Abstract
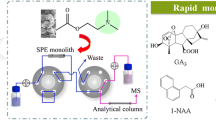
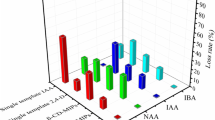
Cytokinins (CTKs) are a class of growth-regulating hormones involved in various physiological and developmental processes. More novel analytical methods for the accurate identification and quantitative determination of trace CTKs in plants have been desired to better elucidate the roles of CTKs. In this work, a novel method based on monolithic molecularly imprinted solid-phase extraction followed by liquid chromatography–electrospray tandem mass spectrometry (mMI-SPE-LC-MS/MS) was developed for accurate determination of four CTKs in plant samples. The molecularly imprinted polymer monolith was prepared by using kinetin as the template in syringes and exhibited specific recognition ability for the four CTKs in comparison with that of non-imprinted polymer monolith. Several factors affecting the extraction performance of mMI-SPE, including the pH of loading sample solution, the nature and volume of elution solvent, the flow rate of sample loading, and sample volume, were investigated, respectively. Under the optimized conditions, the proposed mMI-SPE-LC-MS/MS method was successfully applied in the selective extraction and determination of four CTKs in plant tissues, and it offers detection limits (S/N = 3) of 104, 113, 130, and 89 pg/mL and mean recoveries of 85.9%, 79.3%, 73.5%, and 70.1% for kinetin, kinetin glucoside, trans-zeatin, and meta-topolin (mT), respectively, with the corresponding RSDs less than 15%.

Trace determination of cytokinins in different plant samples by monolithic molecularly imprinted solid-phase extraction and liquid chromatography–mass spectrometry









Similar content being viewed by others
References
Werner IT, Motyka V, Strnad M, Schmülling T (2001) Proc Natl Acad Sci USA 98:10487–10492
Mok DWS, Mok MC (2001) Annu Rev Plant Physiol Plant Mol Biol 52:89–112
Ferreira FJ, Kieber JJ (2005) Curr Opin Plant Biol 8:518–525
Sakakibara H (2006) Annu Rev Plant Biol 57:431–449
Kyozuka J (2007) Curr Opin Plant Biol 10:442–446
Werner T, Schmülling T (2009) Curr Opin Plant Biol 12:527–538
Choi J, Choi D, Lee S, Ryu C, Hwang I (2011) Trends Plant Sci 16:388–394
Kamada-Nobusada T, Sakakibara H (2009) Phytochem 70:444–449
Perilli S, Moubayidin L, Sabatini S (2010) Curr Opin Plant Biol 13:21–26
Barciszewski J, Massino F, Clark BFC (2007) Int J Biol Macromol 40:182–192
Pan XQ, Wang XM (2009) J Chromatogr B 877:2806–2813
Liang Y, Zhao MP, Liu HW (2009) Chin J Anal Chem 37:1232–1339
Tarkowski P, Ge L, Yong JWH, Tan SN (2009) Trends Anal Chem 28:323–335
Du FY, Ruan GH, Liu HW (2012) Anal Bioanal Chem 403:55–74
Du FY, Bai Y, Bai Y, Liu HW (2010) Anal Chem 82:9374–9383
Bai Y, Du FY, Bai Y, Liu HW (2010) Anal Methods 2:1867–1873
Liang Y, Zhu XC, Zhao MP, Liu HW (2012) Methods 56:174–179
Kojima M, Kamada-Nobusada T, Komatsu H, Takei K, Kuroha T, Mizutani M, Ashikari M, Ueguchi-Tanaka M, Matsuoka M, Suzuki K, Sakakibara H (2009) Plant Cell Physiol 50:1201–1214
Chen WQ, Gai Y, Liu SC, Wang RX, Jiang XN (2010) J Integr Plant Biol 52(10):925–932
Novák O, Hauserová E, Amakorová P, Doležal K, Strnad M (2008) Phytochem 69:2214–2224
Caro E, Marcé RM, Borrull F, Cormack PAG, Sherrington DC (2006) Trends Anal Chem 25:143–154
Tamayo FG, Turiel E, Martín-Esteban A (2007) J Chromatogr A 1152:32–40
Harun N, Anderson RA, Cormack PAG (2010) Anal Bioanal Chem 396:2449–2459
Tse Sum Bui B, Haupt K (2010) Anal Bioanal Chem 398:2481–2492
Beltran A, Borrull F, Cormack PAG, Marcé RM (2010) Trends Anal Chem 29:1363–1375
Chen LX, Xu SF, Li JH (2011) Chem Soc Rev 40:2922–2942
Kugimiya A, Takeuchi T (1999) Anal Chim Acta 395:251–255
Chen CB, Chen YJ, Zhou J, Wu CH (2006) Anal Chim Acta 569:58–65
Zhang Y, Li YW, Hu YL, Li GK, Chen YQ (2010) J Chromatogr A 1217:7337–7344
Hu YL, Li YW, Zhang Y, Li GK, Chen YQ (2011) Anal Bioanal Chem 399:3367–3374
Zheng MM, Gong R, Zhao X, Feng YQ (2010) J Chromatogr A 1217:2075–2081
Zhou JF, Ma C, Zhou S, Ma PL, Chen FR, Qi Y, Chen HX (2010) J Chromatogr A 1217:7478–7483
Hoyerová K, Gaudinová A, Malbeck J, Dobrev PI, Kocábek T, Šolcová B, Ková AT, Kamínek M (2006) Phytochem 67:1151–1159
Pichon V (2007) J Chromatogr A 1152:41–53
Djozan D, Baheri T (2007) J Chromatogr A 1166:16–23
Horgan R, Scott IM (1987) In: Rivier L, Crozier A (eds) The principles and practice of plant hormone analysis, chapter 5. Academic, London, pp 303–365
Pacáková V, Štulík K, Vlasáková V, Březinová A (1997) J Chromatogr A 764:331–335
Dobrev PI, Kamínek M (2002) J Chromatogr A 950:21–29
Barták P, Pěchová D, Tarkowski P, Bednář P, Kotouček M, Stránský Z, Vespalec R (2000) Anal Chim Acta 421:221–229
Ye L, Yu YH, Mosbach K (2001) Analyst 126:760–765
Cela-Pérez MC, Castro-López MM, Lasagabáster-Latorre A, López-Vilariño JM, González-Rodríguez MV, Barral-Losada LF (2011) Anal Chim Acta 706:275–284
Zhu GF, Fan J, Gao YB, Gao X, Wang JJ (2011) Talanta 84:1124–1132
Acknowledgments
This research was supported by the China Postdoctoral Science Foundation (Grant 201003008), by the National Natural Science Foundation of China (Grant 21065003) and the Doctoral Research Foundation of Guilin University of Technology (Grant 002401003314).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 311 kb)
Rights and permissions
About this article
Cite this article
Du, F., Ruan, G., Liang, S. et al. Monolithic molecularly imprinted solid-phase extraction for the selective determination of trace cytokinins in plant samples with liquid chromatography–electrospray tandem mass spectrometry. Anal Bioanal Chem 404, 489–501 (2012). https://doi.org/10.1007/s00216-012-6131-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-012-6131-3