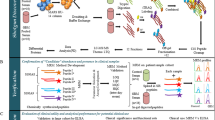
Abstract
The ability to diagnose brain cancer rapidly from serum samples is of great interest; such a diagnosis would allow for rapid testing and time to results providing a responsive diagnostic environment, ability to monitor treatment efficacy, early detection of recurrent tumours and screening techniques. Current methods rely upon subjective, time-consuming tests such as histological grading and are particularly invasive with the diagnostic test requiring hospitalisation of 2–3 days. A rapid diagnostic method based upon serum samples would allow for a relatively non-invasive test and open up the possibility of screening for brain cancer. We report for the first time the use of a Bioplex immunoassay to provide cytokine and angiogenesis factor levels that differ between serum from glioma and non-cancer patients specifically angiopoietin, follistatin, HGF, IL-8, leptin, PDGF-BB and PECAM-1 providing sensitivities and specificities as high as 88 % and 81 %, respectively. We also report, for the first time, the use of serum ATR-FTIR combined with a RBF SVM for the diagnosis of gliomas from non-cancer patients with sensitivities and specificities as high as 87.5 % and 100 %, respectively. We describe the combination of these techniques in an orthogonal diagnostic regime, providing strength to the diagnosis through data combinations, in a rapid diagnostic test within 5 h from serum collection (10 min for ATR-FTIR and 4 h for the Bioplex Immunoassay). This regime has the ability to revolutionise the clinical environment by providing objective measures for diagnosis allowing for increased efficiency with corresponding decreases in mortality, morbidity and economic impact upon the health services.

ᅟ


Similar content being viewed by others
References
Iyer R. V, Golash A., Lea R W, Davies C, Roberts P (2006) Diagnosis and treatment of malignant glioma. Molecular and Cellular Pathology of Cancer Progression and Prognosis: Research Signpost 379–408
Stupp R, Tonn JC, Brada M, Pentheroudakis G (2010) High-grade malignant glioma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 21(5):190–193
Loius DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer W, Kleihues P (2007) The 2007 WHO Classification of Tumours of the Central Nervous System. Acta Neuropathol 114:97–109
XPrize Life Sciences Prize Group (2013) XPRIZE, CA, USA. http://www.xprize.org/prize-development/life-sciences. Accessed 3rd August 2012
Frankel S, Davey Smith G, Donovan J, Neal D (2003) Screening for prostate cancer. Lancet 361(9363):1122–1128
ERSPC Randomized Study of Screening for Prostate Cancer (2009) ERSPC, EU. http://media.erspc-media.org/erspc-background/#psa. Accessed 1st May 2012
Hoffman RM, Smith AY (2011) What we have learned from randomized trials of prostate cancer screening. Asian J Androl 13:369–373
Hessels D, Rittenhouse HG, Schalken JA (2005) Molecular diagnostics in prostate cancer. EAU Update Series 3(4):200–213
Ellis DI, Goodacre R (2006) Metabolic fingerprinting in disease diagnosis: biomedical applications of infrared and Raman spectroscopy. Analyst 131(8):875–885
Byers LA, Kies MS, William WN, Hu J, Lee JJ, El-Naggar A, Papadimitrakopoulou V, Myers JN, Lippman SM, Heymach JV (2008) Profiling of cytokines and angiogenic factors (C/AF) in head and neck (HN) cancer correlates circulating biomarkers with clinical outcomes following induction chemotherapy. Journal of Clinical Oncology, ASCO Annual Meeting Proceedings 26 (#6005)
Rahbari NN, Schmidt T, Falk CS, Hinz U, Herber M, Bork U, Büchler MW, Weitz J, Koch M (2011) Expression and prognostic value of circulating angiogenic cytokines in pancreatic cancer. BMC Cancer 11(286)
Jansen M, Hamer PCD, Witmer AN, Troost D, van Noorden CJF (2004) Current perspectives on antiangiogenesis strategies in the treatment of malignant gliomas. Brain Res Brain Res Rev 45(3):143–163
DaCosta RS, Wilson BC, Macron NC (2005) Optical techniques for the endoscopic detection of dysplastic colonic lesions. Curr Opin Gastroenterol 21:70–79
