDI)-Orbitrap mass spectrometer allowed better detection of desired analytes in the background of matrix and endogenous compounds. Tandem mass spectra were also used to confirm the identification of detected metabolites in complex matrices. The optimization of sample preparation before imaging experiments included the tissue cryogenic sectioning (thickness 20 μm), the transfer to stainless steel or glass slide, and the selection of suitable matrix and its homogenous deposition on the tissue slice. Thirteen matrices typically used for small molecule analysis, e.g., 2,5-dihydroxybenzoic acid (DHB), 1,5-diaminonaphthalene (DAN), 9-aminoacridine (AA), etc., were investigated for the studied drug and its metabolite detection efficiency in both polarity modes. Particular matrices were scored based on the strength of extracted ion current (EIC), relative ratio of AT molecular adducts, and fragment ions. The matrix deposition on the tissue for the most suitable matrices was done by sublimation to obtain the small crystal size and to avoid local variations in the ionization efficiency. UHPLC/MS profiling of drug metabolites in adjacent tissue slices with the previously optimized extraction was performed in parallel to mass spectrometry imaging (MSI) measurements to obtain more detailed information on metabolites in addition to the spatial information from MSI. The quantitation of atorvastatin in rat liver, serum, and feces was also performed.

ᅟ




Similar content being viewed by others
Abbreviations
- AA:
-
9-Aminoacridine
- AAP:
-
3-Aminoacetophenone
- AP:
-
2-Aminopyrazine
- AQ:
-
3-Aminoquinoline
- AT:
-
Atorvastatin
- BuCHCA:
-
Butylamine salt of α-cyano-4-hydroxycinnamic acid
- CHCA:
-
α-Cyano-4-hydroxycinnamic acid
- CID:
-
Collision-induced dissociation
- DAN:
-
1,5-Diaminonaphthalene
- DHA:
-
2,6-Dihydroxyacetophenone
- DHB:
-
2,5-Dihydroxybenzoic acid
- DMAN:
-
N,N,N′,N′-tetramethyl-1,8-naphthalenediamine
- EIC:
-
Extracted ion current
- ESI:
-
Electrospray ionization
- FD:
-
Fragmentation degree
- HCD:
-
Higher energy collisional induced dissociation
- HPLC:
-
High-performance liquid chromatography
- LOD:
-
Limit of detection
- MALDI:
-
Matrix-assisted laser desorption/ionization
- MBT:
-
2-Mercaptobenzothiazole
- MS:
-
Mass spectrometry
- MSI:
-
Mass spectrometry imaging
- MS/MS:
-
Tandem mass spectrometry
- NaDHB:
-
Sodium 2,5-dihydroxybenzoate
- QTOF:
-
Quadrupole-time-of-flight
- THA:
-
2,4,6-Trihydroxyacetophenone
- UHPLC:
-
Ultrahigh-performance liquid chromatography
- UHPLC/MS/MS:
-
Ultrahigh-performance liquid chromatography-tandem mass spectrometry
- UHRMS:
-
Ultrahigh-resolution mass spectrometry
- UHRMSI:
-
Ultrahigh-resolution mass spectrometry imaging
References
Curran MP (2010) Amlodipine/atorvastatin: a review of its use in the treatment of hypertension and dyslipidaemia and the prevention of cardiovascular disease. Drugs 70:191–213
Nováková L, Satinský D, Solich P (2008) HPLC methods for the determination of simvastatin and atorvastatin. Trac Trends Anal Chem 27:352–367
Hermann M, Christensen H, Reubsaet JLE (2005) Determination of atorvastatin and metabolites in human plasma with solid-phase extraction followed by LC-tandem MS. Anal Bioanal Chem 382:1242–1249
Black AE, Sinz MW, Hayes RN, Woolf T (1998) Metabolism and excretion studies in mouse after single and multiple oral doses of the 3-hydroxy-3-methylglutaryl-CoA reductase inhibitor atorvastatin. Drug Metab Dispos 26:755–763
Jirásko R, Mikysek T, Chagovets V, Vokřál I, Holčapek M (2013) Structural characterization of electrochemically and in vitro biologically generated oxidation products of atorvastatin using UHPLC/MS/MS. Anal Bioanal Chem 405:7181–7193
Kracun M, Kocijan A, Bastarda A, Grahek R, Plavec J, Kocjan D (2009) Isolation and structure determination of oxidative degradation products of atorvastatin. J Pharm Biomed 50:729–736
Shah RP, Kumar V, Singh S (2008) Liquid chromatography/mass spectrometric studies on atorvastatin and its stress degradation products. Rapid Commun Mass Spectrom 22:613–622
Holčapek M, Kolářová L, Nobilis M (2008) High-performance liquid chromatography-tandem mass spectrometry in the identification and determination of phase I and phase II drug metabolites. Anal Bioanal Chem 391:59–78