Shim M, Wong Kee Son LM, Marcon NE, Wilson BC (2000) In vivo near-infrared Raman spectroscopy: demonstration of feasibility during clinical gastrointestinal endoscopy. J Photochem Photobiol 72:146–150
Bakker Schutt TC, Puppels GJ, Kraan YM, Greve J, van der Maas LLJ (1997) Intracellular carotenoid levels measure by Raman microspectroscopy: comparison of lympthocytes form lung cancer patients and healthy individuals. Int J Cancer 74(1):20–25
Sulé-Suso J, Forster A, Zholobenko V, Stone N, El Haj A (2004) Effect of CaCl2 and MgCl2 on Fourier transform infrared spectra of lung cancer cells. Appl Spectrosc 58:61–67
Lyng FM, Faoláin EO, Conroy J, Meade AD, Knief P, Duffy B, Hunter MB, Byrne JM, Kelehan P, Byrne HJ (2007) Vibrational spectroscopy for cervical cancer pathology, from biochemical analysis to diagnostic tool. Exp Mol Pathol 82(2):121–129
Podshyvaloy A, Sahv RK, Mark S, Kantarovich K, Guterman H, Goldstein J, Jagannathan R, Argov S, Mordechai S (2005) Distinction of cervical cancer biopsies by use of infrared microspectroscopy and probabilistic neural networks. Applied Optics 44(1–2):3725–3734
Koljenović S, Choo-Smith LP, Schut TC, Kros JM, Berge HJ, Puppels GJ (2002) Discriminating vital tumour from necrotic tissue in human glioblastoma tissue by Raman spectroscopy. Lab Invest 82(10):1265–1277
Beleites C, Steiner G, Sowa MG, Baumgartner R, Sobottka S, Schackert G, Salzer R (2005) Classification of human gliomas by infrared imaging spectroscopy and chemometric image processing. Vibrational Spectroscopy 38:143–149
Manoharan R, Shafer KE, Perelman LT, Wu J, Chen K, Deinum G, Fitzmaurice M, Myles J, Crowe J, Dasari RR, Feld MS (1998) Raman spectroscopy and fluorescence photon migration for breast cancer diagnosis and imaging. Photochemistry and Photobiology 67(1):15–22
Dukor RK, Liebman MN, Johnson BL (1998) A new, non-destructive method for analysis of clinical samples with FT-IR microspectroscopy. Breast cancer tissue as an example. Cell Molecular Biology 44(1):211–217
Baker MJ, Gazi E, Brown MD, Shanks JH, Clarke NW, Gardner P (2009) Investigating FTIR based histopathology for the diagnosis of prostate cancer. Journal of Biophotonics 2(1–2):104–113
Malins DC, Polissar NL, Gunselman SJ (1997) Models of DNA structure achieve almost perfect discrimination between normal prostate, benign prostatic hyperplasia (BPH), and adenocarcinoma and have a high potential for predicting BPH and prostate cancer. Proceedings of the National Academy of Sciences of the United States of America 94(1):259–264
Fernandez DC, Rohit B, Hewitt SM, Levin IW (2005) Infrared spectroscopic imaging for histopathologic recognition. Nature Biotechnology 23:469–474
Isabelle M, Stone N, Barr H, Vipond M, Sheperd N, Rogers K (2008) Lymph node pathology using optical spectroscopy in cancer diagnostics. Spectroscopy 22:97–104
Romeo MJ, Diem M (2005) Infrared spectral imaging of lymph nodes: strategies for analysis and artefact reduction. Vibrational Spectroscopy 38(1–2):115–119
Wolcott M J, Schoepp R J, Norwood D A, Shoemaker D R (2007) Rapid infectious disease diagnostic assays, global infectious disease surveillance and detection: assessing the challenges—Find Solutions Workshop Summary 165–177
Henchel EA, Teska JD, Ludwig GV, Shoemaker DR, Ezzell JW (2001) Current laboratory methods for biological threat agent identification. Clin Lab Med 21(3):661–678
Chang C C, Lin C J (2011) LIBSVM: a library for support vector machines. ACM TIST 2.Doi:10.1145/1961189.1961199
Baker MJ, Clarke CC, Nicholson JM, Lyng FM, Byrne HJ, Hart CA, Brown MD, Clarke NW, Gardner P (2010) An investigation of the RWPE prostate derived family of cell lines using FTIR spectroscopy. Analyst 135(5):887–894
Sattlecker M, Bessant C, Smith J, Stone N (2010) Investigation of support vector machines and Raman spectroscopy for lymph node diagnostics. Analyst 135(5):895–901
Bouloumie A, Schini-Kerth VB, Busse R (1999) Vascular endothelial growth factor up-regulates nitric oxide synthase expression in endothelial cells. Cardiovascular Research 41(3):773–780
Reiss Y, Machein MR, Plate KH (2005) The role of angiopoietins during angiogenesis in gliomas. Brain Pathology 15(4):311–317
Dunn IF, Heese O, Black PM (2000) Growth factors in glioma angiogenesis: FGFs, PDGF, EGF, and TGFs. Journal of Neuro-Oncology 50(1–2):121–137
Becker M, Mertsch S, Schuch R, Paulus W, Senner V (2010) Follistatin promotes migration and proliferation of glioma cells. European Journal of Cell Biology 89:17–17
Liu KZ, Shaw AR, Man A, Dembinski TC, Mantsch HH (2002) Reagent-free, simultaneous determination of serum cholesterol in HDL and LDL by infrared spectroscopy. Clinical Chemistry 48(3):400–506
Baker MJ, Gazi E, Brown MD, Shanks JH, Gardner P, Clarke NW (2008) FTIR-based spectroscopic analysis in the identification of clinically aggressive prostate cancer. British Journal of Cancer 99:1859–1866
Diessel E, Willman S, Kamphaus P, Kurte R, Damm U, Heise HM (2004) Glucose quantification in dried-down nanolitre samples using mid-infrared attenuate total reflection spectroscopy. Applied Spectroscopy 58(4):442–450
Shaw RA, Mantsch HH (2010) Infrared spectroscopy in clinical and diagnostic analysis. Encyclopaedia of Analytical Chemistry. Wiley, Chichester
Goormaghtigh E, Raussens V, Ruysschaert JM (1999) Attenuated total reflection infrared spectroscopy of proteins and lipids in biological membranes. Biochimica et Biophysica Acta 1422:105–185
Rohleder D, Kocherscheidt G, Gerber K, Kiefer W, Kohler W, Mock J, Petrich W (2005) Comparison of mid-infrared and Raman spectroscopy in the quantitative analysis of serum. Journal of Biomedical Optics 10(3):031108
Naumann D (2001) FT-infrared and FT-Raman spectroscopy in biomedical research. Applied Spectroscopy Reviews 36(2–3):239–298
Meade A, Lyng F, Knief P, Byrne H (2007) Growth substrate induced functional changes elucidated by FTIR and Raman spectroscopy in in-vitro cultured human keratinocytes. Analytical and Bioanalytical Chemistry 387(5):1717–1728
Bellisola G, Sorio C (2012) Infrared spectroscopy and microscopy in cancer research and diagnosis. American Journal of Cancer Research 2(1):1–21
Tamm LK, Tatulian SA (1997) Infrared spectroscopy of proteins and peptides in lipid bilayers. Quarterly Reviews of Biophysics 30(4):365–429
Petrich W, Lewandrowski KB, Muhlestein JB, Hammond ME, Januzzi JL, Lewandrowski EL, Pearson RR, Dolenko B, Fruh J, Haass M, Hirschl MM, Kohler W, Mischler R, Mocks J, Ordonez-Llanos J, Quarder O, Somorjai R, Staib A, Sylven C, Werner G, Zerback R (2009) Potential of mid-infrared spectroscopy to aid the triage of patients with acute chest pain. Analyst 134(6):1092–1098
Gajjar K, Trevisan J, Owens G, Keating P, Wood N, Stringfellow H, Martin-Hirsch P, Martin F (2013) Fourier-transform infrared spectroscopy coupled with a classification machine for the analysis of blood plasma or serum: a novel diagnostic approach for ovarian cancer. Analyst. doi:10.1039/c3an36654e
Acknowledgements
The authors acknowledge the support and funding provided by the Brain Tumour North West collaborations (http://www.btnw.org.uk/), the Sydney Driscoll Neuroscience Foundation, School of Forensic and Investigative Sciences, School of Pharmacy and Biomedical Sciences and the Centre for Materials Science at the University of Central Lancashire and to the Association of British Spectroscopists for a travel grant for MJB. The authors also acknowledge Dr Joseph M Hayes for his protein image and thoughtful discussions.
Competing interests
The authors declare no competing interests.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Hands, J.R., Abel, P., Ashton, K. et al. Investigating the rapid diagnosis of gliomas from serum samples using infrared spectroscopy and cytokine and angiogenesis factors. Anal Bioanal Chem 405, 7347–7355 (2013). https://doi.org/10.1007/s00216-013-7163-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-013-7163-z