Norris JL, Caprioli RM (2013) Analysis of tissue specimens by matrix-assisted laser desorption/ionization imaging mass spectrometry in biological and clinical research. Chem Rev 113:2309–2342
Prideaux B, Stoeckli M (2012) Mass spectrometry imaging for drug distribution studies. J Proteome 75:4999–5013
Römpp A, Spengler B (2013) Mass spectrometry imaging with high resolution in mass and space. Histochem Cell Biol 139:759–783
Wu CP, Dill AL, Eberlin LS, Cooks RG, Ifa DR (2013) Mass spectrometry imaging under ambient conditions. Mass Spectrom Rev 32:218–243
Fletcher JS, Lockyer NP, Vickerman JC (2011) Developments in molecular SIMS depth profiling and 3D imaging of biological systems using polyatomic primary ions. Mass Spectrom Rev 30:142–174
Passarelli MK, Winograd N (2011) Lipid imaging with time-of-flight secondary ion mass spectrometry (ToF-SIMS). Biochim Biophys Acta Mol Cell Biol Lipids 1811:976–990
Weidner SM, Falkenhagen J (2009) Imaging mass spectrometry for examining localization of polymeric composition in matrix-assisted laser desorption/ionization samples. Rapid Commun Mass Spectrom 23:653–660
Seeley EH, Caprioli RM (2008) Molecular imaging of proteins in tissues by mass spectrometry. Proc Natl Acad Sci U S A 105:18126–18131
Karas M, Hillenkamp F (1988) Laser desorption ionization of proteins with molecular masses exceeding 10000 daltons. Anal Chem 60:2299–2301
Svatoš A (2010) Mass spectrometric imaging of small molecules. Trends Biotechnol 28:425–434
van Kampen JJA, Burgers PC, de Groot R, Gruters RA, Luider TM (2011) Biomedical application of maldi mass spectrometry for small-molecule analysis. Mass Spectrom Rev 30:101–120
Law KP, Larkin JR (2011) Recent advances in SALDI-MS techniques and their chemical and bioanalytical applications. Anal Bioanal Chem 399:2597–2622
Kawasaki H, Ozawa T, Hisatomi H, Arakawa R (2012) Platinum vapor deposition surface-assisted laser desorption/ionization for imaging mass spectrometry of small molecules. Rapid Commun Mass Spectrom 26:1849–1858
Shroff R, Rulíšek L, Doubský J, Svatoš A (2009) Acid–base-driven matrix-assisted mass spectrometry for targeted metabolomics. Proc Natl Acad Sci U S A 106:10092–10096
Woo HK, Northen TR, Yanes O, Siuzdak G (2008) Nanostructure-initiator mass spectrometry: a protocol for preparing and applying NIMS surfaces for high-sensitivity mass analysis. Nat Protoc 3:1341–1349
Pirman DA, Reich RF, Kiss A, Heeren RMA, Yost RA (2013) Quantitative MALDI tandem mass spectrometric imaging of cocaine from brain tissue with a deuterated internal standard. Anal Chem 85:1081–1089
Cornett DS, Frappier SL, Caprioli RM (2008) MALDI-FTICR imaging mass spectrometry of drugs and metabolites in tissue. Anal Chem 80:5648–5653
Jun JH, Song ZH, Liu ZJ, Nikolau BJ, Yeung ES, Lee YJ (2010) High-spatial and high-mass resolution imaging of surface metabolites of Arabidopsis thaliana by laser desorption-ionization mass spectrometry using colloidal silver. Anal Chem 82:3255–3265
Jungmann JH, Heeren RMA (2012) Emerging technologies in mass spectrometry imaging. J Proteome 75:5077–5092
Hankin JA, Barkley RM, Murphy RC (2007) Sublimation as a method of matrix application for mass spectrometric imaging. J Am Soc Mass Spectrom 18:1646–1652
Thomas A, Charbonneau JL, Fournaise E, Chaurand P (2012) Sublimation of new matrix candidates for high spatial resolution imaging mass spectrometry of lipids: enhanced information in both positive and negative polarities after 1,5-diaminonaphthalene deposition. Anal Chem 84:2048–2054
Acknowledgments
This project was supported by the Czech Science Foundation (Grant No. P206/12/P065). M.H. acknowledges the support of the ERC CZ Project No. LL1302 sponsored by the Ministry of Education, Youth and Sports of the Czech Republic. M.K. acknowledges the support of research project MH CZ-DRO (UHHK, 00179906).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 758 kb)
Rights and permissions
About this article
Cite this article
Jirásko, R., Holčapek, M., Kuneš, M. et al. Distribution study of atorvastatin and its metabolites in rat tissues using combined information from UHPLC/MS and MALDI-Orbitrap-MS imaging. Anal Bioanal Chem 406, 4601–4610 (2014). https://doi.org/10.1007/s00216-014-7880-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-014-7880-y